Europe Heart Valve Devices Market, By Product Type (Mechanical Heart Valves, Biological Heart Valves and Transcatheter Valves), Treatment (Open Surgery and Minimally Invasive Surgery (MIS)), End User (Hospitals & Clinics, Ambulatory Surgical Centers, Cardiac Centers, Research Centers and Others), Distribution Channel (Direct Tender, Third Party Distributors), Industry Trends and Forecast to 2030.
Europe Heart Valve Devices Market Analysis and Insights
Heart valve devices are used to treat blocked heart valves, and implanting these devices is one of the most common procedures. Structural heart valve devices available on the market include mechanical, biological, and transcatheter valves. The Europe heart valve devices market is likely to grow steadily during the forecast period due to the increasing number of heart valve surgeries across the globe. The growth of the Europe heart valve devices market is anticipated to be driven by developments in structural heart devices and procedures such as aortic valves, left atrial occlusion devices and tissue or biological valves. Tissue valves have already revolutionized the market for heart valve devices. Next-generation heart valve surgeries offering low delivery patient profiles, more controlled surgeries, better valve function, reduced paravalvular regurgitation, increased durability, and lower costs. Product innovations by key market players have boosted the growth of market by enabling them to address a larger patient population and achieve better clinical outcomes.
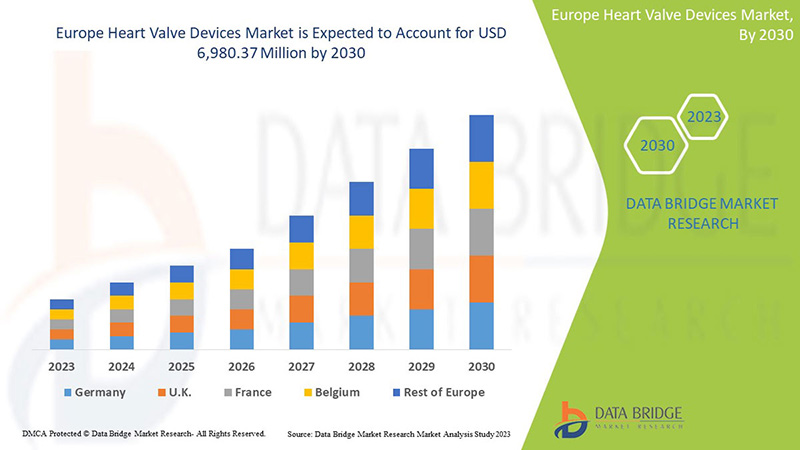
Data Bridge Market Research analyzes that the Europe heart valve devices market is expected to reach the value of USD 6,980.37 million by 2030, at a CAGR of 12.8% during the forecast period. Product type accounts for the largest type segment in the market due to rapid demand of heart valves devices globally. This market report also covers pricing analysis, patent analysis, and technological advancements in depth.
|
Report Metric
|
Details
|
|
Forecast Period
|
2023 to 2030
|
|
Base Year
|
2022
|
|
Historic Years
|
2021 (Customizable to 2020 - 2015)
|
|
Quantitative Units
|
Revenue in USD Million, Volumes in Units, Pricing in USD
|
|
Segments Covered
|
By Product Type (Mechanical Heart Valves, Biological Heart Valves and Transcatheter Valves), Treatment (Open Surgery and Minimally Invasive Surgery (MIS)), End User (Hospitals & Clinics, Ambulatory Surgical Centers, Cardiac Centers, Research Centers and Others), Distribution Channel (Direct Tender, Third Party Distributors).
|
|
Countries Covered
|
Germany, France, U.K., Italy, Russia, Spain, Netherlands, Switzerland, Norway, Poland, Sweden, Belgium, Turkey, Denmark, Finland and the rest of Europe.
|
|
Market Players Covered
|
Abbott, Boston Scientific Corporation or its affiliates, Artivion, Inc., Edwards Lifesciences Corporation, Medtronic, NeoVasc, Micro Interventional Devices Incorporated, XELTIS, TTK, Meril Life Sciences Pvt. Ltd, Foldax, Inc., Venus Medtech (Hangzhou) Inc., Colibri Heart Valve among others.
|
Heart Valve Devices Market Definition
Heart valves are necessary to smooth blood movement in the right direction in the body. Heart valves are responsible for constant blood flow, and maintaining blood pressure. If they don't work properly cutting off the heart causes stenosis. Heart disease generally includes several diseases that significantly affect the heart. The regulation of heart valves has increased rapidly in the last ten years as the number of patients suffering from cardiovascular diseases has increased. The market growth of heart valves is accelerated by factors such as unstable lifestyles, lifestyle diseases, increasing smoking population, aging population, and increasing quality of healthcare, and rapid development of healthcare reimbursement during the forecast period. In addition, the high cost of heart valves and the risk of infection of heart implants may be the reason that is likely to slow down the growth of the heart valve market during the above-mentioned forecast period. Growth in medical business aspects and groundwork in emerging economies is fueling growth in medical tourism, driving the heart valve device market. The need for minimally invasive procedures to treat cardiac abnormalities has greatly increased. Current applications of automation in heart valve surgery, such as Transcatheter Aortic Valve Replacement (TAVR), have given way to a growing variety of similar surgeries. Judging by the increasing proportion of the aging population, the increase in life insurance policies is also expected to contribute to the growth of the heart valve market.
