Global Eclinical Solutions Market
市场规模(十亿美元)
CAGR :
% 
 USD
9.36 Billion
USD
25.79 Billion
2024
2032
USD
9.36 Billion
USD
25.79 Billion
2024
2032
| 2025 –2032 | |
| USD 9.36 Billion | |
| USD 25.79 Billion | |
|
|
|
|
全球電子臨床解決方案市場細分,按產品(電子數據採集和臨床試驗數據管理系統、臨床試驗管理系統、臨床分析平台、護理協調醫療記錄 (CCMR)、隨機化和試驗供應管理、臨床數據集成平台、電子臨床結果評估解決方案、安全解決方案、電子試驗主文件系統、監管信息管理解決方案等)、交付模式(Web 許可(Web )解決方案為基於企業的客戶管理)、基於企業管理系統(Web 許可解決方案)解決方案)、臨床試驗階段(第一階段、第二階段、第三階段和第四階段)、組織規模(小型、中型和大型)、用戶設備(台式機、平板電腦、手持 PDA 設備、智慧型手機等)、最終用戶(製藥和生物製藥公司、合約研究組織、諮詢服務公司、醫療器材製造商、醫院和學術研究機構)- 行業趨勢和預測到 2032 年
電子臨床解決方案市場規模
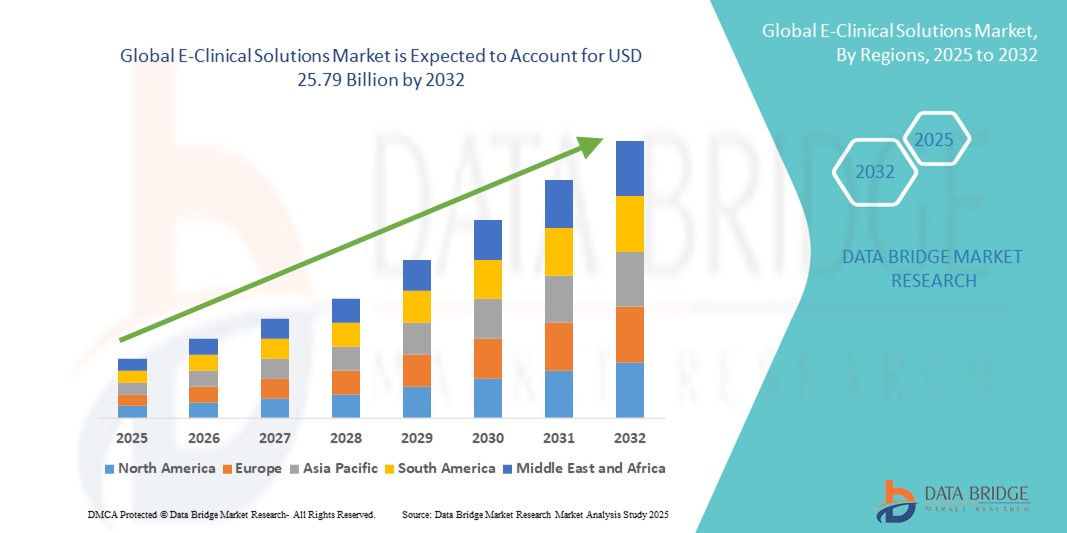
- 2024 年全球電子臨床解決方案市場規模為93.6 億美元 ,預計 到 2032 年將達到 257.9 億美元,預測期內 複合年增長率為 13.50%
- 市場成長主要得益於臨床研究和醫療保健系統中數位技術的日益普及和技術進步,從而增強了製藥和生物技術公司的數據管理、試驗效率和即時決策能力
- 此外,對安全、使用者友好且整合的臨床試驗平台的需求不斷增長,使得電子臨床解決方案成為資料收集、監控和分析的首選。這些因素正在加速電子臨床解決方案的普及,顯著推動產業成長。
電子臨床解決方案市場分析
- 電子臨床解決方案涵蓋 CTMS、EDC、eCOA 和 RTSM 等數位平台,因其在資料管理、營運效率和法規遵循方面發揮的作用,在臨床試驗中正變得越來越重要。簡化複雜試驗流程並支援分散式和虛擬試驗模式的迫切需求推動了市場的擴張。
- 電子臨床解決方案需求的不斷增長主要受到以下因素的推動:全球臨床試驗數量的增加、製藥和生物技術公司研發投資的增加、向基於雲端/行動系統的轉變,以及透過自動化和數據整合加快藥物開發時間表的必要性
- 北美在電子臨床解決方案市場佔據主導地位,2024 年其收入份額最高,達 49.38%。這得歸功於其強大的醫療基礎設施、成熟的製藥和生物技術產業,以及鼓勵數位化應用的良好監管框架。這種主導地位尤其得益於美國的先進應用,2024 年美國佔據了全球市場約 41.4% 的份額。
- 預計亞太地區電子臨床解決方案市場將實現強勁成長,預計複合年增長率為 11.8%,這得益於臨床試驗外包的增加、醫療保健和研發基礎設施的擴大以及中國、印度、日本和韓國等國家試驗活動的增加
- 大型企業部門在 2024 年佔據電子臨床解決方案市場的 63.4% 份額,這反映了它們在研發、全球臨床試驗營運方面的大量投資,以及對管理法規遵從性、複雜工作流程和多國研究協調的完全整合平台的迫切需求
報告範圍和電子臨床解決方案市場細分
