Middle East and Africa e-Clinical Solutions Market, By Product (Electronic Data Capture and Clinical Trial Data Management Systems, Clinical Trial Management Systems, Clinical Analytics Platforms, Care Coordination Medical Records (CCMR), Randomization and Trial Supply Management, Clinical Data Integration Platforms, Electronic Clinical Outcome Assessment Solutions, Safety Solutions, Electronic Trial Master File Systems, Regulatory Information Management Solutions, and Others), Delivery Mode (Web- hosted (On-Demand) Solutions, Licensed Enterprise (On-Premises) Solutions and Cloud-Based (SAAS) Solutions), Clinical Trial Phase (Phase I, Phase II, Phase III, and Phase IV), Organization Size (Small & Medium and Large), User Device (Desktop, Tablet, Handheld PDA Device, Smart Phone, and Others), End User (Pharmaceutical and Biopharmaceutical Companies, Contract Research Organizations, Consulting Service Companies, Medical Device Manufacturers, Hospitals, and Academic Research Institutes), Industry Trends and Forecast to 2030.
Middle East and Africa e-Clinical Solutions Market Analysis and Size
The Middle East and Africa e-clinical solutions market is fragmented, as it consists of many global players such as Oracle, IQVIA Inc., Dassault Systemes, and Clario among others. The presence of these companies produces competitive prices for systems services and software across the region. Due to the presence of these players at regional and international levels, suppliers and manufacturers offer products with different specifications and characteristics in all budgets. The growing adoption of electronic data capture (EDC) systems and cloud-based is driving the market growth. Additionally, the growing focus on patient-centric clinical trials is expected to drive market growth.
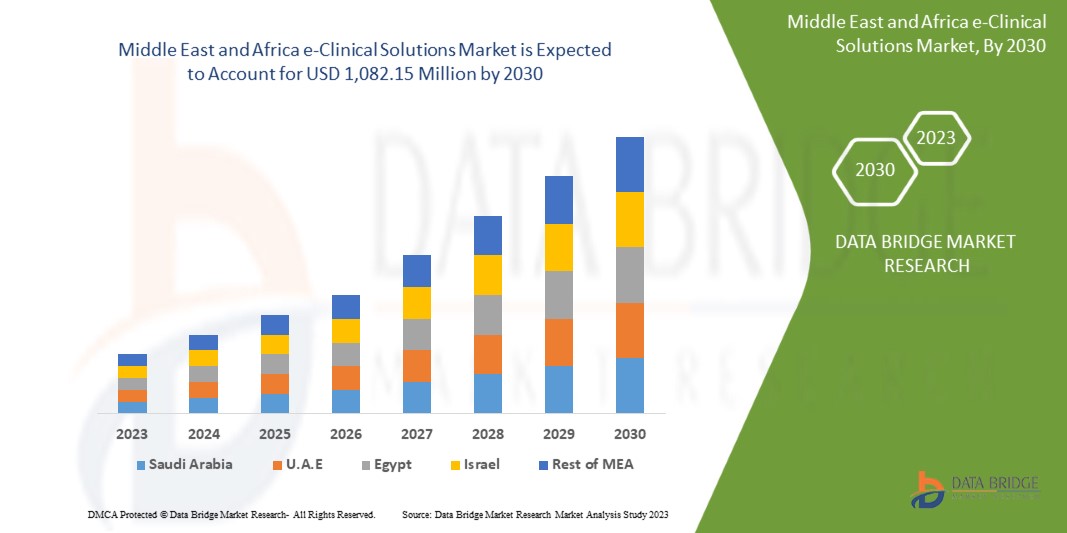
Data Bridge Market Research analyzes that the Middle East and Africa e-clinical solutions market is expected to reach a value of USD 1,082.15 million by 2030, at a CAGR of 11.9% during the forecast period. This market report also covers pricing analysis and technological advancements in depth.
