Supply Chain Ecosystem Analysis now part of DBMR Reports
Global Molecular Point of Care Testing (using NAAT) Market, By Product (Instruments and Consumables & Reagents), Indication (Respiratory Infections Testing, Sexually Transmitted Infection (STI) Testing, Gastrointestinal Tract Infections Testing, and Others), End User (Laboratories, Hospitals, Clinics, Ambulatory Centers, Homecare, Assisted Living Facilities, and Others), Mode of Testing (Prescription-Based Testing and OTC Testing), Distribution Channel (Hospital Pharmacy, Retail Pharmacy, and Online Pharmacy) – Industry Trends and Forecast to 2028.
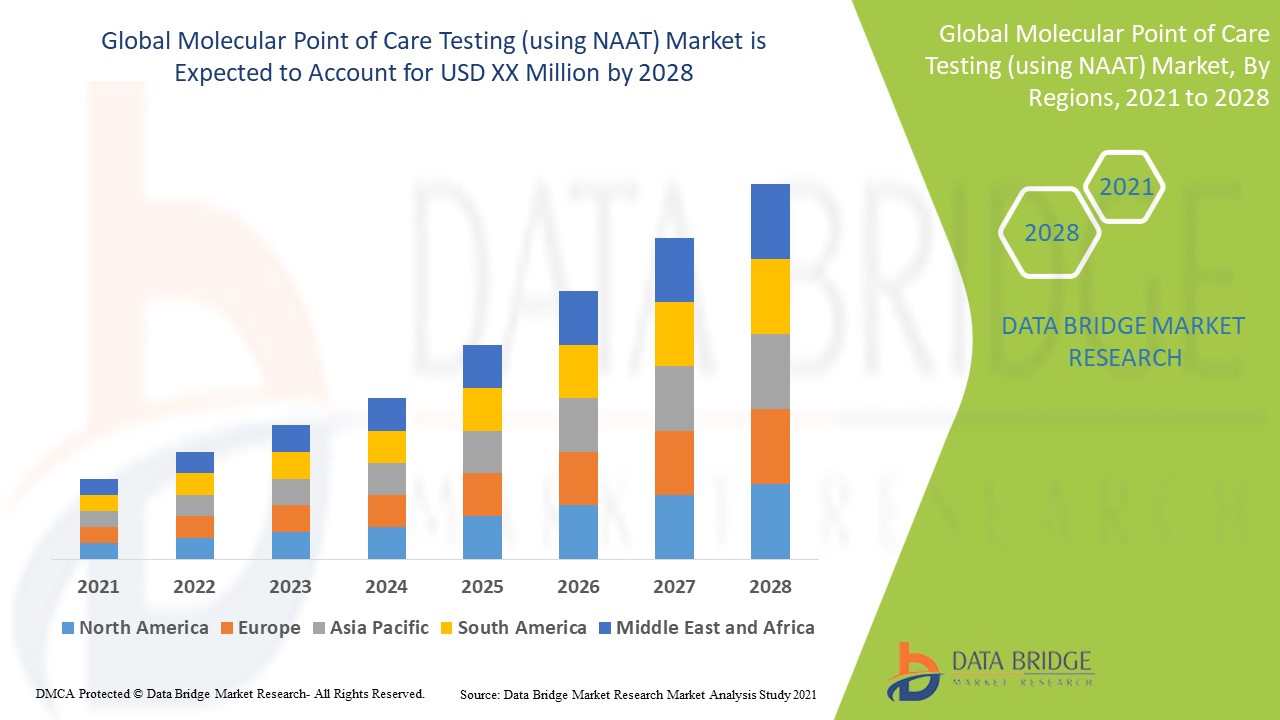
The global molecular point of care testing (using NAAT) market is expected to grow due to improving healthcare facilities and infrastructures and the adoption of end-of-care testing at the hospital, laboratories, and clinics, which can drive the market's growth. The other factors anticipated to propel the development of the molecular point of care testing (using NAAT) market include the rising prevalence of infectious diseases.

The other factors, such as the lack of skilled professionals and the high cost of customized molecular point-of-care testing (using NAAT), hamper the growth of the global molecular end-of-care testing (using NAAT) market. On the other hand, the increased healthcare expenditure and emerging countries with developed hospitals act as an opportunity to grow the global molecular point of care testing (using NAAT) market.
Data Bridge Market Research analyzes that the global molecular point of care testing (using NAAT) market will grow at a CAGR of 10.8% from 2021 to 2028.
|
Report Metric
|
Details
|
|
Forecast Period
|
2021 to 2028
|
|
Base Year
|
2020
|
|
Historic Years
|
2019
|
|
Quantitative Units
|
Revenue in USD Million, Pricing in USD
|
|
Segments Covered
|
By Product (Instruments and Consumables & Reagents), Indication (Respiratory Infections Testing, Sexually Transmitted Infection (STI) Testing, Gastrointestinal Tract Infections Testing, and Others), End User (Laboratories, Hospitals, Clinics, Ambulatory Centers, Homecare, Assisted Living Facilities, and Others), Mode of Testing (Prescription-Based Testing and OTC Testing), Distribution Channel (Hospital Pharmacy, Retail Pharmacy, and Online Pharmacy)
|
|
Countries Covered
|
North America (U.S., Canada), Europe (U.K., Germany, France, Spain, Italy, Rest of Europe), South Asia (Pakistan, Bangladesh, India, Rest of South Asia), South East Asia (Singapore, Malaysia, Vietnam Indonesia, Thailand, Philippines, Rest of South East Asia), Middle East (Saudi Arabia, UAE, Egypt, Israel, and Rest of Middle East), and Africa (South Africa, Kenya, Nigeria, and Rest of Africa)
|
|
Market Players Covered
|
SD Biosensor, Inc., Abbott, F.Hoffmann-La Roche Ltd., Danaher, Thermo Fisher Scientific Inc, binx Health, Inc., Lucira, Meridian Bioscience, Quantumdx Group Ltd., BD, Biomérieux Sa, Qiagen, Quidel Corporation, Sysmex Corporation, Bio-Rad Laboratories, Inc., Sekisui Diagnostic, Randox Laboratories Ltd., GenMark Diagnostics, Seegene Inc. Grifols S.A., Wondfo, Oxford Nanopore Diagnostic, PerkinElmer Inc., and Co-Diagnostic,
|
POCT is characterized as testing which can be done close to the patient where a medical decision can be made immediately, including the results and monitoring. POCT devices now entering clinical use are based on nucleic acid amplification technologies (NAAT), defined here as second-generation devices. These platforms generally have improved sensitivity, typically 60 to 90%, compared to first-generation POCTs, and require portable instrumentation with a footprint of approximately 30cm x 30cm. These instruments are essentially used in molecularly detecting various disorders such as HIV, HPV, and respiratory disease through NAAT technology. NAAT is Nucleic Acid Amplification Techniques that allow the identification of pathogenic organisms by detecting their DNA or RNA.