|
屬性 |
電子臨床解決方案關鍵市場洞察 |
|
涵蓋的領域 |
|
|
覆蓋國家 |
北美洲
歐洲
亞太
中東和非洲
南美洲
|
|
主要市場參與者 |
|
|
市場機會 |
|
|
加值資料資訊集 |
除了對市場價值、成長率、細分、地理覆蓋範圍和主要參與者等市場情景的洞察之外,Data Bridge Market Research 策劃的市場報告還包括深入的專家分析、定價分析、品牌份額分析、消費者調查、人口統計分析、供應鏈分析、價值鏈分析、原材料/消耗品概述、供應商選擇標準、PESTLE 分析、波特分析和監管框架。 |
電子臨床解決方案市場趨勢
透過整合電子臨床平台提高便利性
- 全球電子臨床解決方案市場的一個重要且加速發展的趨勢是臨床試驗管理系統、電子資料收集 (EDC) 和臨床試驗管理系統 (CTMS) 不斷深入整合,形成統一的數位平台。這種融合顯著提升了整個臨床研究生態系統的營運效率、數據品質和決策能力。
- 例如,現代電子臨床平台將EDC與臨床試驗管理功能無縫集成,使申辦者和合約研究組織 (CRO) 能夠透過單一介面即時監控試驗進度、管理患者入組並確保合規性。同樣,先進的解決方案現已整合隨機化和試驗供應管理 (RTSM),從而減少延誤並改善整體試驗協調。
- 電子臨床解決方案的整合進一步實現了資料輸入、進階資料驗證和即時報告的自動化。例如,平台越來越多地整合自適應試驗設計功能和自動查詢解決系統,從而最大限度地減少人工幹預,並提高患者資料處理的準確性。此外,先進的分析和預測模型功能使試驗管理人員能夠預測患者招募率,並提前發現潛在的瓶頸。
- 電子臨床平台與電子健康記錄 (EHR)、實驗室資訊系統 (LIS) 和其他數位健康生態系統的無縫集成,有助於集中存取各種臨床資料來源。透過統一的平台,研究人員可以管理病患記錄、不良事件報告、試驗文件和監管提交,從而創建精簡且合規的臨床試驗流程。
- 這種朝向更智慧、互聯互通和用戶友好的電子臨床生態系統邁進的趨勢,正在從根本上重塑人們對臨床試驗管理的期望。因此,Medidata、Oracle Health Sciences 和 Veeva Systems 等公司正在開發下一代電子臨床解決方案,該解決方案具有增強的互通性、基於雲端的部署模型和行動可訪問性,以支援全球和分散的臨床試驗。
- 隨著利害關係人越來越重視營運效率、法規遵循和加快新療法的上市時間,製藥、生物技術和醫療器材領域對提供整合功能的電子臨床解決方案的需求正在迅速增長
電子臨床解決方案市場動態
司機
對高效臨床試驗管理的需求日益增長,數位化解決方案的採用也日益增多
- 臨床試驗的複雜性和成本日益增加,加上正在開發的候選藥物和生物製劑數量不斷增加,推動了對先進電子臨床解決方案的需求大幅增加。這些平台能夠實現高效的數據管理、無縫的患者參與以及跨地域試驗點的法規合規性。
- 例如,2024年6月,Medidata(達梭系統)在其人工智慧驅動的去中心化臨床試驗 (DCT) 平台中推出了增強功能,旨在改善患者招募和即時試驗監控。此類技術進步正在加速製藥和生物技術產業對電子臨床平台的採用。
- 隨著申辦方和合約研究組織 (CRO) 努力縮短試驗時間並改善決策,電子資料擷取 (EDC)、臨床試驗管理系統 (CTMS)、eCOA 和遠端監控工具等解決方案正變得不可或缺。這些系統提供自動化、數據準確性和即時洞察,從而確保營運效率。
