Global Peptide Synthesis Market
Market Size in USD Billion
CAGR :
% 
 USD
4.70 Billion
USD
8.82 Billion
2022
2030
USD
4.70 Billion
USD
8.82 Billion
2022
2030
| 2023 –2030 | |
| USD 4.70 Billion | |
| USD 8.82 Billion | |
|
|
|
|
Peptide Synthesis Market Analysis and Size
Peptide synthesis is the process of chemically creating peptides by combining amino acids in a specific order. It is used in pharmaceutical research for developing new drugs and studying their interactions. It enables the production of custom-designed peptides for studying protein structure and function, as well as creating peptide-based therapies and diagnostics. Peptide synthesis also aids in high-throughput screening and drug discovery efforts.
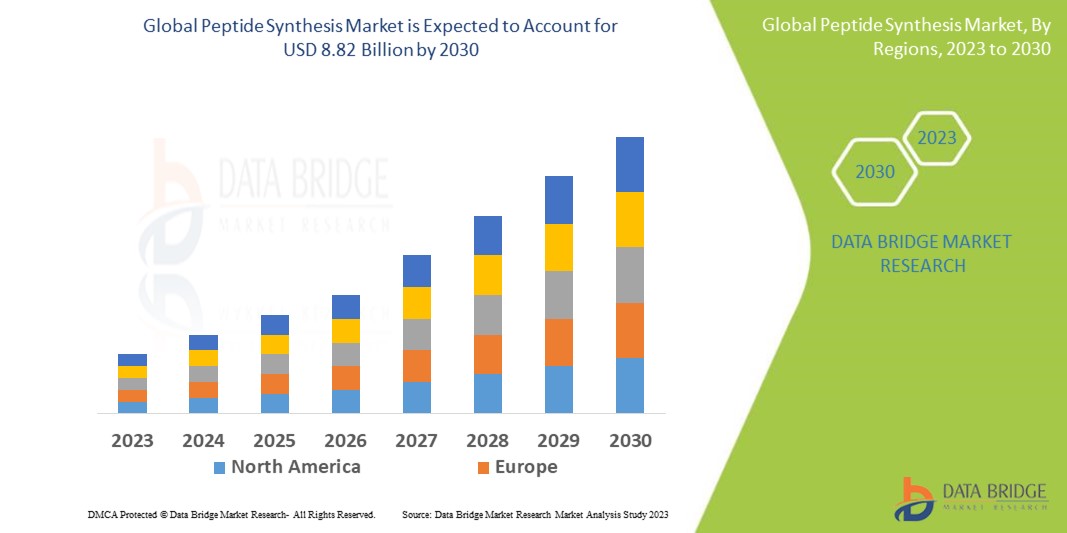
Data Bridge Market Research analyses that the Peptide synthesis Market which was USD 4.7 billion in 2022, would rocket up to USD 8.82 billion by 2030, and is expected to undergo a CAGR of 8.20% during the forecast period. “Therapeutics” dominates the application segment of the peptide synthesis market owing to the growing use of peptides for various disease conditions. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
Peptide Synthesis Market Scope and Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015-2020) |
|
Quantitative Units |
Revenue in USD Million, Volumes in Units, Pricing in USD |
|
Segments Covered |
By Type (Under 75%, 75-85%, Above 85%), Product (Equipment, Reagents and Consumables, Others), Technology (Solid-Phase Peptide Synthesis (SPPS), Solution-Phase Synthesis (SPS), Liquid-Phase Peptide Synthesis (LPPS), Hybrid and Recombinant), Application (Therapeutics, Diagnosis and Research), End User (Pharmaceutical and Biotechnology Companies, Contract Manufacturing Organization (CMO) or Contract Research Organization (CRO), Academic and Research Institutes) |
|
Countries Covered |
U.S., Canada and Mexico in North America, Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe, China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Rest of Asia-Pacific (APAC) in the Asia-Pacific (APAC), Saudi Arabia, U.A.E, South Africa, Egypt, Israel, Rest of Middle East and Africa (MEA) as a part of Middle East and Africa (MEA), Brazil, Argentina and Rest of South America as part of South America. |
|
Market Players Covered |
PolyPeptide Group (Belgium), Merck KGaA (Germany), Thermo Fisher Scientific (U.S.), Enamine Ltd (Ukraine), Alfa Chemistry (China), CEM Corporation (U.S.), GenScript (U.S.), AAPPTec (U.S.), Bachem Holding (Switzerland), AnaSpec, Inc. (U.S.), Biotage (Sweden), GYROS PROTEIN TECHNOLOGIES AB (Sweden), Advanced ChemTech (U.S.), New England Peptide, Inc. (U.S.), Guizhou Xinbang Pharmaceutical Co., Ltd. (China), Hybio Pharmaceutical Co., Ltd. (China), USV Private Limited (India), ScinoPharm Taiwan (Taiwan), CPC Scientific Inc. (U.S.). |
|
Market Opportunities |
|
Market Definition
Peptide synthesis refers to the chemical process of creating peptides, which are short chains of amino acids linked together by peptide bonds. It involves the stepwise assembly of amino acids in a specific order and often requires the protection of certain functional groups to control the reaction. Peptide synthesis can be achieved through various methods, such as solid-phase synthesis, solution-phase synthesis, or recombinant DNA technology. The goal of peptide synthesis is to produce peptides with precise sequences and desired modifications for various applications, including research, drug development, diagnostics, and therapeutics.