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015 – 2020) |
|
Quantitative Units |
Revenue in USD Million, Volumes in Units, and Pricing in USD |
|
Segments Covered |
Por produto (captura eletrónica de dados e sistemas de gestão de dados de ensaios clínicos, sistemas de gestão de ensaios clínicos, plataformas de análise clínica, registos médicos de coordenação de cuidados (CCMR), randomização e gestão da prestação de ensaios, plataformas de integração de dados clínicos, soluções eletrónicas de avaliação de resultados clínicos, soluções de segurança, Sistemas de ficheiros principais de testes eletrónicos, soluções de gestão de informação regulamentar e outros), modo de entrega (soluções alojadas na Web (on-demand), soluções empresariais licenciadas (on-site) e soluções baseadas na cloud (SAAS)), fase de ensaio clínico (Fase I, Fase II, Fase III e Fase IV), Tamanho da organização (Pequeno, Médio e Grande), Dispositivo do utilizador (desktop, tablet , dispositivo PDA portátil, smartphone e outros), Utilizador final (empresas farmacêuticas e biofarmacêuticas , organizações de investigação por contrato, empresas de serviços de consultoria, fabricantes de dispositivos médicos, hospitais e institutos de investigação académica) |
|
Países abrangidos |
África do Sul, Arábia Saudita, Emirados Árabes Unidos, Egito, Israel e o resto do Médio Oriente e África |
|
Participantes do mercado abrangidos |
Oracle, Signant Health, MaxisIT, Paraxel International Corporation, Dassault Systemes, Clario, Mednet, OpenClinica, LLC, 4G Clinical, Veeva Systems, Saama Technologies, LLC, Anju, Castor, Medrio, Inc., ArisGlobal, Merative, Advarra, eClinical Solutions , LLC, Y-Prime LLC, RealTime Software Solutions LLC, Quretec, Research Manager, Datatrack Int., e IQVIA Inc.
|
Definição de Mercado
O objetivo das soluções e-clínicas é revolucionar o campo da investigação clínica. A organização de gestão clínica está a tentar melhorar a eficácia, eficiência e acessibilidade dos dados de investigação clínica para tratar os doentes mais rapidamente. A ideia por detrás da criação de eClinical Solutions foi identificar os problemas com os dados de investigação clínica, corrigi-los e oferecer soluções criativas para tornar os dados de ensaios clínicos úteis e simples de obter, bem como facilitar a padronização, a elaboração de relatórios e a operação do domínio da investigação clínica. Espera-se que a crescente adoção e o crescente foco em soluções e software e-clínico impulsionem o crescimento do mercado.
As soluções e-clínicas, como a captura eletrónica de dados (EDC), os resultados eletrónicos reportados pelo doente (ePRO), os sistemas de gestão de ensaios clínicos (CTMS) e os sistemas de ficheiro mestre de ensaios eletrónicos (eTMF), são programas de software utilizados. em investigação clínica para gerir dados clínicos. No entanto, espera-se que os regulamentos e normas rigorosos para a aprovação de estudos de ensaios clínicos e aprovações de produtos restrinjam o crescimento do mercado.
Dinâmica do mercado de soluções e-clínicas no Médio Oriente e África
Esta secção trata da compreensão dos impulsionadores, oportunidades, restrições e desafios do mercado. Tudo isto é discutido em detalhe abaixo:
MOTORISTAS
Aumento das atividades de I&D e das despesas correspondentes
A investigação e o desenvolvimento são cruciais em todos os setores, incluindo as ciências da vida e os cuidados de saúde. A indústria das ciências da vida realiza uma extensa investigação e desenvolvimento para gerar receitas e, muitas vezes, traz resultados que incluem salvar ou melhorar a vida de um paciente. O desenvolvimento de medicamentos exige a realização de ensaios clínicos para verificar a análise de dados clínicos e rever o seu estudo e novos insights de dados.
As soluções clínicas eletrónicas são utilizadas para a revisão abrangente dos dados, automatizando o processo de transformação de dados e análises para acelerar os prazos. As empresas farmacêuticas e biofarmacêuticas conduzem muitos ensaios clínicos para concluir o desenvolvimento bem-sucedido de medicamentos ou produtos biológicos. As empresas farmacêuticas e biofarmacêuticas aumentaram o seu foco na investigação e desenvolvimento, aumentando a procura por soluções clínicas eletrónicas.
Melhoria das infraestruturas de saúde e estabelecimento de laboratórios avançados
As infraestruturas são um pilar fundamental que apoia o objetivo fundamental de promover melhores padrões de cuidados e bem-estar para todos os doentes, juntamente com uma boa experiência no sistema de saúde. Paralelamente, o sistema de saúde e o pessoal devem apoiar a promoção eficaz da saúde, a prevenção e o autocuidado de toda a população.