In the worldwide pandemic, attempt to synergize the sensitivity of NAATs and the ease of use of POCT assays, miniaturized NAAT-based POCT devices and assays were devised for faster screening and diagnosis of COVID-19, which also propelled the growth of the market.
This section deals with understanding the market drivers, advantages, opportunities, restraints, and challenges. All of this is discussed in detail below:
Drivers
It is a common fact that health degeneration starts with an increase in age. As the rate of chronic disease grows with time, the factors associated with the disease rates also increase. The high number of obese/ overweight population also has increased the prevalence of chronic diseases in various regions across the globe. The rise in the number of chronic diseases similar to HPV, STI, and TB among more, has led to people opting for NAAT-based point-of-care and products for testing. Therefore, this will drive the growth of the Global molecular point of care testing (using NAAT) market in the coming years.
With the increase in the successful diagnosis of chronic diseases, including cancer, anemia, type 2 diabetes, and others, the improved health care professionals provide regular training sessions for the professionals for conducting tests in account for the improvement of diagnosis rate in the region. With the increase in the burden of diseases, the diagnosis rate is also increasing. With the advancements in the diagnosis, the rate is also growing for people going through the diagnosis.
Home healthcare preferences make clinical information available at the right place and time. It will also help in reducing error and increasing safety with ensured quality. They also help elders in the case of a medical emergency. Many strategic changes have been made in health care to address point-of-care testing benefits. As per the World Health Organization (WHO), the number of people aged 65 and above is expected to rise from an estimated 524 million in 2010 to approximately 1.5 billion in 2050, with developed countries accounting for the majority of the increase.
Opportunities
An increase in the burden of chronic and non-chronic disease globally coupled with the rise in the geriatric population in Africa and Asia has created more demand for the development of new kits and solutions for molecular point-of-care testing with better efficacy and less time consumption. To provide diagnosis and screening in this region, the players in the market undergo partnership and agreement to provide quality care through kits and instruments to the global population.
Increasing healthcare expenditure and infrastructure can help patients take hassle-free advanced treatment to receive better diagnoses, procedures, and treatment for fast recovery. Point-of-care testing devices are beneficial for detecting various diseases and supporting treatment plans using POC software and solutions. Healthcare spending comprises out-of-pocket payments (people paying for their care), government expenditure, and sources including health insurance and activities by non-governmental organizations.
Restraints/Challenges
The point-of-care testing products require a substantial considerable cost for the development process. As the concept needs to be locked down, the materials must be sourced, and it is essential to consider lead times. Additionally, the process is time-consuming as the device design needs sufficient time to fully mature before bringing to the market for use.
When compared with the results of laboratory testing standards, the NAAT testing results differ. Laboratory testing is more advanced, follows the process, protocol, and science for laboratory testing, and fully integrates with the technology necessary to analyze, validate, and document effects accurately. However, POCT/NAAT is undoubtedly associated with a lack of precise results. Molecular POCT cannot complete the standardization, and other critical criteria cannot achieve the standardization and different essential standards. It is a significant challenge for the global molecular point of care testing market. The PoC tests might sometimes provide erroneous results and suffers test results variability.
COVID-19 created a major impact on the molecular point of care testing (using NAAT) market. COVID-19 has positively affected the price and demand of molecular point of care testing (using NAAT) products at a higher level because of progressive online sales and technological advancement in the products. The COVID-19 pandemic outbreak positively impacted the market's growth owing to the high requirement for molecular point of care testing (using NAAT), especially in various hospitals. The current priority in most hospitals is given to COVID-19 treatments as the surge of COVID-19 cases is still persistent, which also increases the usage of molecular point of care testing (using NAAT). Moreover, many international governments and healthcare organizations have supported the supply of these products due to their high priority in crucial cases.
Recent Developments
The global molecular point of care testing (using NAAT) market is categorized into five notable segments based on the product, indication, end user, mode of testing, and distribution channel. The growth amongst these segments will help to analyze market growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Product
Based on product, the global molecular point of care testing (using NAAT) market is segmented into instruments and consumables and reagents.
Indication
Based on indication, the global molecular point of care testing (using NAAT) market is segmented into sexually transmitted infection (STI) testing, respiratory infection testing, gastrointestinal tract infection testing, and others.
End User
Based on end user, the global molecular point of care testing (using NAAT) market is segmented into laboratories, hospitals, clinics, ambulatory centers, home care, assisted living facilities, and others.