Peptide Synthesis Market Dynamics
Drivers
- Increasing demand for peptide-based therapeutics
Peptides offer unique advantages as therapeutics, such as high specificity, low toxicity, and favorable pharmacokinetic properties. The growing prevalence of chronic diseases, including cancer, diabetes, and cardiovascular disorders, is driving the demand for peptide-based drugs. The development of innovative peptide therapeutics and the need for efficient peptide synthesis methods are key drivers for the market.
- Expanding applications in diagnostics and imaging
Peptides play a crucial role in diagnostic applications, including the development of peptide-based biosensors, imaging agents, and biomarker. The rising demand for non-invasive and targeted diagnostic tools, such as peptide-based imaging agents for molecular imaging and peptide-based biosensors for disease detection, is driving the market growth.
Opportunities
- Advancement in therapeutics for rare diseases
Peptides offer potential solutions for treating rare genetic disorders and orphan diseases. The growth of the peptide synthesis market provides an opportunity to develop and manufacture peptide-based therapeutics targeting these conditions. Companies can focus on designing and synthesizing peptides that address specific molecular targets associated with rare diseases, thereby catering to unmet medical needs.
- Personalized medicine and precision therapeutics
Peptides can be tailored to specific patient profiles, leading to the concept of personalized medicine. With advances in genomics, proteomics, and bioinformatics, there is an opportunity for peptide synthesis companies to develop customized peptide therapies that match individual patient characteristics, such as genetic mutations or protein expression patterns. This approach can lead to more effective and targeted treatment strategies.
Restraints/Challenges
- Purification and quality control
Obtaining peptides of high purity is crucial for their successful application in research and therapeutics. Purification methods, such as high-performance liquid chromatography (HPLC), can be time-consuming and require optimization for each peptide sequence. Quality control measures are essential to ensure the accuracy, consistency, and safety of synthesized peptides. Maintaining high-quality standards and efficient purification processes can be a challenge for the peptide synthesis market.
This peptide synthesis market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the peptide synthesis market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Recent Development
- In May 2023, PolyPeptide, a focused global CDMO for peptide- and oligonucleotide-based active pharmaceutical ingredients (APIs), and Numaferm, a German biotech company specialized in process development and production of peptides and proteins, have signed a Preferred Partner Collaboration Agreement for peptide development and production, leveraging PolyPeptide’s cGMP manufacturing capacities, regulatory know-how and market access and Numaferm’s biochemical production platform and expertise in sustainable peptide manufacturing.
- In June 2023, IRBM, an innovative contract research organization, today announced it has signed a new agreement with Merck & Co. Inc., Rahway NJ USA, known as MSD outside the USA and Canada, to continue their collaboration in the peptide therapeutics area.
Global Peptide synthesis Market Scope
The peptide synthesis market is segmented on the basis of type, product, technology, application, end-user. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Type
- Under 75%
- 75-85%
- Above 85%
Product
- Equipment
- Reagents and Consumables
- Others
Technology
- Solid-Phase Peptide Synthesis (SPPS)
- Solution-Phase Synthesis (SPS)
- Liquid-Phase Peptide Synthesis (LPPS)
- Hybrid and Recombinant
Application
- Therapeutics
- Diagnosis
- Research
End-User
- Pharmaceutical and Biotechnology Companies
- Contract Manufacturing Organization (CMO) or Contract Research Organization (CRO)
- Academic and Research Institutes
Peptide synthesis Market Regional Analysis/Insights
The peptide synthesis market is analysed and market size insights and trends are provided by country, type, product, technology, application, and end user referenced above.
The countries covered in the peptide synthesis market report are U.S., Canada and Mexico in North America, Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe, China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Rest of Asia-Pacific (APAC) in the Asia-Pacific (APAC), Saudi Arabia, U.A.E, South Africa, Egypt, Israel, Rest of Middle East and Africa (MEA) as a part of Middle East and Africa (MEA), Brazil, Argentina and Rest of South America as part of South America.