A infra-estrutura deve integrar o hospital, como centro de cuidados agudos e de internamento, no sistema de saúde mais amplo. Deve facilitar os sete domínios da experiência de qualidade do doente, eficácia, eficiência, oportunidade, segurança, equidade e sustentabilidade. A infraestrutura inclui o ambiente construído e elementos de suporte, como equipamentos, acesso, tecnologia de informação (TI), sistemas e processos, iniciativas de sustentabilidade e pessoal. Isto melhorou a infraestrutura de cuidados de saúde e proporcionou mais oportunidades para que as soluções clínicas eletrónicas crescessem e produzissem melhores resultados.
OPORTUNIDADE
Aumentar os investimentos de vários governos em ensaios clínicos
O financiamento dos ensaios clínicos provém de diversas fontes, incluindo o governo, investidores comerciais, organizações sem fins lucrativos, instituições académicas e outras organizações de investigação. Historicamente, os Institutos Nacionais de Saúde (NIH) fizeram os maiores investimentos governamentais em investigação fundamental para o desenvolvimento de medicamentos. A agência de investigação avançada de defesa (DARPA) também contribuiu para a fase de descoberta ao aceitar algumas iniciativas biológicas de risco relativamente elevado. Os governos estaduais também estão a assumir cada vez mais a liderança nesta área, em parte devido à frustração do público com o longo processo de descoberta. Assim, espera-se que as iniciativas governamentais e os investimentos para a descoberta de novos medicamentos com estudos experimentais criem uma oportunidade para o crescimento do mercado.
RESTRIÇÃO / DESAFIO
Questões de segurança e privacidade de dados
Os sistemas de informação estão fortemente preocupados com a privacidade e a segurança. O acesso a informação individual de saúde é possível através da digitalização dos serviços de saúde a partir de qualquer dispositivo eletrónico com ligação à Internet em qualquer parte do mundo. Os utilizadores normalmente não têm conhecimento de como os seus dados são tratados. Os dados de saúde na cloud chamaram a atenção dos hackers, que visam sistemas para lançar ataques e roubar dados confidenciais em troca de benefícios financeiros.
As violações de dados têm sido comuns desde 2019, mais de 50 milhões de registos médicos eletrónicos foram pirateados em 2021 e prevê-se que o número de violações de segurança aumente a cada ano. A criação de acordos interjurisdicionais de partilha de dados e o armazenamento e manipulação de acervos de dados são significativamente dificultados por preocupações sobre a privacidade pessoal e a confidencialidade da informação e pela recente promulgação de legislação sobre privacidade e confidencialidade nas províncias e territórios (excepcionalmente registos de doentes). Assim, é uma restrição para o mercado de soluções clínicas electrónicas do Médio Oriente e de África e deverá dificultar o crescimento do mercado.
Impacto pós-COVID-19 no mercado de soluções e-clínicas no Médio Oriente e em África
A COVID-19 impactou positivamente o mercado de soluções e-clínicas no Médio Oriente e em África. A restrição do bloqueio levou ao surgimento de várias oportunidades, como o desenvolvimento de medicamentos, a instalação de aplicações EMR e EHR, e muitas outras.
No entanto, espera-se que o aumento do apoio governamental e as técnicas avançadas e inovadoras proporcionem oportunidades lucrativas para o crescimento do mercado. Além disso, espera-se que o aumento das parcerias, aquisições e colaboração entre os participantes do mercado alimente ainda mais o crescimento do mercado. Além disso, o crescimento tem sido elevado desde a abertura do mercado após a COVID-19, e prevê-se que haja um crescimento considerável no sector. Os participantes do mercado estão a realizar diversas atividades para melhorar as técnicas de estudo experimental. Com isto, as empresas trarão avanço e inovação ao mercado.