Mode of Testing
Based on mode of testing, the global molecular point of care testing (using NAAT) market is segmented into prescription-based testing and OTC testing.
Distribution Channel
Based on the distribution channel, the global molecular point of care testing (using NAAT) market is segmented into hospital pharmacy, retail pharmacy, and online pharmacy.
The global molecular point of care testing (using NAAT) market is categorized into five notable segments based on the product, indication, end user, mode of testing and distribution channel.
The global molecular point of care testing (using NAAT) market is categorized into countries North America (U.S., Canada), Europe (U.K., Germany, France, Spain, Italy, Rest of Europe), South Asia (Pakistan, Bangladesh, India, Rest of South Asia), South East Asia (Singapore, Malaysia, Vietnam Indonesia, Thailand, Philippines, Rest of South East Asia), Middle East (Saudi Arabia, UAE, Egypt, Israel, and Rest of Middle East), and Africa (South Africa, Kenya, Nigeria, and Rest of Africa).
North America dominates the global molecular point of care testing (using NAAT) market with a CAGR of around 12.1% in terms of market share and market revenue. It will continue to flourish its dominance during the forecast period. This is due to the rising prevalence of infectious and chronic diseases and the hospital adoption of point-of-care testing.
The global molecular point of care testing (using NAAT) market competitive landscape provides details about the competitor. Components included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, company strengths and weaknesses, product launch, regulatory guidelines, brand analysis, product approvals, product payload, application dominance, technology lifeline curve. The above data points provided are only related to the company’s focus on the global molecular topic of care testing (using NAAT) market.
Danaher is the dominating market player. Some of the other major players operating in the global molecular point of care testing (using NAAT) market are SD Biosensor, Inc., Abbott, F.Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc, binx Health, Inc., Lucira, Meridian Bioscience, Quantumdx Group Ltd., BD, Biomérieux Sa, Qiagen, Quidel Corporation, Sysmex Corporation, Bio-Rad Laboratories, Inc., Sekisui Diagnostic, Randox Laboratories Ltd., GenMark Diagnostics, Seegene Inc. Grifols S.A., Wondfo, Oxford Nanopore Diagnostic, PerkinElmer Inc., and Co-Diagnostic among others.
SKU-
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET
1.4 LIMITATIONS
1.5 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 CURRENCY AND PRICING
2.5 DBMR TRIPOD DATA VALIDATION MODEL
2.6 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.7 MULTIVARIATE MODELLING