- 此外,以患者為中心的試驗日益增長,混合和分散試驗模式的擴展,使得電子臨床平台成為臨床研究生態系統不可或缺的一部分,從而提高患者的保留率和依從性
- 進階分析、雲端運算和基於行動的介面的整合進一步增強了電子臨床解決方案的價值主張,使其在滿足對更快、更安全、更有效率的藥物開發流程日益增長的需求方面發揮關鍵作用
克制/挑戰
對資料安全、整合複雜性和高實施成本的擔憂
- 對資料隱私和安全漏洞的擔憂是電子臨床解決方案市場發展的一大限制。由於這些系統處理敏感的患者和試驗數據,它們仍然容易受到網路安全威脅、未經授權的存取和潛在的數據洩露,這引起了申辦方、合約研究組織 (CRO) 和監管機構的擔憂。
- 例如,醫療保健和臨床研究機構遭受網路攻擊的事件引起了人們的廣泛關注,他們意識到,在沒有強有力的保障措施的情況下採用基於雲端的數位平台存在風險。
- 透過進階加密、多因素身份驗證、GDPR/HIPAA合規性以及定期系統審核來解決這些問題,對於維護用戶信任至關重要。 Oracle Health Sciences和Veeva Systems等供應商強調其資料保護能力,以讓利害關係人放心。
- 此外,將電子臨床解決方案與現有遺留系統和多樣化試驗管理工作流程相結合的挑戰往往會延遲採用,尤其是在中小型 CRO 中
- 高昂的初始實施成本(包括許可、客製化和人員培訓)仍然是採用的另一個主要障礙,尤其是在發展中地區或預算有限的試驗申辦者。雖然基於 SaaS 的訂閱模式正在降低前期成本,但具備 AI、分析和互通性功能的先進平台仍需要支付高昂的費用。
- 透過經濟實惠的模組化解決方案、更好的互通性標準和更強大的網路安全框架克服這些障礙對於實現電子臨床解決方案的全球持續採用至關重要
電子臨床解決方案市場範圍
市場根據產品、交付模式、臨床試驗階段、組織規模、使用者設備和最終用戶進行細分。
- 按產品
根據產品,電子臨床解決方案市場細分為電子資料收集和臨床試驗資料管理系統、臨床試驗管理系統、臨床分析平台、護理協調醫療記錄 (CCMR)、隨機化和試驗供應管理、臨床資料整合平台、電子臨床結果評估解決方案、安全解決方案、電子試驗主文件系統、監管資訊管理解決方案等。資料管理系統細分市場在 2024 年佔據市場主導地位,收入份額最高,達到 28.6%,這得益於其在高效收集、清理和管理臨床試驗數據方面的核心作用。這些系統對於維護資料完整性、法規遵循和準確報告至關重要,使其成為小型和大型臨床試驗中不可或缺的一部分。多地點和全球研究中試驗資料量的不斷增長,對強大的資料管理的需求也日益增長。與電子健康記錄 (EHR) 和其他試驗平台的整合進一步增強了它們的應用。先進的分析和即時監控功能也使數據管理系統成為高效臨床營運的支柱。
預計臨床分析平台領域在2025年至2032年間將實現最快的複合年增長率,達到22.1%,這得益於臨床研究中人工智慧分析、機器學習和預測模型的日益普及。這些平台使利害關係人能夠從複雜的試驗資料集中提取可操作的洞察,優化患者招募,並即時改善試驗結果。分析解決方案還支援基於風險的監測,識別方案偏差,並增強法規遵循。整合包括eCOA、穿戴式裝置和實驗室系統在內的多種資料來源的能力推動了其成長。先進的視覺化工具和儀表板可協助試驗管理人員快速做出明智的決策。對自適應和精準醫學試驗的日益關注進一步加速了臨床分析平台的普及。