Heart Valve Devices Market Dynamics
This section deals with understanding the market drivers, advantages, opportunities, restraints, and challenges. All of this is discussed in detail below:
Drivers
- Innovations in Heart Valve Devices Offer Improved Clinical Outcome
Launching new products for minimally invasive procedures will likely drive the Europe heart valve devices market. Tissue valves have revolutionized the heart valve market. The next generation of heart valve surgery offers fewer delivery curves, more controlled placement, improved valve function, reduced valve regurgitation, increased durability, and reduced cost. Product innovation has boosted the growth prospects of heart valve device market players as it can treat a larger number of patients with superior clinical outcomes. Despite significant advances in recent years, structural interventional cardiology remains an emerging market with great potential.
Moreover, intelligent designs, new technologies, and biomaterials applications continue to push the boundaries of new product development, ensuring that these devices will be at the forefront of interventional product innovation for years to come. Design innovations are helping market players tap profitable growth opportunities in heart valve devices.
Thus, the growth of the Europe heart valve devices market is expected to propel due to the rise in innovations in heart valve devices.
- Rising number of various heart diseases
Heart attacks and strokes are usually acute events and are primarily caused by a blockage that blocks blood flow to the heart or brain. During the first year of the COVID-19 pandemic, heart disease and stroke deaths increased by 5.8% and 6.8%, respectively. However, the age-associated increases were 1.6% and 1.7% for heart disease and stroke, respectively. The most common cause is the build-up of fatty deposits in the lining of the blood vessels that supply the heart or brain. A stroke can be caused by bleeding or blood clots in a blood vessel in the brain. This trend suggests that the incidence of cardiovascular disease may increase dramatically due to population growth and aging.
Furthermore, High diabetes and blood pressure, high blood cholesterol, and smoking are key risk factors for heart disease. About half of people in the United States (47%) have at least one of these three risk factors. Other medical conditions and lifestyle choices can also put people at a higher risk for heart disease, including unhealthy diet, physical inactivity, and excessive alcohol use.
Moreover, the rising number of heart diseases is expected to act as a driver for the Europe heart valve devices market.
Opportunity
- Increasing Awareness of Prosthetic Devices
A prosthesis is a device designed to replace a missing body part or to improve the function of a body part. Prosthetic heart valves are increasingly used in cases of abnormalities of the natural valves that require intervention. Generally, they can be divided into mechanical heart valves, biological valves, and allogeneic grafts. The goal of artificial valves is to act hemodynamically like a natural valve with minimal side effects. Cardiovascular prostheses devices have been developed to replace damaged heart tissue. These medical devices are designed to mimic the function of normal cardiovascular organs. Artificial hearts allow heart surgeons to increase the treatment of heart blocks.
Moreover, the prevalence of prosthetic valves ranges from 0.2 per 1,000 in people years of age or younger to 5.3 in people years of age or younger.
Thus the increasing awareness about prosthetic devices acts as an opportunity for the market's growth.
Restraint/Challenge
- Expensive Production Cost of Equipment
The number of patients treated for aortic valve disease in the United States is growing quickly. Transcatheter aortic valve replacement (TAVR) replaces surgical aortic valve replacement (SAVR) and medical treatment (M.T.). The economic consequences of these trends are unknown. Therefore, the total cost of TAVR is higher than SAVR and much more expensive than M.T. alone. TAVR costs have decreased over time, while SAVR and M.T. costs have remained the same.
- In addition, the high surgical cost of TAVI is mainly due to the high cost of the production. However, due to the shorter hospital stay, the cost of non-surgical TAVI is lower compared to AVR. The cost of the TAVI implant kit alone (valve, balloon, sheath) is $32,500, while the surgical valve only costs about $5,000, just like in Switzerland, the TAVI implant set costs about 32,000 francs (around of $35,000), while the cost of a biosurgical prosthesis is about 3,000 Swiss francs (about US$3,300). Depending on comorbidities and complications, reimbursement in the United States is between $ 0,000 and $ 5,000, TAVI in Switzerland is about 72,000 francs ($78,000), and AVR is about 3,000 francs ($ 7,000), which means a financial loss for the hospital.
Thus, the rising production cost of equipment may hamper the market's growth.
Recent Developments
- In September 2023, Abbott released data from five late-stage presentations showing the benefits of its minimally invasive devices in treating people with various structural heart diseases. The data includes findings that support the value of MitraClip™. It is the world's first and leading transcatheter edge-to-edge repair (TEER) to treat leaky valves in people with mitral regurgitation (MR). New data on Abbott's structural heart therapies were presented at the Cardiovascular Research Foundation's 34th Annual Transcatheter Cardiovascular Therapy (TCT) Scientific Symposium in Boston. This has helped the company to increase its business position in the market.