Desenvolvimentos recentes
- Em abril de 2023, a subsidiária Medidata da Dassault Systèmes anunciou que a Lambda Therapeutics está a implementar os produtos clínicos baseados na cloud da Medidata, Rave EDC, Rave RTSM e Rave Imaging, de acordo com um comunicado da Medidata, subsidiária da Dassault Systèmes. Automatizar e otimizar as operações de gestão de dados e fornecer com segurança dados de maior qualidade para obter insights mais rápidos melhorará ainda mais a produtividade dos ensaios clínicos. Isto ajudou a empresa a promover as suas ofertas em todo o mundo
- Em março de 2023, a Clario lançou uma ferramenta Image Viewer baseada na cloud que ajuda os patrocinadores e os CRO a ver as imagens dos seus ensaios clínicos. Anteriormente, várias organizações tinham de participar no procedimento de transferência de imagens para ver as fotos de um ensaio clínico. Isto complicou o processo já arriscado e aumentou a possibilidade de atrasos e erros. Isto ajudou a empresa a aumentar a sua oferta de serviços
Âmbito de mercado das soluções e-clínicas no Médio Oriente e África
O mercado de soluções e-clínicas do Médio Oriente e África está segmentado em seis segmentos notáveis com base no produto, modo de entrega, fase de ensaio clínico, tamanho da organização, dispositivo do utilizador e utilizador final. O crescimento entre estes segmentos irá ajudá-lo a analisar os escassos segmentos de crescimento nas indústrias e fornecer aos utilizadores uma valiosa visão geral do mercado e insights para os ajudar a tomar decisões estratégicas para identificar as principais aplicações do mercado.
Produto
- Sistemas eletrónicos de captura de dados e gestão de dados clínicos
- Sistemas de gestão de ensaios clínicos
- Plataformas de análise clínica
- Registo Médico da Coordenação de Cuidados (CCMR)
- Randomização e gestão de fornecimento de testes
- Plataformas de integração de dados clínicos
- Soluções eletrónicas de avaliação de resultados clínicos
- Soluções de segurança
- Sistemas de ficheiros mestre de teste eletrónico
- Soluções de gestão de informações regulamentares
- Outros
Com base no produto, o mercado está segmentado em captura eletrónica de dados e gestão de dados clínicos, sistemas de gestão de ensaios clínicos, plataformas de análise clínica, registo médico de coordenação de cuidados (CCMR), randomização e gestão de fornecimento de ensaios, plataformas de integração de dados clínicos, soluções eletrónicas de avaliação de resultados clínicos , soluções de segurança, sistemas de arquivo mestre de testes eletrónicos, soluções de gestão de informação regulamentar e outros.
Modo de entrega
- Soluções alojadas na Web (on demand)
- Soluções empresariais licenciadas (on-site)
- Soluções baseadas na cloud (SAAS)
Com base no modo de entrega, o mercado está segmentado em soluções alojadas na web (on-demand), soluções empresariais licenciadas (on-premises) e soluções baseadas na cloud (SAAS).
Fase de ensaio clínico
- Fase I
- Fase II
- Fase III
- Fase IV
Com base na fase de ensaio clínico, o mercado está segmentado em Fase I, Fase II, Fase III e Fase IV.
Tamanho da organização
- Pequeno e médio
- Grande
Com base no tamanho da organização, o mercado está segmentado em pequena, média e grande dimensão.
Dispositivo do utilizador
- Área de trabalho
- Comprimido
- Dispositivo PDA portátil
- Telefone inteligente
- Outros
Com base no dispositivo do utilizador, o mercado está segmentado em desktop, tablet, dispositivo PDA portátil, smartphone, entre outros.
Utilizador final
- Empresas farmacêuticas e biofarmacêuticas
- Organizações de investigação contratada
- Empresas de serviços de consultoria
- Fabricantes de dispositivos médicos
- Hospitais
- Institutos de Investigação Académica
Com base no utilizador final, o mercado está segmentado em empresas farmacêuticas e biofarmacêuticas, organizações de investigação por contrato, empresas de serviços de consultoria, fabricantes de dispositivos médicos, hospitais e institutos de investigação académica.