North America is expected to dominate the peptide synthesis market due to the sturdy occurrence of major key players. Furthermore, the rise in the alertness regarding the accessible peptide synthesis technologies and the rising attention on moving the peptide drugs on a commercial scale will further boost the growth of the peptide synthesis market in the region during the forecast period.
Asia-Pacific is projected to observe significant amount of growth of the peptide synthesis market due to the rise in the investments by major key players. Moreover, the increasing consciousness regarding the novel peptide treatments and the growing healthcare expenditure is further anticipated to propel the growth of the peptide synthesis market in the region in the coming years.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points like down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Healthcare Infrastructure growth Installed base and New Technology Penetration
The Peptide synthesis Market also provides you with detailed market analysis for every country growth in healthcare expenditure for capital equipment, installed base of different kind of products for Peptide synthesis Market, impact of technology using life line curves and changes in healthcare regulatory scenarios and their impact on the Peptide synthesis Market. The data is available for historic period 2010-2020.
Competitive Landscape and Peptide synthesis Market Share Analysis
The peptide synthesis market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to the market.
Some of the major players operating in the peptide synthesis market are:
- PolyPeptide Group (Belgium)
- Merck KGaA (Germany)
- Thermo Fisher Scientific (U.S.)
- Enamine Ltd (Ukraine)
- Alfa Chemistry (China)
- CEM Corporation (U.S.)
- GenScript (U.S.)
- AAPPTec (U.S.)
- Bachem Holding (Switzerland)
- AnaSpec, Inc. (U.S.)
- Biotage (Sweden)
- GYROS PROTEIN TECHNOLOGIES AB (Sweden)
- Advanced ChemTech (U.S.)
- New England Peptide, Inc. (U.S.)
- Guizhou Xinbang Pharmaceutical Co., Ltd. (China)
- Hybio Pharmaceutical Co., Ltd. (China)
- USV Private Limited (India)
- ScinoPharm Taiwan (Taiwan)
- CPC Scientific Inc. (U.S.)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Table of Content
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF GLOBAL PEPTIDE SYNTHESIS MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATION
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 KEY TAKEAWAYS
2.2 ARRIVING AT THE GLOBAL PEPTIDE SYNTHESIS MARKET SIZE
2.2.1 VENDOR POSITIONING GRID
2.2.2 TECHNOLOGY LIFE LINE CURVE
2.2.3 TRIPOD DATA VALIDATION MODEL
2.2.4 MARKET GUIDE