- In September 2020, Boston Scientific Corporation announced that it had introduced a controlled launch of the ACURATE neo2™ aortic valve system in Europe. This next-generation transcatheter aortic valve implantation (TAVI) technology is a new platform designed with multiple features to improve the clinical performance of the original ACURATE new platform. Compared to the previous generation, the ACURATE neo2 valve system has an expanded indication for patients with aortic stenosis. This has helped the company to gain its product portfolio.
Heart Valve Devices Market Scope
The heart valve devices market is segmented into product type, treatment, end user, and distribution channel. The growth among segments helps you analyze niche pockets of growth and strategies to approach the market and determine your core application areas and the difference in your target markets.
BY PRODUCT TYPE
- Mechanical Heart Valves
- Biological Heart Valve
- Transcatheter Valves
On the basis of product type, the heart valve devices market is segmented into mechanical heart valve, biological heart valve and transcatheter valve.
BY TREATMENT
- OPEN SURGERY
- MINIMALLY INVASIVE SURGERY
On the basis of treatment, the heart valve devices market is segmented into open surgery and minimal invasive surgery.
BY END USER
- HOSPITAL & CLINICS
- AMBULATORY SURGICAL CENTERS
- CARDIAC CENTERS
- RESEARCH CENTERS
- OTHERS
On the basis of end user, the heart valve devices market is segmented into hospitals & clinics, ambulatory surgical centers, cardiac centers, research centers and others.
BY DISTRIBUTION CHANNEL
- DIRECT TENDER
- THIRD PARTY DISTRIBUTORS
On the basis of distribution channel, the heart valve devices market is segmented into direct tender and third party distributors.
Europe Heart Valve Devices Market Regional Analysis/Insights
The heart valve devices market is analyzed and market size information is provided product type, treatment, end user, and distribution channel.
The countries covered in this market report Germany, France, U.K., Italy, Russia, Spain, Netherlands, Switzerland, Norway, Poland, Sweden, Belgium, Turkey, Denmark, Finland and the rest of Europe.
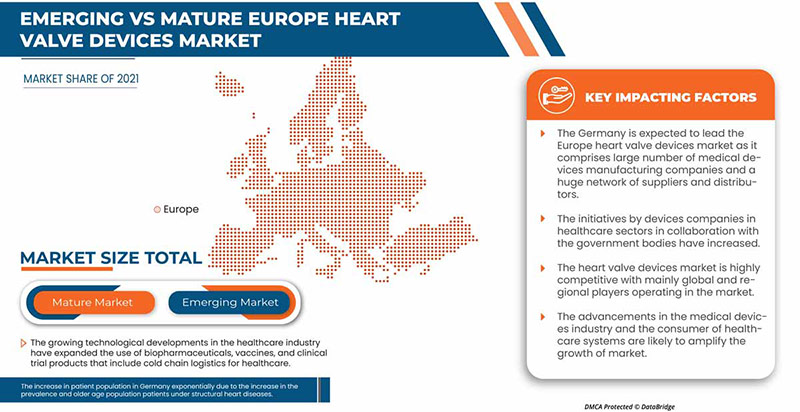
Germany dominates Europe region due to the mass production of heart valve devices and increasing demand from emerging markets and expansion of healthcare industries.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impact the current and future trends of the market. Data points such as new sales, replacement sales, country demographics, regulatory acts, and import-export tariffs are some of the major pointers used to forecast the market scenario for individual countries. Also, presence and availability of Europe brands and their challenges faced due to large or scarce competition from local and domestic brands, and impact of sales channels are considered while providing forecast analysis of the country data.
Competitive Landscape and Heart Valve Devices Market Share Analysis
The heart valve devices market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in R&D, new market initiatives, production sites and facilities, company strengths and weaknesses, product launch, product trials pipelines, product approvals, patents, product width and breath, application dominance, technology lifeline curve. The above data points provided are only related to the company’s focus on the heart valve devices market.
Some of the key players operating in the heart valve devices market are Abbott, Boston Scientific Corporation or its affiliates, Artivion, Inc., Edwards Lifesciences Corporation, Medtronic, NeoVasc, Micro Interventional Devices Incorporated, XELTIS, TTK, Meril Life Sciences Pvt. Ltd, Foldax, Inc., Venus Medtech (Hangzhou) Inc., Colibri Heart Valve among others.
Research Methodology: Heart Valve Devices Market
Data collection and base year analysis are done using data collection modules with large sample sizes. The market data is analyzed and estimated using market statistical and coherent models. In addition, market share analysis and key trend analysis are the major success factors in the market report. The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market, and primary (industry expert) validation. Apart from this, data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Company Market Share Analysis, Standards of Measurement, Europe vs Regional, and Vendor Share Analysis. Please request analyst call in case of further inquiry.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Factbook) or can assist you in creating presentations from the data sets available in the report.
SKU-

