Europe Viscosupplementation Market
Market Size in USD Billion
CAGR :
% 
 USD
479.07 Million
USD
886.72 Million
2023
2031
USD
479.07 Million
USD
886.72 Million
2023
2031
| 2024 –2031 | |
| USD 479.07 Million | |
| USD 886.72 Million | |
|
|
|
Europe Viscosupplementation Market, By Source (Non-Animal Origin and Animal Origin), Molecular Weight (Intermediate Molecular Weight, Low Molecular Weight, and High Molecular Weight), Injection (Single Injection, Three Injection, and Five Injection), Age Group (Geriatric and Adult), End User (Hospitals, Orthopaedic Clinics, Ambulatory Care Center, and Others), Distribution Channel (Direct Tender and Retail Sales) – Industry Trends and Forecast to 2031.
Europe Viscosupplementation Market Analysis and Insights
In the viscosupplementation market, advancements in product formulations and delivery methods have led to improved efficacy and patient outcomes. These innovations benefit both patients and healthcare providers by offering enhanced pain relief, prolonged joint functionality, and increased treatment options for osteoarthritis. Overall, the evolution of viscosupplementation contributes to better quality of life for individuals suffering from joint-related conditions.
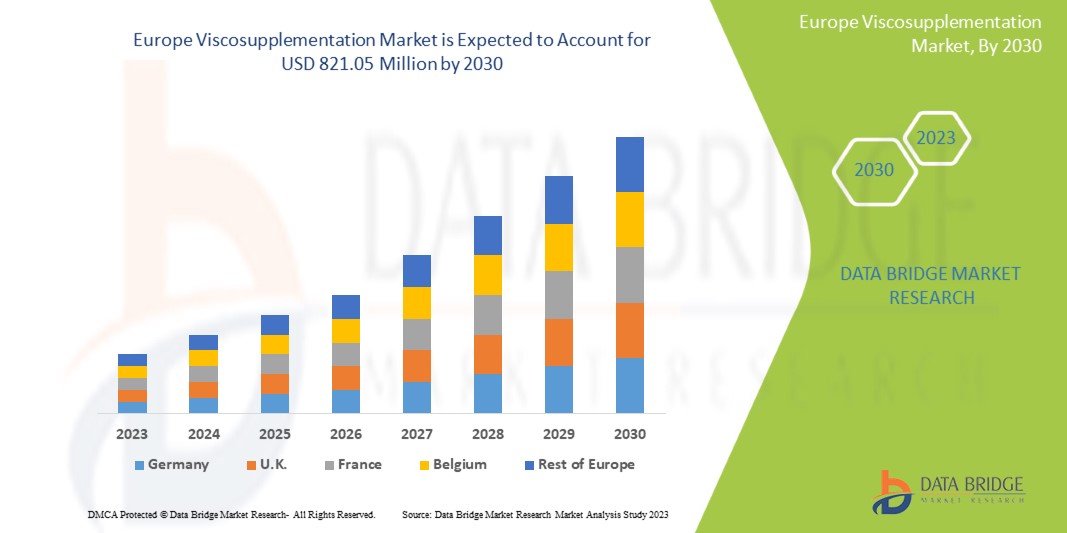
The Europe viscosupplementation market size was valued at USD 479.07 million in 2023, is projected to reach USD 886.72 million by 2031, with a CAGR of 8.00% during the forecast period 2024 to 2031. This indicates that the market value. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
Report Scope and Market Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2024 to 2031 |
|
Base Year |
2023 |
|
Historic Years |
2022 (Customizable to 2016-2021) |
|
Quantitative Units |
Revenue in USD Million, Volumes in Units, Pricing in USD |
|
Segments Covered |
Source (Non-Animal Origin and Animal Origin), Molecular Weight (Intermediate Molecular Weight, Low Molecular Weight, and High Molecular Weight), Injection (Single Injection, Three Injection, and Five Injection), Age Group (Geriatric and Adult), End User (Hospitals, Orthopaedic Clinics, Ambulatory Care Center, and Others), Distribution Channel (Direct Tender and Retail Sales) |
|
Countries Covered |
Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe |
|
Market Players Covered |
Anika Therapeutics, Inc. (U.S.), SEIKAGAKU CORPORATION (Japan), Bioventus (U.S.), Fidia Farmaceutici S.p.A (Italy), Ferring B.V (Netherlands), Sanofi-Aventis U.S. LLC (France), Zimmer Biomet (U.S.), APTISSEN (U.S.), Johnson & Johnson Services, Inc. (U.S.), LG Chem (South Korea), Viatris Inc. (U.S.), IBSA Institut Biochimique SA (Switzerland), TRB CHEMEDICA SA (Switzerland), and Teva Pharmaceutical Industries Ltd. (Israel) |
|
Market Opportunities |
|
Market Definition
Viscosupplementation is a therapeutic process in which the pathological synovial fluid or effusion is removed from the joint by arthrocentesis and is replaced with a highly purified hyaluronan solution which has a 16- to 30-times greater concentration than the pathological joint fluid, and a 2- to 5-times than that of the hyaluronan in the healthy synovial fluid. In some preparations of hyaluronan for therapeutic purposes, the rheological properties (viscosity and elasticity) are low, and therefore the elastoviscosity of the fluid is similar to that of fluid removed from an arthritic joint. Another preparation available to patients worldwide is composed of hyaluronan derivatives (hylans) with substantially greater elastoviscosity than hyaluronan solutions and has comparable rheological properties to those of the fluid found in young, healthy individuals.