2.8 DISTRIBUTION CHANNEL LIFELINE CURVE
2.9 DBMR MARKET POSITION GRID
2.1 VENDOR SHARE ANALYSIS
2.11 MARKET END USER COVERAGE GRID
2.12 SECONDARY SOURCES
2.13 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PORTERS FIVE FORCES
4.2 PESTEL ANALYSIS
4.3 ANNUAL TEST VOLUME: FOR ALL COUNTRIES
4.3.1 CEPHEID
4.3.2 THERMO FISHER SCIENTIFIC INC.
4.3.3 HOLOGIC, INC.
4.3.4 BD
4.3.5 F. HOFFMANN-LA ROCHE LTD
4.4 NUMBER OF POCT INSTRUMENTS INSTALLED IN EACH COUNTRY
4.5 DISTRIBUTORS
4.5.1 NORTH AMERICA
4.5.2 EUROPE
4.5.3 SOUTH ASIA
4.5.4 SOUTH EAST ASIA
4.5.5 MIDDLE EAST
4.5.6 AFRICA
5 REGULATORY FRAMEWORK
6 MARKET OVERVIEW
6.1 DRIVERS
6.1.1 INCREASING PREVALENCE OF CHRONIC DISEASES
6.1.2 IMPROVING DIAGNOSIS SEEKING RATE
6.1.3 RISING TECHNOLOGICAL ADVANCEMENTS
6.1.4 RISING GERIATRIC POPULATION ACROSS GLOBE
6.1.5 RISING PREFERENCE FOR HOMECARE TESTING
6.2 RESTRAINTS
6.2.1 PRODUCT RECALLS
6.2.2 HIGH COSTS OF PRODUCTS
6.2.3 NON-AVAILABILITY OF RELEVANT AND APPROPRIATE KITS
6.3 OPPORTUNITIES
6.3.1 STRATEGIC INITIATIVES TAKEN BY MARKET PLAYERS
6.3.2 RISING HEALTHCARE EXPENDITURE
6.3.3 PANDEMIC OUTBREAK OF COVID-19
6.3.4 GROWING DEMAND FOR BETTER QUALITY HEALTHCARE AND EARLY DIAGNOSIS
6.4 CHALLENGES
6.4.1 LACK OF ALIGNMENT WITH TEST RESULTS OBTAINED FROM LABORATORIES
6.4.2 LACK OF ACCESSIBILITY
7 IMPACT OF COVID-19 ON THE GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET
7.1 IMPACT ON PRICE
7.2 IMPACT ON DEMAND
7.3 IMPACT ON SUPPLY CHAIN
7.4 KEY INITIATIVES BY MARKET PLAYERS DURING THE COVID 19
7.5 CONCLUSION:
8 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT
8.1 OVERVIEW
8.2 CONSUMABLES & REAGENTS
8.3 INSTRUMENTS
8.3.1 MOBILE PLATFORM (SMALL & PORTABLE)
8.3.2 FACILITY-BASED PLATFORMS (LARGE POCT PLATFORM)
9 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION
9.1 OVERVIEW
9.2 RESPIRATORY INFECTIONS TESTING
9.2.1 COVID-19
9.2.2 FLU B
9.2.3 FLU A
9.2.4 RSV
9.2.5 TUBERCULOSIS
9.2.6 PNEUMONIA (PNEUMOCOCCAL PNEUMONIA)
9.2.7 OTHERS
9.3 SEXUALLY TRANSMITTED INFECTION (STI) TESTING
9.3.1 TV
9.3.2 CT
9.3.3 NG
9.3.4 HPV
9.3.5 HSV
9.3.6 MG
9.3.7 UU
9.3.8 MH
9.3.9 OTHERS
9.4 GASTROINTESTINAL TRACT INFECTIONS TESTING
9.4.1 GROUP B STREPTOCOCCUS
9.4.2 SHIGA TOXIN
9.4.3 H. PYLORI
9.5 OTHERS
10 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER
10.1 OVERVIEW
10.2 LABORATORIES
10.2.1 POLYMERASE CHAIN REACTION
10.2.2 ISOTHERMAL AMPLIFICATION
10.3 HOSPITAL
10.3.1 POLYMERASE CHAIN REACTION
10.3.2 ISOTHERMAL AMPLIFICATION
10.4 CLINICS
10.4.1 POLYMERASE CHAIN REACTION
10.4.2 ISOTHERMAL AMPLIFICATION
10.5 AMBULATORY CENTER
10.5.1 POLYMERASE CHAIN REACTION
10.5.2 ISOTHERMAL AMPLIFICATION
10.6 HOMECARE
10.6.1 POLYMERASE CHAIN REACTION
10.6.2 ISOTHERMAL AMPLIFICATION
10.7 ASSISTED LIVING FACILITIES
10.7.1 POLYMERASE CHAIN REACTION
10.7.2 ISOTHERMAL AMPLIFICATION
10.8 OTHERS
11 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING
11.1 OVERVIEW
11.2 PRESCRIPTION-BASED TESTING
11.3 OTC TESTING
12 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL
12.1 OVERVIEW
12.2 HOSPITAL PHARMACY
12.3 RETAIL PHARMACY
12.4 ONLINE PHARMACY
13 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION
13.1 OVERVIEW
13.2 NORTH AMERICA
13.2.1 U.S.
13.2.2 CANADA
13.3 EUROPE
13.3.1 U.K.
13.3.2 GERMANY
13.3.3 ITALY
13.3.4 FRANCE
13.3.5 SPAIN
13.3.6 REST OF EUROPE
13.4 SOUTH ASIA
13.4.1 INDIA
13.4.2 PAKISTAN
13.4.3 BANGLADESH
13.4.4 REST OF SOUTH ASIA
13.5 SOUTH EAST ASIA
13.5.1 SINGAPORE
13.5.2 VIETNAM
13.5.3 THAILAND
13.5.4 MALAYSIA
13.5.5 INDONESIA
13.5.6 PHILIPPINES
13.5.7 REST OF SOUTH EAST ASIA
13.6 MIDDLE EAST
13.6.1 SAUDI ARABIA
13.6.2 U.A.E
13.6.3 ISRAEL
13.6.4 EGYPT
13.6.5 REST OF MIDDLE EAST
13.7 AFRICA
13.7.1 SOUTH AFRICA
13.7.2 KENYA
13.7.3 NIGERIA
13.7.4 REST OF AFRICA
14 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: COMPANY LANDSCAPE
14.1 COMPANY SHARE ANALYSIS: GLOBAL
14.2 COMPANY SHARE ANALYSIS: NORTH AMERICA
14.3 COMPANY SHARE ANALYSIS: EUROPE
14.4 COMPANY SHARE ANALYSIS: SOUTH ASIA
15 SWOT ANALYSIS
16 COMPANY PROFILE
16.1 DANAHER
16.1.1 COMPANY SNAPSHOT
16.1.2 REVENUE ANALYSIS
16.1.3 COMPANY SHARE ANALYSIS
16.1.4 PRODUCT PORTFOLIO
16.1.5 RECENT DEVELOPMENTS
16.2 THERMO FISHER SCIENTIFIC INC.
16.2.1 COMPANY SNAPSHOT
16.2.2 REVENUE ANALYSIS
16.2.3 COMPANY SHARE ANALYSIS
16.2.4 PRODUCT PORTFOLIO
16.2.5 RECENT DEVELOPMENTS
16.3 BD
16.3.1 COMPANY SNAPSHOT
16.3.2 REVENUE ANALYSIS
16.3.3 COMPANY SHARE ANALYSIS
16.3.4 PRODUCT PORTFOLIO
16.3.5 RECENT DEVELOPMENTS
16.4 F.HOFFMANN-LA-ROCHE LTD.
16.4.1 COMPANY SNAPSHOT
16.4.2 REVENUE ANALYSIS
16.4.3 COMPANY SHARE ANALYSIS
16.4.4 PRODUCT PORTFOLIO
16.4.5 RECENT DEVELOPMENTS
16.5 ABBOTT
16.5.1 COMPANY SNAPSHOT
16.5.2 REVENUE ANALYSIS
16.5.3 PRODUCT PORTFOLIO
16.5.4 RECENT DEVELOPMENTS
16.6 BINX HEALTH, INC.
16.6.1 COMPANY SNAPSHOT
16.6.2 PRODUCT PORTFOLIO
16.6.3 RECENT DEVELOPMENT
16.7 BIOMERIEUX SA
16.7.1 COMPANY SNAPSHOT
16.7.2 REVENUE ANALYSIS
16.7.3 PRODUCT PORTFOLIO
16.7.4 RECENT DEVELOPMENT
16.8 CO-DIAGNOSTICS, INC.
16.8.1 COMPANY SNAPSHOT
16.8.2 REVENUE ANALYSIS
16.8.3 PRODUCT PORTFOLIO
16.8.4 RECENT DEVELOPMENTS
16.9 GENMARK DIAGNOSTICS, INC
16.9.1 COMPANY SNAPSHOT
16.9.2 REVENUE ANALYSIS
16.9.3 PRODUCT PORTFOLIO
16.9.4 RECENT DEVELOPMENT
16.1 GRIFOLS, S.A.
16.10.1 COMPANY SNAPSHOT
16.10.2 REVENUE ANALYSIS
16.10.3 PRODUCT PORTFOLIO
16.10.4 RECENT DEVELOPMENT
16.11 LUCIRA HEALTH INC.
16.11.1 COMPANY SNAPSHOT
16.11.2 PRODUCT PORTFOLIO
16.11.3 RECENT DEVELOPMENTS
16.12 MERIDIAN BIOSCIENCE
16.12.1 COMPANY SNAPSHOT
16.12.2 REVENUE ANALYSIS
16.12.3 PRODUCT PORTFOLIO
16.12.4 RECENT DEVELOPMENTS
16.13 QUANTUMDX GROUP LTD.
16.13.1 COMPANY SNAPSHOT
16.13.2 PRODUCT PORTFOLIO
16.13.3 RECENT DEVELOPMENTS
16.14 QUIDEL CORPORATION
16.14.1 COMPANY SNAPSHOT
16.14.2 REVENUE ANALYSIS
16.14.3 PRODUCT PORTFOLIO
16.14.4 RECENT DEVELOPMENT
16.15 SD BIOSENSOR, INC.
16.15.1 COMPANY SNAPSHOT
16.15.2 PRODUCT PORTFOLIO
16.15.3 RECENT DEVELOPMENT
16.16 SEKISUI DIAGNOSTICS (A SUBSIDIARY OF SEKISUI MEDICAL)
16.16.1 COMPANY SNAPSHOT
16.16.2 PRODUCT PORTFOLIO
16.16.3 RECENT DEVELOPMENT
16.17 SYSMEX (A SUBSIDIARY OF SYSMEX CORPORATION
16.17.1 COMPANY SNAPSHOT
16.17.2 REVENUE ANALYSIS
16.17.3 PRODUCT PORTFOLIO
16.17.4 RECENT DEVELOPMENT
16.18 WONDFO
16.18.1 COMPANY SNAPSHOT
16.18.2 PRODUCT PORTFOLIO
16.18.3 RECENT DEVELOPMENT
17 QUESTIONNAIRE
18 RELATED REPORTS
TABLE 1 CEPHEID: ANNUAL TEST VOLUME, FOR ALL COUNTRIES, 2020 (UNITS)
TABLE 2 THERMO FISHER SCIENTIFIC INC. : ANNUAL TEST VOLUME, FOR ALL COUNTRIES, 2020 (UNITS)
TABLE 3 HOLOGIC, INC. : ANNUAL TEST VOLUME, FOR ALL COUNTRIES, 2020 (UNITS)
TABLE 4 BD : ANNUAL TEST VOLUME, FOR ALL COUNTRIES, 2020 (UNITS)
TABLE 5 F. HOFFMANN-LA ROCHE LTD : ANNUAL TEST VOLUME, FOR ALL COUNTRIES, 2020 (UNITS)
TABLE 6 NUMBER OF POCT INSTRUMENTS INSTALLED IN EACH COUNTRY, 2020 (UNITS)
TABLE 7 DISTRIBUTORS ACROSS NORTH AMERICA
TABLE 8 DISTRIBUTORS ACROSS EUROPE
TABLE 9 DISTRIBUTORS ACROSS SOUTH ASIA
TABLE 10 DISTRIBUTORS ACROSS SOUTH EAST ASIA
TABLE 11 DISTRIBUTORS ACROSS MIDDLE EAST
TABLE 12 DISTRIBUTORS ACROSS AFRICA
TABLE 13 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 14 GLOBAL CONSUMABLES & REAGENTS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 15 GLOBAL INSTRUMENTS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 16 GLOBAL INSTRUMENTS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 17 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 18 GLOBAL RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 19 GLOBAL RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 20 GLOBAL SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 21 GLOBAL SEXUALLY TRANSMITTED IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 22 GLOBAL GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 23 GLOBAL RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 24 GLOBAL OTHERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 25 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 26 GLOBAL LABOROTORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 27 GLOBAL LABOROTORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 28 GLOBAL HOSPITAL IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 29 GLOBAL HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 30 GLOBAL CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 31 GLOBAL CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 32 GLOBAL AMBULATORY CENTER IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 33 GLOBAL AMBULATORY CENTER IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 34 GLOBAL HOME CARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 35 GLOBAL HOME CARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 36 GLOBAL ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 37 GLOBAL ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 38 GLOBAL ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 39 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 40 GLOBAL PRESCRIPTION-BASED TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION 2019-2028 (USD MILLION)
TABLE 41 GLOBAL OTC TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 42 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 43 GLOBAL HOSPITAL PHARMACY IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 44 GLOBAL RETAIL PHARMACY IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 45 GLOBAL ONLINE PHARMACY IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 46 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 47 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY COUNTRY, 2019-2028 (USD MILLION)