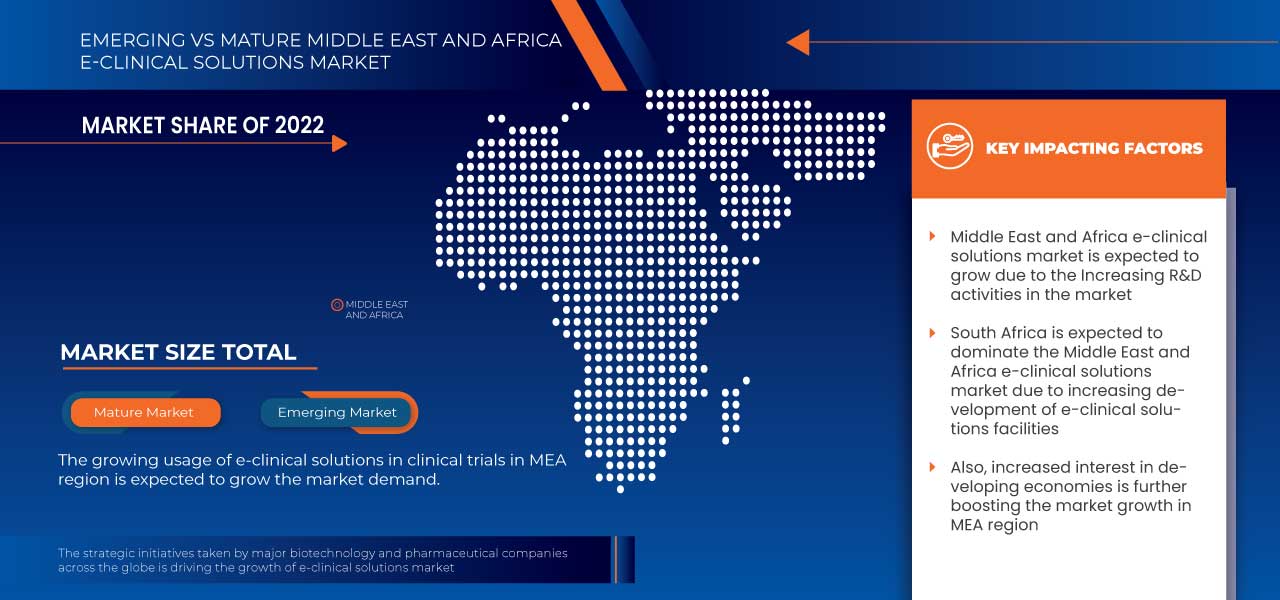
Análise/perspetivas regionais do mercado de soluções e-clínicas no Médio Oriente e África
O mercado de soluções e-clínicas do Médio Oriente e África é analisado, e as informações sobre o tamanho do mercado são baseadas no produto, modo de entrega, fase de ensaio clínico, tamanho da organização, dispositivo do utilizador e utilizador final.
Os países abordados neste relatório de mercado são a África do Sul, Arábia Saudita, Emirados Árabes Unidos, Egito, Israel e o resto do Médio Oriente e África.
Espera-se que a África do Sul domine a região do Médio Oriente e de África devido à crescente adopção de sistemas electrónicos de captura de dados e de soluções baseadas na nuvem.
A secção de países do relatório também fornece fatores individuais que impactam o mercado e alterações na regulamentação do mercado interno que impactam as tendências atuais e futuras do mercado. Dados como novas vendas, vendas de reposição, demografia do país, atos regulamentares e tarifas de importação e exportação são alguns dos principais indicadores utilizados para prever o cenário de mercado para países individuais. Além disso, são considerados a presença e disponibilidade de marcas da Ásia-Pacífico e os desafios enfrentados devido à concorrência grande ou escassa de marcas locais e nacionais e o impacto dos canais de vendas, ao mesmo tempo que se fornece uma análise de previsão dos dados do país.
Cenário competitivo e análise da quota de mercado de soluções e-clínicas no Médio Oriente e África
O panorama competitivo do mercado de soluções e-clínicas no Médio Oriente e em África fornece detalhes do concorrente. Os detalhes incluem a visão geral da empresa, finanças, receitas geradas, potencial de mercado, novas iniciativas de mercado, presença global, localizações e instalações de produção, capacidades de produção, pontos fortes e fracos da empresa, lançamento de produtos, largura e amplitude do produto e domínio de aplicação. Os dados acima fornecidos estão apenas relacionados com o foco das empresas em relação ao mercado.