- 按交付方式
根據交付模式,電子臨床解決方案市場細分為網路託管(隨選)解決方案、授權企業(本地)解決方案和基於雲端(軟體即服務/SAAS)的解決方案。網路託管(按需)解決方案憑藉其可擴展性、成本效益和易於部署的特點,在2024年佔據了41.3%的最大市場份額。這些解決方案允許使用者遠端存取試驗資料和管理工具,無需複雜的IT基礎設施,尤其有利於中小型研究機構。易於與現有IT系統整合且維護需求極低,進一步增強了其應用。能夠根據試驗需求靈活擴展資源也是該細分市場受到青睞的原因之一。集中式資料存取和即時監控功能可提高營運效率。
預計2025年至2032年,基於雲端(SaaS)的解決方案細分市場將以21.4%的複合年增長率保持最快增長,這得益於分散式和混合式臨床試驗模式的日益普及。雲端平台提供跨多個地點和國家/地區安全、即時的試驗資料訪問,從而促進申辦方、合約研究組織(CRO)和臨床研究中心之間的更有效率協作。自動化工作流程、數據標準化和法規遵循功能支援高效的試驗營運。雲端技術的採用還可以降低IT基礎設施成本並加快部署進度。雲端平台整合高級分析和行動存取功能,加強了營運監管。監管機構對數位化試驗管理解決方案的鼓勵力度不斷加大,進一步加速了市場成長。
- 按臨床試驗階段
依臨床試驗階段,電子臨床解決方案市場細分為I期、II期、III期及IV期。 III期市場在2024年佔據了最大的收入份額,達到46.7%,這得益於複雜的試驗方案、龐大的患者群體以及後期研究中嚴格的監管要求。 III期試驗會產生大量數據,因此需要先進的電子臨床解決方案來實現高效的數據管理、病患監測和法規遵循。試驗管理系統、分析平台和eCOA工具的整合可提高操作準確性。申辦者高度依賴III期系統來確保研究及時完成並最大限度地減少延誤。對全球多中心試驗的日益關注鞏固了該市場的主導地位。
預計2025年至2032年,II期臨床試驗領域將以19.6%的複合年增長率保持最快增長,這得益於腫瘤學、標靶治療和精準醫療領域中期試驗數量的不斷增長。 II期臨床試驗需要自適應設計、詳細的患者監測以及來自多個來源的無縫資料整合。電子臨床解決方案簡化了這些流程,提供即時洞察並提高試驗效率。增強的患者參與工具以及與穿戴式裝置的整合加速了其應用。監管機構對病人安全和數據準確性的重視也促進了該領域的成長。中期試驗對快速決策的需求進一步刺激了對複雜電子臨床平台的需求。
- 按組織規模
根據組織規模,電子臨床解決方案市場可細分為中小型企業 (SME) 和大型企業。大型企業在 2024 年佔據 63.4% 的市場份額,佔據主導地位,這反映了其廣泛的研發投入、全球試驗營運以及對全面整合平台的需求,以管理法規遵從性、多國研究和複雜的工作流程。大型企業利用電子臨床解決方案來減少操作錯誤、簡化資料收集並確保整個試驗的標準化流程。集中監控和分析支援大規模決策。與 EHR 和其他醫院系統的整合可提高效率。雲端運算和 Web 託管解決方案的廣泛採用進一步鞏固了其主導地位。
預計中小企業市場在2025年至2032年間將實現最快的複合年增長率,達到20.2%,這得益於可擴展、經濟實惠且基於雲端的電子臨床解決方案的日益普及。中小企業受益於自動化工作流程、遠端存取和降低的基礎設施成本,從而能夠利用有限的資源進行高效的試驗。 SaaS平台的快速部署加速了其應用。這些解決方案無需巨額IT投資即可實現合規性。在藥物開發時間方面與大型企業競爭的需求進一步推動了市場成長。與行動和分析工具的整合可提高小型企業的營運效率。