Europe Viscosupplementation Market Dynamics
Drivers
- Increasing Preference for Non-Surgical Treatments
With a growing emphasis on minimally invasive procedures and avoiding surgery whenever possible, viscosupplementation offers a viable alternative to invasive joint surgeries for osteoarthritis management, boosting its adoption. For instance, in the United States, where over 30 million adults are affected by osteoarthritis, there's a growing inclination towards non-surgical options. Viscosupplementation injections provide symptomatic relief and improve joint function without the risks and recovery time associated with surgery, fostering its widespread adoption.
- Advancement in Technology
Ongoing advancements in medical technology are leading to the development of more advanced viscosupplements with improved efficacy and longer-lasting effects. These innovations attract both patients and healthcare providers, contributing to market growth. For instance, the development of cross-linked hyaluronic acid formulations has led to longer-lasting effects compared to traditional viscosupplements. These innovations not only attract patients seeking more effective pain relief and improved joint function but also garner the interest of healthcare providers, driving the expansion of the viscosupplementation market.
Opportunities
- Patient Preference for Pain Management
Patients are increasingly seeking non-opioid options for managing osteoarthritis pain to enhance their quality of life. Viscosupplementation stands out as a preferred choice due to its ability to alleviate joint pain without the risks associated with opioids. For instance, a recent study published in the Journal of Arthroplasty demonstrated that viscosupplementation for knee are effective in reducing pain scores for osteoarthritis patients, highlighting its appeal as a safe and effective pain management solution.
- Increasing Prevalence of Osteoarthritis
The global prevalence of osteoarthritis is escalating, driven by sedentary lifestyles, rising obesity rates, and sports-related injuries. This surge in affected individuals creates a substantial demand for viscosupplementation treatments. For instance, in the U.S. alone, osteoarthritis affects over 32.5 million adults, a figure expected to climb due to these contributing factors. Consequently, the market for viscosupplementation therapies is propelled by the expanding pool of patients seeking relief from joint pain and stiffness.
Restraints/Challenges
- Rising Competition from Alternative Therapies
Alternative therapies such as oral pain medications, physical therapy, and surgery pose significant competition to viscosupplementation. They are often perceived as more cost-effective and reliable options by patients and healthcare providers. This perception hampers the adoption of viscosupplementation, particularly in cases where alternatives offer similar or better outcomes.
- High Cost of Treatment
Viscosupplementation injection cost are high which obstruct treatments, especially for uninsured patients, severely restrict accessibility and adoption rates. This financial barrier hampers market growth, as many individuals cannot afford the expensive procedures, thereby limiting the potential reach and impact of viscosupplementation in addressing joint-related conditions.
This market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Recent Development
- In May 2022, Fidia Farmaceutici S.p.A. introduced its innovative Aesthetic Care portfolio featuring ACP technology at the 20th AMWC in Monte Carlo, showcasing its Hyal System and Hy-Tissue in Spain, highlighting hyaluronic acid research
- In December 2021, Avanos Medical acquired OrthogenRx, Inc., bolstering its viscosupplementation therapies with FDA-approved GenVisc850 and TriVisc for knee osteoarthritis treatment
- In August 2021, SEIKAGAKU CORPORATION launched HyLink in Taiwan, an intra-articular single-injection for viscosupplementation knee osteoarthritis, expanding its portfolio through TCM Biotech International Corp. HyLink, containing a cross-linked hyaluronate hydrogel, demonstrates Seikagaku's advanced cross-linking technology application
Europe Viscosupplementation Market Scope
The market is segmented on the basis of source, age group, molecular weight, injection, end user, and distribution channel. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Source
- Non-Animal Origin
- Animal Origin
Molecular Weight
- Intermediate Molecular Weight
- Low Molecular Weight
- High Molecular Weight
Injection
- Single Injection
- Three Injection
- Five Injection
Age Group
- Geriatric
- Adult
End User
- Hospitals
- Orthopedic Clinics
- Ambulatory Care Center
- Others
Distribution Channel
- Direct Tender
- Retail Sales
Europe Viscosupplementation Market Regional Analysis/Insights
The market is analysed and market size insights and trends are provided by country, source, age group, molecular weight, injection, end user, and distribution channel as referenced above.
The countries covered in the market report are Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, and Rest of Europe in Europe.
Germany is expected to dominate the viscosupplementation market, driven by a preference for shorter treatment regimens. Despite high prices, the rising prevalence of diseases such as osteoarthritis and increasing government approvals for three-injection viscosupplements are projected to fuel segment growth positively.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points such as down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Healthcare Infrastructure growth Installed base and New Technology Penetration
The market also provides you with detailed market analysis for every country growth in healthcare expenditure for capital equipment, installed base of different kind of products for market, impact of technology using life line curves and changes in healthcare regulatory scenarios and their impact on the market. The data is available for historic period 2016-2021.
Competitive Landscape and Europe Viscosupplementation Market Share Analysis
The market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to market.
Some of the major players operating in the market are:
- Anika Therapeutics, Inc. (U.S.)
- SEIKAGAKU CORPORATION (Japan)
- Bioventus (U.S.)
- Fidia Farmaceutici S.p.A (Italy)
- Ferring B.V (Netherlands)
- Sanofi-Aventis U.S. LLC (France)
- Zimmer Biomet (U.S.)
- APTISSEN (U.S.)
- Johnson & Johnson Services, Inc. (U.S.)
- LG Chem (South Korea)
- Viatris Inc. (U.S.)
- IBSA Institut Biochimique SA (Switzerland)
- TRB CHEMEDICA SA (Switzerland)
- Teva Pharmaceutical Industries Ltd. (Israel)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.