Alguns dos principais players de mercado que operam no mercado de soluções clínicas eletrónicas no Médio Oriente e em África são a Oracle, Signant Health, MaxisIT, Paraxel International Corporation, Dassault Systemes, Clario, Mednet, OpenClinica, LLC, 4G Clinical, Veeva Systems, Saama Technologies, LLC, Anju, Castor, Medrio, Inc., ArisGlobal, Merative, Advarra, eClinical Solutions, LLC, Y-Prime LLC e RealTime Software Solutions, entre outros.
SKU-
Obtenha acesso online ao relatório sobre a primeira nuvem de inteligência de mercado do mundo
- Painel interativo de análise de dados
- Painel de análise da empresa para oportunidades de elevado potencial de crescimento
- Acesso de analista de pesquisa para personalização e customização. consultas
- Análise da concorrência com painel interativo
- Últimas notícias, atualizações e atualizações Análise de tendências
- Aproveite o poder da análise de benchmark para um rastreio abrangente da concorrência
Índice
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW
1.4 CURRENCY AND PRICING
1.5 LIMITATIONS
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 DBMR TRIPOD DATA VALIDATION MODEL
2.5 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.6 MULTIVARIATE MODELLING
2.7 MARKET END USER COVERAGE GRID
2.8 PRODUCT LIFELINE CURVE
2.9 DBMR MARKET POSITION GRID
2.1 VENDOR SHARE ANALYSIS
2.11 SECONDARY SOURCES
2.12 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PORTER'S FIVE FORCES MODEL
4.2 ECOSYSTEMS ANALYSIS OF E CLINICAL SOLUTIONS
4.3 USE CASES
5 VALUE CHAIN ANALYSIS
6 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: REGULATIONS
7 MARKET OVERVIEW
7.1 DRIVERS
7.1.1 THE GROWING USAGE OF E-CLINICAL SOLUTIONS IN CLINICAL TRIALS
7.1.2 INCREASING R&D ACTIVITIES AND CORRESPONDING EXPENDITURE
7.1.3 STRATEGIC INITIATIVES TAKEN BY MAJOR BIOTECHNOLOGY AND PHARMACEUTICAL COMPANIES
7.1.4 IMPROVED HEALTHCARE INFRASTRUCTURE AND ESTABLISHMENT OF ADVANCED LABORATORIES
7.2 RESTRAINTS
7.2.1 DATA SAFETY AND PRIVACY ISSUES
7.2.2 LACK OF SKILLED PROFESSIONALS
7.2.3 LIMITED ADOPTION OF E-CLINICAL SOLUTIONS IN EMERGING COUNTRIES DUE TO BUDGETARY CONSTRAINTS AND POOR MANAGEMENT POLICIES