2.2.5 MULTIVARIATE MODELLING
2.2.6 TOP TO BOTTOM ANALYSIS
2.2.7 CHALLENGE MATRIX
2.2.8 APPLICATION COVERAGE GRID
2.2.9 STANDARDS OF MEASUREMENT
2.2.10 VENDOR SHARE ANALYSIS
2.2.11 DATA POINTS FROM KEY PRIMARY INTERVIEWS
2.2.12 DATA POINTS FROM KEY SECONDARY DATABASES
2.3 GLOBAL PEPTIDE SYNTHESIS MARKET : RESEARCH SNAPSHOT
2.4 ASSUMPTIONS
3 MARKET OVERVIEW
3.1 DRIVERS
3.2 RESTRAINTS
3.3 OPPORTUNITIES
3.4 CHALLENGES
4 EXECUTIVE SUMMARY
5 PREMIUM INSIGHTS
5.1 PESTEL ANALYSIS
5.2 PORTER’S FIVE FORCES MODEL
5.3 DETAILS OF CMO (CDMO)
5.4 PEPTIDE DRUG TRENDS
6 INDUSTRY INSIGHTS
6.1 MICRO AND MACRO ECONOMIC FACTORS
6.2 PENETRATION AND GROWTH PROSPECT MAPPING
6.3 KEY PRICING STRATEGIES
6.4 INTERVIEWS WITH SPECIALIST
6.5 ANALYIS AND RECOMMENDATION
7 INTELLECTUAL PROPERTY (IP) PORTFOLIO
7.1 PATENT QUALITY AND STRENGTH
7.2 PATENT FAMILIES
7.3 LICENSING AND COLLABORATIONS
7.4 COMPETITIVE LANDSCAPE
7.5 IP STRATEGY AND MANAGEMENT
7.6 OTHER
8 COST ANALYSIS BREAKDOWN
9 TECHNONLOGY ROADMAP
10 INNOVATION TRACKER AND STRATEGIC ANALYSIS
10.1 MAJOR DEALS AND STRATEGIC ALLIANCES ANALYSIS
10.1.1 JOINT VENTURES
10.1.2 MERGERS AND ACQUISITIONS
10.1.3 LICENSING AND PARTNERSHIP
10.1.4 TECHNOLOGY COLLABORATIONS
10.1.5 STRATEGIC DIVESTMENTS
10.2 NUMBER OF PRODUCTS IN DEVELOPMENT
10.3 STAGE OF DEVELOPMENT
10.4 TIMELINES AND MILESTONES
10.5 INNOVATION STRATEGIES AND METHODOLOGIES
10.6 RISK ASSESSMENT AND MITIGATION
10.7 FUTURE OUTLOOK
11 REGULATORY COMPLIANCE
11.1 REGULATORY AUTHORITIES
11.2 REGULATORY CLASSIFICATIONS
11.2.1 CLASS I
11.2.2 CLASS II
11.2.3 CLASS III
11.3 REGULATORY SUBMISSIONS
11.4 INTERNATIONAL HARMONIZATION
11.5 COMPLIANCE AND QUALITY MANAGEMENT SYSTEMS
11.6 REGULATORY CHALLENGES AND STRATEGIES
12 REIMBURSEMENT FRAMEWORK
13 OPPUTUNITY MAP ANALYSIS
14 VALUE CHAIN ANALYSIS
15 HEALTHCARE ECONOMY
15.1 HEALTHCARE EXPENDITURE
15.2 CAPITAL EXPENDITURE
15.3 CAPEX TRENDS
15.4 CAPEX ALLOCATION
15.5 FUNDING SOURCES
15.6 INDUSTRY BENCHMARKS
15.7 GDP RATION IN OVERALL GDP
15.8 HEALTHCARE SYSTEM STRUCTURE
15.9 GOVERNMENT POLICIES
15.1 ECONOMIC DEVELOPMENT
16 GLOBAL PEPTIDE SYNTHESIS MARKET , BY PRODCUCT TYPE
16.1 OVERVIEW
16.2 EQUIPMENT
16.2.1 PEPTIDE SYNTHESIZER
16.2.1.1. MANUAL PEPTIDE SYNTHESIZER
16.2.1.1.1. MARKET VALUE (USD MILLION)
16.2.1.1.2. MARKET VOLUME (UNIT)
16.2.1.1.3. AVERAGE SELLING PRICE (USD)
16.2.1.2. AUTOMATED PEPTIDE SYNTHESIZER
16.2.1.2.1. BATCH SYNTHESIZERS
16.2.1.2.1.1 MARKET VALUE (USD MILLION)
16.2.1.2.1.2 MARKET VOLUME (UNIT)
16.2.1.2.1.3 AVERAGE SELLING PRICE (USD)
16.2.1.2.2. CONTINUOUS FLOW SYNTHESIZERS
16.2.1.2.2.1 MARKET VALUE (USD MILLION)
16.2.1.2.2.2 MARKET VOLUME (UNIT)
16.2.1.2.2.3 AVERAGE SELLING PRICE (USD)