- 按用戶設備
根據用戶設備,電子臨床解決方案市場細分為桌上型電腦、平板電腦、手持PDA設備、智慧型手機等。 2024年,桌上型電腦市場佔最大份額,達到37.9%,因為桌上型電腦仍然是試驗管理人員、資料分析師和臨床協調員執行複雜資料輸入、分析和監管報告的主要工具。其強大的運算能力、大螢幕顯示介面和安全的環境使其成為管理敏感試驗資料的理想選擇。桌上型電腦還支援與多個試驗平台和電子病歷(EHR)系統整合。可靠性和穩定性對於處理大規模、多地點的試驗操作至關重要。
預計2025年至2032年,智慧型手機市場將以23.5%的複合年增長率保持最快成長,這得益於相容行動裝置的eCOA解決方案、患者參與應用程式和遠端監控工具的普及。智慧型手機可以提供即時試驗更新,提高患者依從性,並使臨床醫生、監查員和申辦者能夠隨時隨地存取資料。行動裝置的可存取性增強了分散式和混合式試驗模式。與雲端平台和穿戴式裝置的整合支援持續資料收集。對靈活、以患者為中心的試驗管理的需求日益增長,加速了基於智慧型手機的解決方案的普及。智慧型手機還能減少數據報告和監控的延遲,進而提高營運效率。
- 按最終用戶
根據最終用戶,電子臨床解決方案市場細分為製藥和生物製藥公司、合約研究組織 (CRO)、諮詢服務公司、醫療器材製造商、醫院和學術研究機構。製藥和生物製藥公司佔據主導地位,2024 年的市場份額為 51.8%,這得益於其採用電子臨床平台來管理複雜的多地點試驗、確保法規遵從性並加快藥物開發進度。大型製藥公司依靠這些解決方案進行整合試驗管理、即時數據存取和標準化工作流程,以減少錯誤並提高效率。
預計合約研究組織 (CRO) 領域在 2025 年至 2032 年期間的複合年增長率將達到 21.9%,為行業最高。這得益於臨床試驗營運外包趨勢的日益增長以及對經濟高效的試驗管理的需求。 CRO 使用電子臨床平台來管理多個客戶試驗、簡化工作流程、優化資源並確保全球研究的準確報告。即時數據存取、集中監控以及利害關係人之間更佳的協作將提升試驗效率。分散式試驗和混合模式的日益普及將加速該領域的成長。此外,該領域還受益於基於人工智慧的分析和行動監控解決方案等技術創新。
電子臨床解決方案市場區域分析
- 北美在電子臨床解決方案市場佔據主導地位,2024 年其收入份額最大,為 49.38%,這得益於其強大的醫療保健基礎設施、成熟的製藥和生物技術行業以及鼓勵數位化應用的有利監管框架
- 這種主導地位尤其受到美國先進應用的推動,這得益於電子資料擷取系統、臨床試驗管理平台和分析驅動解決方案的持續實施
- 該地區對法規遵從性、病人安全和運營效率的重視繼續推動醫院、CRO 和研究機構廣泛採用電子臨床平台
美國電子臨床解決方案市場洞察
美國電子臨床解決方案市場佔據了北美最大的收入份額,這得益於數位化臨床試驗解決方案的廣泛採用、試驗營運外包的不斷增加以及對臨床研究基礎設施的大力投資。雲端平台、電子臨床結果評估 (eCOA) 工具和整合試驗管理系統等先進技術正在加快患者招募速度、提高資料管理效率並提高試驗透明度。對分散式和混合式試驗的日益關注進一步推動了製藥和生物技術公司對綜合電子臨床解決方案的採用。