7.3 OPPORTUNITIES
7.3.1 RISING ADOPTION OF ELECTRONIC DATA CAPTURE (EDC) SYSTEMS AND CLOUD-BASED SOLUTIONS
7.3.2 INCREASING INVESTMENTS BY VARIOUS GOVERNMENTS IN CLINICAL TRIALS
7.3.3 GROWING FOCUS ON PATIENT-CENTRIC CLINICAL TRIALS
7.4 CHALLENGES
7.4.1 REGULATORY CHALLENGES ASSOCIATED WITH E-CLINICAL SOLUTIONS
7.4.2 HIGH IMPLEMENTATION COSTS
8 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY PRODUCT
8.1 OVERVIEW
8.2 ELECTRONIC DATA CAPTURE AND CLINICAL DATA MANAGEMENT SYSTEMS
8.3 RANDOMIZATION AND TRIAL SUPPLY MANAGEMENT
8.4 CLINICAL TRIAL MANAGEMENT SYSTEMS
8.5 ELECTRONIC TRIAL MASTER FILE SYSTEMS
8.6 ELECTRONIC CLINICAL OUTCOME ASSESSMENT SOLUTIONS
8.7 SAFETY SOLUTIONS
8.8 REGULATORY INFORMATION MANAGEMENT SOLUTIONS
8.9 CLINICAL DATA INTEGRATION PLATFORMS
8.1 CLINICAL ANALYTICS PLATFORMS
8.11 CARE COORDINATION MEDICAL RECORD (CCMR)
8.12 OTHERS
9 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY DELIVERY MODE
9.1 OVERVIEW
9.2 WEB-HOSTED (ON-DEMAND) SOLUTIONS
9.3 CLOUD-BASED (SAAS) SOLUTIONS
9.4 LICENSED ENTERPRISE (ON-PREMISES) SOLUTIONS
10 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY CLINICAL TRIAL PHASE
10.1 OVERVIEW
10.2 PHASE III
10.3 PHASE I
10.4 PHASE II
10.5 PHASE IV
11 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY ORGANIZATION SIZE
11.1 OVERVIEW
11.2 MEDIUM AND SMALL
11.3 LARGE
12 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY USER DEVICE
12.1 OVERVIEW
12.2 DESKTOP
12.3 TABLET
12.4 SMART PHONE
12.5 HANDHELD PDA DEVICE
12.6 OTHERS
13 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY END USER
13.1 OVERVIEW
13.2 CONTRACT RESEARCH ORGANIZATIONS
13.3 PHARMACEUTICAL AND BIOPHARMACEUTICAL COMPANIES
13.4 MEDICAL DEVICE MANUFACTURERS
13.5 HOSPITAL
13.6 CONSULTING SERVICE COMPANIES
13.7 ACADEMIC RESEARCH INSTITUTES
14 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY REGION
14.1 MIDDLE EAST AND AFRICA
14.1.1 SOUTH AFRICA
14.1.2 SAUDI ARABIA
14.1.3 U.A.E.
14.1.4 EGYPT
14.1.5 ISRAEL
14.1.6 REST OF MIDDLE EAST AND AFRICA
15 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, COMPANY LANDSCAPE
15.1 COMPANY SHARE ANALYSIS: MIDDLE EAST & AFRICA
16 SWOT ANALYSIS
17 COMPANY PROFILE
17.1 ORACLE
17.1.1 COMPANY SNAPSHOT
17.1.2 REVENUE ANALYSIS
17.1.3 COMPANY SHARE ANALYSIS
17.1.4 PRODUCT PORTFOLIO
17.1.5 RECENT DEVELOPMENT
17.2 DASSAULT SYSTEMES
17.2.1 COMPANY SNAPSHOT
17.2.2 REVENUE ANALYSIS
17.2.3 COMPANY SHARE ANALYSIS
17.2.4 PRODUCT PORTFOLIO
17.2.5 RECENT DEVELOPMENTS
17.3 CLARIO
17.3.1 COMPANY SNAPSHOT
17.3.2 COMPANY SHARE ANALYSIS
17.3.3 PRODUCT PORTFOLIO
17.3.4 RECENT DEVELOPMENT
17.4 VEEVA SYSTEMS
17.4.1 COMPANY SNAPSHOT
17.4.2 REVENUE ANALYSIS
17.4.3 COMPANY SHARE ANALYSIS
17.4.4 PRODUCT PORTFOLIO
17.4.5 RECENT DEVELOPMENTS
17.5 ARISMIDDLE EAST & AFRICA
17.5.1 COMPANY SNAPSHOT
17.5.2 COMPANY SHARE ANALYSIS
17.5.3 PRODUCT PORTFOLIO
17.5.4 RECENT DEVELOPMENTS
17.6 4G CLINICAL
17.6.1 COMPANY SNAPSHOT
17.6.2 PRODUCT PORTFOLIO
17.6.3 RECENT DEVELOPMENT
17.7 ADVARRA.
17.7.1 COMPANY SNAPSHOT
17.7.2 PRODUCT PORTFOLIO
17.7.3 RECENT DEVELOPMENT
17.8 ANJU
17.8.1 COMPANY SNAPSHOT