16.2.1.2.3. LIBRARY SYNTHESIZERS
16.2.1.2.3.1 MARKET VALUE (USD MILLION)
16.2.1.2.3.2 MARKET VOLUME (UNIT)
16.2.1.2.3.3 AVERAGE SELLING PRICE (USD)
16.2.2 CHROMATOGRAPHY EQUIPMENT
16.2.2.1. RP-HPLC CHROMATOGRAPHY
16.2.2.2. FLASH CHROMATOGRAPHY
16.2.2.3. ION-EXCHANGE CHROMATOGRAPHY
16.2.2.4. HYDROPHOBIC INTERACTION CHROMATOGRAPHY
16.2.2.5. GEL FILTRATION CHROMATOGRAPHY
16.2.2.6. SIZE EXCLUSION CHROMATOGRAPHY
16.2.2.7. HYDROPHILIC INTERACTION CHROMATOGRAPHY
16.2.2.8. OTHERS
16.2.3 LYOPHILIZER
16.2.3.1. NON-STERILE FREEZE DRYERS
16.2.3.2. STERILIZABLE FREEZE DRYERS
16.3 REAGENTS AND CONSUMABLES
16.3.1 RESINS
16.3.2 AMINO ACID
16.3.3 DYES
16.3.4 COUPLING REAGENTS
16.3.5 DYES & FLUORESCENT LABELING REAGENTS
16.3.6 OTHERS
16.4 SERVICES
16.4.1 MINIMIZE AMINO ACID OXIDATION
16.4.2 DYE-LABELLED PEPTIDE SERVICES
16.4.3 PEPTIDE SERVICES
16.4.4 TFA REMOVAL
16.4.5 ANALYTICAL HPLC
16.4.6 CHECK PURITY LEVEL
16.4.7 MODIFICATION OF SYNTHETIC PEPTIDES
16.4.8 OTHERS
16.5 OTHERS
17 GLOBAL PEPTIDE SYNTHESIS MARKET , BY TECHNOLOGY
17.1 OVERVIEW
17.2 SOLID-PHASE
17.3 SOLUTION-PHASE
17.4 HYBRID
17.5 RECOMBINANT
18 GLOBAL PEPTIDE SYNTHESIS MARKET , BY APPLICATION
18.1 OVERVIEW
18.2 THERAPEUTICS
18.2.1 CANCER
18.2.1.1. BY EQUIPMENT
18.2.1.1.1. PEPTIDE SYNTHESIZER
18.2.1.1.2. CHROMATOGRAPHY EQUIPMENTS
18.2.1.1.3. LYOPHILIZERS
18.2.1.2. BY TECHNOLOGY
18.2.1.2.1. SOLID-PHASE
18.2.1.2.2. SOLUTION-PHASE
18.2.1.2.3. HYBRID
18.2.1.2.4. RECOMBINANT
18.2.2 METABOLIC DISORDER
18.2.2.1. BY EQUIPMENT
18.2.2.1.1. PEPTIDE SYNTHESIZER
18.2.2.1.2. CHROMATOGRAPHY EQUIPMENTS
18.2.2.1.3. LYOPHILIZERS
18.2.2.2. BY TECHNOLOGY
18.2.2.2.1. SOLID-PHASE
18.2.2.2.2. SOLUTION-PHASE
18.2.2.2.3. HYBRID
18.2.2.2.4. RECOMBINANT
18.2.3 CARDIOVASCULAR DISORDER
18.2.3.1. BY EQUIPMENT
18.2.3.1.1. PEPTIDE SYNTHESIZER
18.2.3.1.2. CHROMATOGRAPHY EQUIPMENTS
18.2.3.1.3. LYOPHILIZERS
18.2.3.2. BY TECHNOLOGY
18.2.3.2.1. SOLID-PHASE
18.2.3.2.2. SOLUTION-PHASE
18.2.3.2.3. HYBRID
18.2.3.2.4. RECOMBINANT
18.2.4 RESPIRATORY DISORDER
18.2.4.1. BY EQUIPMENT
18.2.4.1.1. PEPTIDE SYNTHESIZER
18.2.4.1.2. CHROMATOGRAPHY EQUIPMENTS
18.2.4.1.3. LYOPHILIZERS
18.2.4.2. BY TECHNOLOGY
18.2.4.2.1. SOLID-PHASE
18.2.4.2.2. SOLUTION-PHASE
18.2.4.2.3. HYBRID
18.2.4.2.4. RECOMBINANT
18.2.5 GIT (GASTROINTESTINAL DISORDERS)
18.2.5.1. BY EQUIPMENT
18.2.5.1.1. PEPTIDE SYNTHESIZER
18.2.5.1.2. CHROMATOGRAPHY EQUIPMENTS
18.2.5.1.3. LYOPHILIZERS
18.2.5.2. BY TECHNOLOGY
18.2.5.2.1. SOLID-PHASE
18.2.5.2.2. SOLUTION-PHASE
18.2.5.2.3. HYBRID
18.2.5.2.4. RECOMBINANT
18.2.6 INFECTIOUS DISEASES
18.2.6.1. BY EQUIPMENT
18.2.6.1.1. PEPTIDE SYNTHESIZER
18.2.6.1.2. CHROMATOGRAPHY EQUIPMENTS