TABLE 48 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 49 NORTH AMERICA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 50 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 51 NORTH AMERICA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 52 NORTH AMERICA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 53 NORTH AMERICA GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 54 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 55 NORTH AMERICA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 56 NORTH AMERICA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 57 NORTH AMERICA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 58 NORTH AMERICA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 59 NORTH AMERICA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 60 NORTH AMERICA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 61 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 62 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 63 U.S. MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 64 U.S. INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 65 U.S. MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 66 U.S. RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 67 U.S. SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 68 U.S. GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 69 U.S. MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 70 U.S. LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 71 U.S. HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 72 U.S. CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 73 U.S. AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 74 U.S. HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 75 U.S. ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 76 U.S. MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 77 U.S. MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 78 CANADA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 79 CANADA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 80 CANADA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 81 CANADA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 82 CANADA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 83 CANADA GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 84 CANADA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 85 CANADA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 86 CANADA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 87 CANADA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 88 CANADA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 89 CANADA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 90 CANADA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 91 CANADA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 92 CANADA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 93 EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY COUNTRY, 2019-2028 (USD MILLION)
TABLE 94 EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 95 EUROPE INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 96 EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 97 EUROPE RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 98 EUROPE SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 99 EUROPE GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 100 EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 101 EUROPE LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 102 EUROPE HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 103 EUROPE CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 104 EUROPE AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 105 EUROPE HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 106 EUROPE ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 107 EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 108 CANADA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 109 U.K. MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 110 U.K. INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 111 U.K. MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 112 U.K. RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 113 U.K. SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 114 U.K. GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 115 U.K. MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 116 U.K. LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 117 U.K. HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 118 U.K. CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 119 U.K. AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 120 U.K. HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 121 U.K. ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 122 U.K. MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 123 CANADA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 124 GERMANY MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 125 GERMANY INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 126 GERMANY MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 127 GERMANY RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 128 GERMANY SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 129 GERMANY GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 130 GERMANY MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 131 GERMANY LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 132 GERMANY HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 133 GERMANY CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 134 GERMANY AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 135 GERMANY HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 136 GERMANY ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 137 GERMANY MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 138 CANADA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 139 ITALY MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 140 ITALY INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 141 ITALY MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 142 ITALY RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 143 ITALY SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 144 ITALY GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 145 ITALY MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 146 ITALY LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 147 ITALY HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 148 ITALY CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 149 ITALY AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 150 ITALY HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 151 ITALY ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 152 ITALY MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 153 ITALY MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 154 FRANCE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 155 FRANCE INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 156 FRANCE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 157 FRANCE RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 158 FRANCE SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 159 FRANCE GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 160 FRANCE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 161 FRANCE LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 162 FRANCE HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 163 FRANCE CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 164 FRANCE AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 165 FRANCE HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 166 FRANCE ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 167 FRANCE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 168 FRANCE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 169 SPAIN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 170 SPAIN INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 171 SPAIN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 172 SPAIN RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 173 SPAIN SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 174 SPAIN GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 175 SPAIN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 176 SPAIN LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 177 SPAIN HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 178 SPAIN CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 179 SPAIN AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 180 SPAIN HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 181 SPAIN ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 182 SPAIN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 183 SPAIN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 184 REST OF EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 185 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY COUNTRY, 2019-2028 (USD MILLION)