歐洲電子臨床解決方案市場洞察
預計歐洲電子臨床解決方案市場在整個預測期內將以顯著的複合年增長率擴張,主要驅動力在於臨床試驗營運外包的增加、監管機構對數位醫療技術的支持以及先進試驗管理系統的採用。由於強大的醫藥研發發生態系統、日益複雜的試驗以及對整合式即時試驗管理解決方案的需求,德國、法國和英國等國家正經歷顯著成長。歐洲利益相關者正在利用基於雲端、分析驅動且相容行動裝置的電子臨床平台來提高營運效率並確保多地點研究的合規性。
英國電子臨床解決方案市場洞察
英國電子臨床解決方案市場預計將在預測期內實現顯著的複合年增長率,這得益於英國強大的臨床研究基礎設施和數位化試驗平台的日益普及。申辦者和合約研究組織 (CRO) 正在投資整合式電子臨床解決方案,以簡化患者監測、集中管理試驗數據並加速藥物開發流程。此外,分散式和混合式試驗模式的興起也推動了基於雲端和行動裝置的系統的採用,從而支援即時決策和高效的試驗監督。
德國電子臨床解決方案市場洞察
預計在預測期內,德國電子臨床解決方案市場將以可觀的複合年增長率擴張,這得益於臨床試驗活動的不斷增長、嚴格的監管標準以及先進數位平台的日益普及。德國強大的製藥和生物技術產業,加上其對創新和數據完整性的重視,推動了綜合電子臨床解決方案的使用,包括電子數據採集、臨床試驗管理系統和整合分析平台。該地區的學術研究機構、合約研究組織 (CRO) 和製藥組織正經歷著強勁的成長。
亞太地區電子臨床解決方案市場洞察
受臨床試驗外包業務的不斷增長、醫療保健和研發基礎設施的不斷擴張以及中國、印度、日本和韓國等國家試驗活動的不斷增加的推動,亞太地區電子臨床解決方案市場在預測期內有望實現11.8%的強勁複合年增長率。該地區對基於雲端的試驗管理系統、分析平台以及相容行動裝置的電子臨床解決方案的需求日益增長,這些解決方案旨在支援分散式研究、優化患者招募並增強數據收集。亞太地區製藥和生物技術公司不斷增加的投資,進一步推動了先進電子臨床技術的普及。
日本電子臨床解決方案市場洞察
日本電子臨床解決方案市場正蓬勃發展,得益於其先進的醫療基礎設施、高品質的臨床研究標準以及日益普及的技術驅動型試驗解決方案。申辦者和合約研究組織 (CRO) 正在利用整合試驗管理平台、eCOA 工具和雲端系統來改善病患監測、提高試驗效率並確保合規性。精準醫療和複雜臨床研究的日益增長的關注度正在進一步加速市場成長。
中國電子臨床解決方案市場洞察
2024年,中國電子臨床解決方案市場佔據亞太地區最大市場收入份額,這得益於中國臨床試驗活動的不斷擴展、強勁的醫藥研發投入以及對數位醫療技術日益增強的監管支持。中國正在快速採用電子資料收集系統、臨床試驗管理解決方案和即時分析平台,以支援多中心試驗、改善病患招募並簡化試驗流程。政府推動醫療保健和生命科學領域數位化的措施進一步加強了該地區的市場擴張。
電子臨床解決方案市場份額
電子臨床解決方案產業主要由知名公司主導,包括:
- 甲骨文(美國)
- Signant Health(美國)
- MaxisIT(美國)
- Parexel International Corporation(美國)
- 達梭系統(法國)
- Clario(美國)
- Mednet(美國)
- OpenClinica, LLC(美國)
- 4G臨床(美國)