18.2.6.1.3. LYOPHILIZERS
18.2.6.2. BY TECHNOLOGY
18.2.6.2.1. SOLID-PHASE
18.2.6.2.2. SOLUTION-PHASE
18.2.6.2.3. HYBRID
18.2.6.2.4. RECOMBINANT
18.2.7 PAIN MANAGEMENT
18.2.7.1. BY EQUIPMENT
18.2.7.1.1. PEPTIDE SYNTHESIZER
18.2.7.1.2. CHROMATOGRAPHY EQUIPMENTS
18.2.7.1.3. LYOPHILIZERS
18.2.7.2. BY TECHNOLOGY
18.2.7.2.1. SOLID-PHASE
18.2.7.2.2. SOLUTION-PHASE
18.2.7.2.3. HYBRID
18.2.7.2.4. RECOMBINANT
18.2.8 DERMATOLOGY DISORDER
18.2.8.1. BY EQUIPMENT
18.2.8.1.1. PEPTIDE SYNTHESIZER
18.2.8.1.2. CHROMATOGRAPHY EQUIPMENTS
18.2.8.1.3. LYOPHILIZERS
18.2.8.2. BY TECHNOLOGY
18.2.8.2.1. SOLID-PHASE
18.2.8.2.2. SOLUTION-PHASE
18.2.8.2.3. HYBRID
18.2.8.2.4. RECOMBINANT
18.2.9 CNS DISORDER
18.2.9.1. BY EQUIPMENT
18.2.9.1.1. PEPTIDE SYNTHESIZER
18.2.9.1.2. CHROMATOGRAPHY EQUIPMENTS
18.2.9.1.3. LYOPHILIZERS
18.2.9.2. BY TECHNOLOGY
18.2.9.2.1. SOLID-PHASE
18.2.9.2.2. SOLUTION-PHASE
18.2.9.2.3. HYBRID
18.2.9.2.4. RECOMBINANT
18.2.10 RENAL DISORDER
18.2.10.1. BY EQUIPMENT
18.2.10.1.1. PEPTIDE SYNTHESIZER
18.2.10.1.2. CHROMATOGRAPHY EQUIPMENTS
18.2.10.1.3. LYOPHILIZERS
18.2.10.2. BY TECHNOLOGY
18.2.10.2.1. SOLID-PHASE
18.2.10.2.2. SOLUTION-PHASE
18.2.10.2.3. HYBRID
18.2.10.2.4. RECOMBINANT
18.2.11 OTHERS
18.3 DIAGNOSIS
18.3.1 BY EQUIPMENT
18.3.1.1. PEPTIDE SYNTESIZER
18.3.1.2. CHROMATOGRAPHY EQUIPMENT
18.3.1.3. LYOPHILIZERS
18.3.2 BY TECHNOLOGY
18.3.2.1. SOLID-PHASE
18.3.2.2. SOLUTION-PHASE
18.3.2.3. HYBRID
18.3.2.4. RECOMBINANT
18.4 RESEARCH
18.4.1 BY EQUIPMENT
18.4.1.1. PEPTIDE SYNTESIZER
18.4.1.2. CHROMATOGRAPHY EQUIPMENT
18.4.1.3. LYOPHILIZERS
18.4.2 BY TECHNOLOGY
18.4.2.1. SOLID-PHASE
18.4.2.2. SOLUTION-PHASE
18.4.2.3. HYBRID
18.4.2.4. RECOMBINANT
19 GLOBAL PEPTIDE SYNTHESIS MARKET , BY END USER
19.1 OVERVIEW
19.2 CONTRACT DEVELOPMENT & MANUFACTURING ORGANIZATIONS AND CONTRACT RESEARCH ORGANIZATIONS
19.3 PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES
19.4 ACADEMIC AND GOVERNMENT RESEARCH INSTITUTES
19.5 OTHERS
20 GLOBAL PEPTIDE SYNTHESIS MARKET , BY DISTRIBUTION CHANNEL
20.1 OVERVIEW
20.2 DIRECT TENDER
20.3 RETAIL SALES
20.4 OTHERS
21 GLOBAL PEPTIDE SYNTHESIS MARKET , SWOT AND DBMR ANALYSIS
22 GLOBAL PEPTIDE SYNTHESIS MARKET , COMPANY LANDSCAPE
22.1 COMPANY SHARE ANALYSIS: GLOBAL
22.2 COMPANY SHARE ANALYSIS: NORTH AMERICA
22.3 COMPANY SHARE ANALYSIS: EUROPE
22.4 COMPANY SHARE ANALYSIS: ASIA-PACIFIC
22.5 MERGERS & ACQUISITIONS
22.6 NEW PRODUCT DEVELOPMENT & APPROVALS
22.7 EXPANSIONS
22.8 REGULATORY CHANGES
22.9 PARTNERSHIP AND OTHER STRATEGIC DEVELOPMENTS
23 GLOBAL PEPTIDE SYNTHESIS MARKET , BY REGION
Global Peptide synthesis market , (ALL SEGMENTATION PROVIDED ABOVE IS REPRESENTED IN THIS CHAPTER BY COUNTRY)