TABLE 186 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 187 SOUTH ASIA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 188 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 189 SOUTH ASIA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 190 SOUTH ASIA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 191 SOUTH ASIA GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 192 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 193 SOUTH ASIA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 194 SOUTH ASIA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 195 SOUTH ASIA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 196 SOUTH ASIA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 197 SOUTH ASIA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 198 SOUTH ASIA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 199 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 200 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 201 INDIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 202 INDIA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 203 INDIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 204 INDIA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 205 INDIA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 206 INDIA GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 207 INDIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 208 INDIA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 209 INDIA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 210 INDIA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 211 INDIA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 212 INDIA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 213 INDIA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 214 INDIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 215 INDIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 216 PAKISTAN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 217 PAKISTAN INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 218 PAKISTAN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 219 PAKISTAN RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 220 PAKISTAN SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 221 PAKISTAN GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 222 PAKISTAN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 223 PAKISTAN LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 224 PAKISTAN HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 225 PAKISTAN CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 226 PAKISTAN AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 227 PAKISTAN HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 228 PAKISTAN ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 229 PAKISTAN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 230 PAKISTAN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 231 BANGLADESH MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 232 BANGLADESH INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 233 BANGLADESH MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 234 BANGLADESH RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 235 BANGLADESH SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 236 BANGLADESH GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 237 BANGLADESH MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 238 BANGLADESH LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 239 BANGLADESH HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 240 BANGLADESH CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 241 BANGLADESH AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 242 BANGLADESH HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 243 BANGLADESH ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 244 BANGLADESH MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 245 BANGLADESH MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 246 REST OF SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 247 SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY COUNTRY, 2019-2028 (USD MILLION)
TABLE 248 SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 249 SOUTH EAST ASIA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 250 SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 251 SOUTH EAST ASIA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 252 SOUTH EAST ASIA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 253 SOUTH EAST ASIA GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 254 SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 255 SOUTH EAST ASIA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 256 SOUTH EAST ASIA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 257 SOUTH EAST ASIA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 258 SOUTH EAST ASIA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 259 SOUTH EAST ASIA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 260 SOUTH EAST ASIA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 261 SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 262 SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 263 SINGAPORE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 264 SINGAPORE INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 265 SINGAPORE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 266 SINGAPORE RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 267 SINGAPORE SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 268 SINGAPORE GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 269 SINGAPORE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 270 SINGAPORE LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 271 SINGAPORE HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 272 SINGAPORE CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 273 SINGAPORE AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 274 SINGAPORE HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 275 SINGAPORE ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 276 SINGAPORE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 277 SINGAPORE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 278 VIETNAM MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 279 VIETNAM INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 280 VIETNAM MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 281 VIETNAM RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 282 VIETNAM SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 283 VIETNAM GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 284 VIETNAM MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 285 VIETNAM LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 286 VIETNAM HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 287 VIETNAM CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 288 VIETNAM AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 289 VIETNAM HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 290 VIETNAM ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 291 VIETNAM MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 292 VIETNAM MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 293 THAILAND MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 294 THAILAND INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 295 THAILAND MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 296 THAILAND RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 297 THAILAND SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 298 THAILAND GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 299 THAILAND MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 300 THAILAND LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 301 THAILAND HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 302 THAILAND CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 303 THAILAND AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 304 THAILAND HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 305 THAILAND ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 306 THAILAND MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 307 THAILAND MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 308 MALAYSIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 309 MALAYSIA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 310 MALAYSIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 311 MALAYSIA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 312 MALAYSIA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 313 MALAYSIA GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 314 MALAYSIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 315 MALAYSIA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 316 MALAYSIA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 317 MALAYSIA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 318 MALAYSIA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 319 MALAYSIA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 320 MALAYSIA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 321 MALAYSIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 322 MALAYSIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 323 INDONESIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 