- Veeva Systems(美國)
- Saama Technologies, LLC(美國)
- Anju(美國)
- 卡斯特(荷蘭)
- Medrio, Inc.(美國)
- ArisGlobal(美國)
- Merative(美國)
- Advarra(美國)
- eClinical Solutions, LLC(美國)
- Y-Prime LLC(美國)
- RealTime Software Solutions LLC(美國)
- Quretec(愛沙尼亞)
- 研究經理(荷蘭)
- Datatrack Int.(荷蘭)
- IQVIA Inc.(美國)
全球電子臨床解決方案市場的最新發展
- 2021年12月,甲骨文公司宣布以約283億美元現金收購領先的電子健康記錄 (EHR) 系統供應商 Cerner Corporation。這項策略性措施旨在增強甲骨文在醫療保健領域的影響力,並加速臨床環境的數位轉型。
- 2025年7月,知名門診雲端電子病歷 (EHR) 供應商 eClinicalWorks 成為美國醫療保險和醫療補助服務中心 (CMS) 數位醫療技術生態系統的積極參與者。這項由白宮高級衛生官員宣布的計劃,旨在促進無縫醫療數據共享,並建立新的行業互通性標準。
- 2023年8月,提供病患參與和臨床工作流程解決方案的雲端平台OceanMD與不列顛哥倫比亞省衛生服務管理局簽署了一份價值3,850萬美元的合約。該協議旨在提供電子轉診、電子諮詢和電子訂單等數位服務,以增強全省的醫療保健服務。
- 2025年3月,歐盟關於建立歐洲健康資料空間的2025/327號條例(EU)生效。該條例旨在使歐盟公民能夠更好地控制其個人健康數據,並增強成員國之間電子健康記錄的互通性。
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
研究方法
数据收集和基准年分析是使用具有大样本量的数据收集模块完成的。该阶段包括通过各种来源和策略获取市场信息或相关数据。它包括提前检查和规划从过去获得的所有数据。它同样包括检查不同信息源中出现的信息不一致。使用市场统计和连贯模型分析和估计市场数据。此外,市场份额分析和关键趋势分析是市场报告中的主要成功因素。要了解更多信息,请请求分析师致电或下拉您的询问。
DBMR 研究团队使用的关键研究方法是数据三角测量,其中包括数据挖掘、数据变量对市场影响的分析和主要(行业专家)验证。数据模型包括供应商定位网格、市场时间线分析、市场概览和指南、公司定位网格、专利分析、定价分析、公司市场份额分析、测量标准、全球与区域和供应商份额分析。要了解有关研究方法的更多信息,请向我们的行业专家咨询。
可定制
Data Bridge Market Research 是高级形成性研究领域的领导者。我们为向现有和新客户提供符合其目标的数据和分析而感到自豪。报告可定制,包括目标品牌的价格趋势分析、了解其他国家的市场(索取国家列表)、临床试验结果数据、文献综述、翻新市场和产品基础分析。目标竞争对手的市场分析可以从基于技术的分析到市场组合策略进行分析。我们可以按照您所需的格式和数据样式添加您需要的任意数量的竞争对手数据。我们的分析师团队还可以为您提供原始 Excel 文件数据透视表(事实手册)中的数据,或者可以帮助您根据报告中的数据集创建演示文稿。