23.1 NORTH AMERICA
23.1.1 U.S.
23.1.2 CANADA
23.1.3 MEXICO
23.2 EUROPE
23.2.1 GERMANY
23.2.2 U.K.
23.2.3 ITALY
23.2.4 FRANCE
23.2.5 SPAIN
23.2.6 RUSSIA
23.2.7 SWITZERLAND
23.2.8 TURKEY
23.2.9 BELGIUM
23.2.10 NETHERLANDS
23.2.11 DENMARK
23.2.12 SWEDEN
23.2.13 POLAND
23.2.14 NORWAY
23.2.15 FINLAND
23.2.16 REST OF EUROPE
23.3 ASIA-PACIFIC
23.3.1 JAPAN
23.3.2 CHINA
23.3.3 SOUTH KOREA
23.3.4 INDIA
23.3.5 SINGAPORE
23.3.6 THAILAND
23.3.7 INDONESIA
23.3.8 MALAYSIA
23.3.9 PHILIPPINES
23.3.10 AUSTRALIA
23.3.11 NEW ZEALAND
23.3.12 VIETNAM
23.3.13 TAIWAN
23.3.14 REST OF ASIA-PACIFIC
23.4 SOUTH AMERICA
23.4.1 BRAZIL
23.4.2 ARGENTINA
23.4.3 REST OF SOUTH AMERICA
23.5 MIDDLE EAST AND AFRICA
23.5.1 SOUTH AFRICA
23.5.2 EGYPT
23.5.3 BAHRAIN
23.5.4 UNITED ARAB EMIRATES
23.5.5 KUWAIT
23.5.6 OMAN
23.5.7 QATAR
23.5.8 SAUDI ARABIA
23.5.9 REST OF MEA
23.6 KEY PRIMARY INSIGHTS: BY MAJOR COUNTRIES
24 GLOBAL PEPTIDE SYNTHESIS MARKET , COMPANY PROFILE
24.1 THERMOFISHER SCIENTIFIC INC.
24.1.1 COMPANY OVERVIEW
24.1.2 REVENUE ANALYSIS
24.1.3 GEOGRAPHIC PRESENCE
24.1.4 PRODUCT PORTFOLIO
24.1.5 RECENT DEVELOPMENTS
24.2 MERCK KGAA
24.2.1 COMPANY OVERVIEW
24.2.2 REVENUE ANALYSIS
24.2.3 GEOGRAPHIC PRESENCE
24.2.4 PRODUCT PORTFOLIO
24.2.5 RECENT DEVELOPMENTS
24.3 BACHEM
24.3.1 COMPANY OVERVIEW
24.3.2 REVENUE ANALYSIS
24.3.3 GEOGRAPHIC PRESENCE
24.3.4 PRODUCT PORTFOLIO
24.3.5 RECENT DEVELOPMENTS
24.4 BIOTAGE
24.4.1 COMPANY OVERVIEW
24.4.2 REVENUE ANALYSIS
24.4.3 GEOGRAPHIC PRESENCE
24.4.4 PRODUCT PORTFOLIO
24.4.5 RECENT DEVELOPMENTS
24.5 CEM CORPORATION
24.5.1 COMPANY OVERVIEW
24.5.2 REVENUE ANALYSIS
24.5.3 GEOGRAPHIC PRESENCE
24.5.4 PRODUCT PORTFOLIO
24.5.5 RECENT DEVELOPMENTS
24.6 MESA LABS
24.6.1 COMPANY OVERVIEW
24.6.2 REVENUE ANALYSIS
24.6.3 GEOGRAPHIC PRESENCE
24.6.4 PRODUCT PORTFOLIO
24.6.5 RECENT DEVELOPMENTS
24.7 KANEKA CORPORATION
24.7.1 COMPANY OVERVIEW
24.7.2 REVENUE ANALYSIS
24.7.3 GEOGRAPHIC PRESENCE
24.7.4 PRODUCT PORTFOLIO
24.7.5 RECENT DEVELOPMENTS
24.8 ADVANCED CHEM TECH
24.8.1 COMPANY OVERVIEW
24.8.2 REVENUE ANALYSIS
24.8.3 GEOGRAPHIC PRESENCE
24.8.4 PRODUCT PORTFOLIO
24.8.5 RECENT DEVELOPMENTS
24.9 WUXI STA
24.9.1 COMPANY OVERVIEW
24.9.2 REVENUE ANALYSIS
24.9.3 GEOGRAPHIC PRESENCE
24.9.4 PRODUCT PORTFOLIO
24.9.5 RECENT DEVELOPMENTS
24.1 SYNGENE INTERNATIONAL LIMITED
24.10.1 COMPANY OVERVIEW
24.10.2 REVENUE ANALYSIS
24.10.3 GEOGRAPHIC PRESENCE
24.10.4 PRODUCT PORTFOLIO
24.10.5 RECENT DEVELOPMENTS
24.11 PROTEOGENIX
24.11.1 COMPANY OVERVIEW
24.11.2 REVENUE ANALYSIS
24.11.3 GEOGRAPHIC PRESENCE
24.11.4 PRODUCT PORTFOLIO
24.11.5 RECENT DEVELOPMENTS
24.12 DALTON PHARMA SERVICES
24.12.1 COMPANY OVERVIEW
24.12.2 REVENUE ANALYSIS
24.12.3 GEOGRAPHIC PRESENCE