324 INDONESIA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 325 INDONESIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 326 INDONESIA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 327 INDONESIA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 328 INDONESIA GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 329 INDONESIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 330 INDONESIA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 331 INDONESIA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 332 INDONESIA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 333 INDONESIA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 334 INDONESIA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 335 INDONESIA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 336 INDONESIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 337 INDONESIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 338 PHILIPPINES MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 339 PHILIPPINES INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 340 PHILIPPINES MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 341 PHILIPPINES RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 342 PHILIPPINES SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 343 PHILIPPINES GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 344 PHILIPPINES MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 345 PHILIPPINES LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 346 PHILIPPINES HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 347 PHILIPPINES CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 348 PHILIPPINES AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 349 PHILIPPINES HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 350 PHILIPPINES ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 351 PHILIPPINES MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 352 PHILIPPINES MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 353 REST OF SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 354 MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY COUNTRY, 2019-2028 (USD MILLION)
TABLE 355 MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 356 MIDDLE EAST INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 357 MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 358 MIDDLE EAST RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 359 MIDDLE EAST SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 360 MIDDLE EAST GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 361 MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 362 MIDDLE EAST LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 363 MIDDLE EAST HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 364 MIDDLE EAST CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 365 MIDDLE EAST AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 366 MIDDLE EAST HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 367 MIDDLE EAST ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 368 MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 369 MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 370 SAUDI ARABIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 371 SAUDI ARABIA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 372 SAUDI ARABIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 373 SAUDI ARABIA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 374 SAUDI ARABIA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 375 SAUDI ARABIAGASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 376 SAUDI ARABIAMOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 377 SAUDI ARABIA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 378 SAUDI ARABIA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 379 SAUDI ARABIA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 380 SAUDI ARABIA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 381 SAUDI ARABIA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 382 SAUDI ARABIA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 383 SAUDI ARABIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 384 SAUDI ARABIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 385 U.A.E MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 386 U.A.E INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 387 U.A.E MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 388 U.A.E RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 389 U.A.E SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 390 U.A.E GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 391 U.A.E MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 392 U.A.E LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 393 U.A.E HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 394 U.A.E CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 395 U.A.E AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 396 U.A.E HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 397 U.A.E ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 398 U.A.E MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 399 U.A.E MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 400 ISRAEL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 401 ISRAEL INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 402 ISRAEL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 403 ISRAEL RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 404 ISRAEL SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 405 ISRAEL GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 406 ISRAEL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 407 ISRAEL LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 408 ISRAEL HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 409 ISRAEL CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 410 ISRAEL AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 411 ISRAEL HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 412 ISRAEL ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 413 ISRAEL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 414 ISRAEL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 415 EGYPT MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 416 EGYPT INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 417 EGYPT MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 418 EGYPT RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 419 EGYPT SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 420 EGYPT GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 421 EGYPT MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 422 EGYPT LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 423 EGYPT HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 424 EGYPT CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 425 EGYPT AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 426 EGYPT HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 427 EGYPT ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 428 EGYPT MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 429 EGYPT MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 430 REST OF MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 431 AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY COUNTRY, 2019-2028 (USD MILLION)
TABLE 432 AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 433 AFRICA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 434 AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 435 AFRICA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 436 AFRICA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 437 AFRICA GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 438 AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 439 AFRICA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 440 AFRICA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 441 AFRICA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 442 AFRICA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 443 AFRICA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 444 AFRICA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 445 AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 446 AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 447 SOUTH AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 448 SOUTH AFRICA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 449 SOUTH AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 450 SOUTH AFRICA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 451 SOUTH AFRICA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 452 SOUTH AFRICA GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 453 SOUTH AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 454 SOUTH AFRICA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 455 SOUTH AFRICA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 456 SOUTH AFRICA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 457 SOUTH AFRICA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 458 SOUTH AFRICA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 459 SOUTH AFRICA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 460 SOUTH AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 461 SOUTH AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 462 KENYA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 463 KENYA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 464 KENYA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 465 KENYA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 466 KENYA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 467 KENYA GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 468 KENYA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 469 KENYA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 470 KENYA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 471 KENYA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 472 KENYA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 473 KENYA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 474 KENYA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 475 KENYA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 476 KENYA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 477 NIGERIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 478 NIGERIA INSTRUMENT IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
TABLE 479 NIGERIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 480 NIGERIA RESPIRATORY INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 481 NIGERIA SEXUALLY TRANSMITTED INFECTION (STI) TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY INDICATION, 2019-2028 (USD MILLION)
TABLE 482 NIGERIA GASTROINTESTINAL TRACT INFECTIONS TESTING IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, INDICATION, 2019-2028 (USD MILLION)
TABLE 483 NIGERIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY END USER, 2019-2028 (USD MILLION)
TABLE 484 NIGERIA LABORATORIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 485 NIGERIA HOSPITALS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 486 NIGERIA CLINICS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 487 NIGERIA AMBULATORY CENTERS IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 488 NIGERIA HOMECARE IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 489 NIGERIA ASSISTED LIVING FACILITIES IN MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY TECHNOLOGY, 2019-2028 (USD MILLION)
TABLE 490 NIGERIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY MODE OF TESTING, 2019-2028 (USD MILLION)
TABLE 491 NIGERIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY DISTRIBUTION CHANNEL, 2019-2028 (USD MILLION)
TABLE 492 REST OF AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET, BY PRODUCT, 2019-2028 (USD MILLION)
FIGURE 1 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: SEGMENTATION
FIGURE 2 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: DATA TRIANGULATION
FIGURE 3 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: DROC ANALYSIS
FIGURE 4 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: GLOBAL VS REGIONAL MARKET ANALYSIS
FIGURE 5 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: MULTIVARIATE MODELLING
FIGURE 8 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: DBMR MARKET POSITION GRID
FIGURE 9 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: VENDOR SHARE ANALYSIS
FIGURE 10 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: END USER OVERAGE GRID
FIGURE 11 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: SEGMENTATION
FIGURE 12 NORTH AMERICA IS EXPECTED TO DOMINATE THE GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET AND IS EXPECTED TO GROW WITH THE HIGHEST CAGR IN THE FORECAST PERIOD
FIGURE 13 INCREASING PREVALENCE OF CHRONIC DISEASE AND IMPROVING DIAGNOSIS-SEEKING RATE ARE EXPECTED TO DRIVE THE GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET IN THE FORECAST PERIOD
FIGURE 14 CONSUMABLES AND REAGENTS SEGMENT IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET IN 2021 & 2028
FIGURE 15 ASIA-PACIFIC IS THE FASTEST GROWING MARKET FOR THE MOLECULAR POINT OF CARE TESTING (USING NAAT) MANUFACTURERS IN THE FORECAST PERIOD
FIGURE 16 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET
FIGURE 17 MEDICAL DEVICE SPENDING VS NATIONAL HEALTH EXPENDITURES, 2016
FIGURE 18 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY PRODUCT, 2020
FIGURE 19 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY PRODUCT, 2021-2028 (USD MILLION)
FIGURE 20 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY PRODUCT, CAGR (2021-2028)
FIGURE 21 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY PRODUCT, LIFELINE CURVE
FIGURE 22 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY INDICATION, 2020
FIGURE 23 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY INDICATION, 2021-2028 (USD MILLION)
FIGURE 24 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY INDICATION, CAGR (2021-2028)
FIGURE 25 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY INDICATION, LIFELINE CURVE
FIGURE 26 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY END USER, 2020
FIGURE 27 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY END USER, 2021-2028 (USD MILLION)
FIGURE 28 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY END USER, CAGR (2021-2028)
FIGURE 29 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY END USER, LIFELINE CURVE
FIGURE 30 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY MODE OF TESTING, 2020
FIGURE 31 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY MODE OF TESTING, 2021-2028 (USD MILLION)
FIGURE 32 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY MODE OF TESTING, CAGR (2021-2028)
FIGURE 33 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY MODE OF TESTING, LIFELINE CURVE
FIGURE 34 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT)MARKET: BY DISTRIBUTION CHANNEL, 2020
FIGURE 35 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT)MARKET: BY DISTRIBUTION CHANNEL, 2021-2028 (USD MILLION)
FIGURE 36 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT)MARKET: BY DISTRIBUTION CHANNEL, CAGR (2021-2028)
FIGURE 37 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT)MARKET: BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 38 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: SNAPSHOT (2020)
FIGURE 39 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY REGION (2020)
FIGURE 40 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY REGION (2021 & 2028)
FIGURE 41 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY REGION (2020 & 2028)
FIGURE 42 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY PRODUCT (2021-2028)
FIGURE 43 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: SNAPSHOT (2020)
FIGURE 44 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020)
FIGURE 45 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2021 & 2028)
FIGURE 46 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020 & 2028)
FIGURE 47 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY PRODUCT (2021-2028)
FIGURE 48 EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: SNAPSHOT (2020)
FIGURE 49 EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020)
FIGURE 50 EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2021 & 2028)
FIGURE 51 EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020 & 2028)
FIGURE 52 EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY PRODUCT (2021-2028)
FIGURE 53 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: SNAPSHOT (2020)
FIGURE 54 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020)
FIGURE 55 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2021 & 2028)
FIGURE 56 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020 & 2028)
FIGURE 57 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY PRODUCT (2021-2028)
FIGURE 58 SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: SNAPSHOT (2020)
FIGURE 59 SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020)
FIGURE 60 SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2021 & 2028)
FIGURE 61 SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020 & 2028)
FIGURE 62 SOUTH EAST ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY PRODUCT (2021-2028)
FIGURE 63 MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: SNAPSHOT (2020)
FIGURE 64 MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020)
FIGURE 65 MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2021 & 2028)
FIGURE 66 MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020 & 2028)
FIGURE 67 MIDDLE EAST MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY PRODUCT (2021-2028)
FIGURE 68 AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: SNAPSHOT (2020)
FIGURE 69 AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020)
FIGURE 70 AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2021 & 2028)
FIGURE 71 AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY COUNTRY (2020 & 2028)
FIGURE 72 AFRICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: BY PRODUCT (2021-2028)
FIGURE 73 GLOBAL MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: COMPANY SHARE 2020 (%)
FIGURE 74 NORTH AMERICA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: COMPANY SHARE 2020 (%)
FIGURE 75 EUROPE MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: COMPANY SHARE 2020 (%)
FIGURE 76 SOUTH ASIA MOLECULAR POINT OF CARE TESTING (USING NAAT) MARKET: COMPANY SHARE 2020 (%)
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.