24.12.4 PRODUCT PORTFOLIO
24.12.5 RECENT DEVELOPMENTS
24.13 BIOSYNTH
24.13.1 COMPANY OVERVIEW
24.13.2 REVENUE ANALYSIS
24.13.3 GEOGRAPHIC PRESENCE
24.13.4 PRODUCT PORTFOLIO
24.13.5 RECENT DEVELOPMENTS
24.13.6
24.14 CSBIO
24.14.1 COMPANY OVERVIEW
24.14.2 REVENUE ANALYSIS
24.14.3 GEOGRAPHIC PRESENCE
24.14.4 PRODUCT PORTFOLIO
24.14.5 RECENT DEVELOPMENTS
24.15 GENSCRIPT
24.15.1 COMPANY OVERVIEW
24.15.2 REVENUE ANALYSIS
24.15.3 GEOGRAPHIC PRESENCE
24.15.4 PRODUCT PORTFOLIO
24.15.5 RECENT DEVELOPMENTS
24.16 ACES PHARMA INC.
24.16.1 COMPANY OVERVIEW
24.16.2 REVENUE ANALYSIS
24.16.3 GEOGRAPHIC PRESENCE
24.16.4 PRODUCT PORTFOLIO
24.16.5 RECENT DEVELOPMENTS
24.17 JPT PEPTIDE TECHNOLOGIES
24.17.1 COMPANY OVERVIEW
24.17.2 REVENUE ANALYSIS
24.17.3 GEOGRAPHIC PRESENCE
24.17.4 PRODUCT PORTFOLIO
24.17.5 RECENT DEVELOPMENTS
24.18 LUXEMBOURG BIO TECHNOLOGIES
24.18.1 COMPANY OVERVIEW
24.18.2 REVENUE ANALYSIS
24.18.3 GEOGRAPHIC PRESENCE
24.18.4 PRODUCT PORTFOLIO
24.18.5 RECENT DEVELOPMENTS
24.19 AMBIOPHARM INC.
24.19.1 COMPANY OVERVIEW
24.19.2 REVENUE ANALYSIS
24.19.3 GEOGRAPHIC PRESENCE
24.19.4 PRODUCT PORTFOLIO
24.19.5 RECENT DEVELOPMENTS
24.2 AURIGENE PHARMACEUTICAL SERVICES LTD
24.20.1 COMPANY OVERVIEW
24.20.2 REVENUE ANALYSIS
24.20.3 GEOGRAPHIC PRESENCE
24.20.4 PRODUCT PORTFOLIO
24.20.5 RECENT DEVELOPMENTS
24.21 CORDENPHARMA INTERNATIONAL
24.21.1 COMPANY OVERVIEW
24.21.2 REVENUE ANALYSIS
24.21.3 GEOGRAPHIC PRESENCE
24.21.4 PRODUCT PORTFOLIO
24.21.5 RECENT DEVELOPMENTS
24.22 CHEMPEP INC.
24.22.1 COMPANY OVERVIEW
24.22.2 REVENUE ANALYSIS
24.22.3 GEOGRAPHIC PRESENCE
24.22.4 PRODUCT PORTFOLIO
24.22.5 RECENT DEVELOPMENTS
24.23 CPC SCIENTIFIC INC.
24.23.1 COMPANY OVERVIEW
24.23.2 REVENUE ANALYSIS
24.23.3 GEOGRAPHIC PRESENCE
24.23.4 PRODUCT PORTFOLIO
24.23.5 RECENT DEVELOPMENTS
24.24 BIO-SYNTHESIS INC
24.24.1 COMPANY OVERVIEW
24.24.2 REVENUE ANALYSIS
24.24.3 GEOGRAPHIC PRESENCE
24.24.4 PRODUCT PORTFOLIO
24.24.5 RECENT DEVELOPMENTS
24.25 ALTA BIOSCIENCE
24.25.1 COMPANY OVERVIEW
24.25.2 REVENUE ANALYSIS
24.25.3 GEOGRAPHIC PRESENCE
24.25.4 PRODUCT PORTFOLIO
24.25.5 RECENT DEVELOPMENTS
24.26 AUSPEP
24.26.1 COMPANY OVERVIEW
24.26.2 REVENUE ANALYSIS
24.26.3 GEOGRAPHIC PRESENCE
24.26.4 PRODUCT PORTFOLIO
24.26.5 RECENT DEVELOPMENTS
24.27 ALMAC GROUP
24.27.1 COMPANY OVERVIEW
24.27.2 REVENUE ANALYSIS
24.27.3 GEOGRAPHIC PRESENCE
24.27.4 PRODUCT PORTFOLIO
24.27.5 RECENT DEVELOPMENTS
24.28 ABI SCIENTIFIC INC.
24.28.1 COMPANY OVERVIEW
24.28.2 REVENUE ANALYSIS
24.28.3 GEOGRAPHIC PRESENCE
24.28.4 PRODUCT PORTFOLIO
24.28.5 RECENT DEVELOPMENTS
25 RELATED REPORTS
26 CONCLUSION
27 QUESTIONNAIRE
28 ABOUT DATA BRIDGE MARKET RESEARCH

Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.