17.8.2 PRODUCT PORTFOLIO
17.8.3 RECENT DEVELOPMENTS
17.9 CASTOR.
17.9.1 COMPANY SNAPSHOT
17.9.2 PRODUCT PORTFOLIO
17.9.3 RECENT DEVELOPMENTS
17.1 DATATRAK INT.
17.10.1 COMPANY SNAPSHOT
17.10.2 PRODUCT PORTFOLIO
17.10.3 RECENT DEVELOPMENT
17.11 ECLINICAL SOLUTIONS LLC.
17.11.1 COMPANY SNAPSHOT
17.11.2 PRODUCT PORTFOLIO
17.11.3 RECENT DEVELOPMENT
17.12 IQVIA INC
17.12.1 COMPANY SNAPSHOT
17.12.2 REVENUE ANALYSIS
17.12.3 PRODUCT PORTFOLIO
17.12.4 RECENT DEVELOPMENTS
17.13 MAXISIT
17.13.1 COMPANY SNAPSHOT
17.13.2 PRODUCT PORTFOLIO
17.13.3 RECENT DEVELOPMENT
17.14 MEDNET
17.14.1 COMPANY SNAPSHOT
17.14.2 PRODUCT PORTFOLIO
17.14.3 RECENT DEVELOPMENT
17.15 MEDRIO, INC.
17.15.1 COMPANY SNAPSHOT
17.15.2 PRODUCT PORTFOLIO
17.15.3 RECENT DEVELOPMENTS
17.16 MERATIVE
17.16.1 COMPANY SNAPSHOT
17.16.2 PRODUCT PORTFOLIO
17.16.3 RECENT DEVELOPMENT
17.17 OPENCLINICA, LLC
17.17.1 COMPANY SNAPSHOT
17.17.2 PRODUCT PORTFOLIO
17.17.3 RECENT DEVELOPMENT
17.18 PAREXEL INTERNATIONAL CORPORATION
17.18.1 COMPANY SNAPSHOT
17.18.2 PRODUCT PORTFOLIO
17.18.3 RECENT DEVELOPMENT
17.19 QURETEC
17.19.1 COMPANY SNAPSHOT
17.19.2 PRODUCT PORTFOLIO
17.19.3 RECENT DEVELOPMENT
17.2 REALTIME SOFTWARE SOLUTIONS, LLC
17.20.1 COMPANY SNAPSHOT
17.20.2 PRODUCT PORTFOLIO
17.20.3 RECENT DEVELOPMENT
17.21 RESEARCH MANAGER
17.21.1 COMPANY SNAPSHOT
17.21.2 PRODUCT PORTFOLIO
17.21.3 RECENT DEVELOPMENT
17.22 SAAMA TECHNOLOGIES, LLC
17.22.1 COMPANY SNAPSHOT
17.22.2 PRODUCT PORTFOLIO
17.22.3 RECENT DEVELOPMENT
17.23 SIGNANT HEALTH
17.23.1 COMPANY SNAPSHOT
17.23.2 PRODUCT PORTFOLIO
17.23.3 RECENT DEVELOPMENT
17.24 Y-PRIME, LLC.
17.24.1 COMPANY SNAPSHOT
17.24.2 PRODUCT PORTFOLIO
17.24.3 RECENT DEVELOPMENT
18 QUESTIONNAIRE
19 RELATED REPORTS
Lista de Tabela
TABLE 1 THE COSTS OF IMPLEMENTATION ARE AS FOLLOWS, ACCORDING TO SIMPLETRIALS:
TABLE 2 ACCORDING TO ECLINICAL WORKS – ECLINICAL OFFERS TWO ENTERPRISE PRICING PACKAGES:
TABLE 3 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY PRODUCT, 2021-2030 (USD MILLION)
TABLE 4 MIDDLE EAST & AFRICA ELECTRONIC DATA CAPTURE AND CLINICAL DATA MANAGEMENT SYSTEMS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 5 MIDDLE EAST & AFRICA RANDOMIZATION AND TRIAL SUPPLY MANAGEMENT IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 6 MIDDLE EAST & AFRICA CLINICAL TRIAL MANAGEMENT SYSTEMS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 7 MIDDLE EAST & AFRICA ELECTRONIC TRIAL MASTER FILE SYSTEMS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 8 MIDDLE EAST & AFRICA ELECTRONIC CLINICAL OUTCOME ASSESSMENT SOLUTIONS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 9 MIDDLE EAST & AFRICA SAFETY SOLUTIONS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 10 MIDDLE EAST & AFRICA REGULATORY INFORMATION MANAGEMENT SOLUTIONS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 11 MIDDLE EAST & AFRICA CLINICAL DATA INTEGRATION PLATFORMS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 12 MIDDLE EAST & AFRICA CLINICAL ANALYTICS PLATFORMS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 13 MIDDLE EAST & AFRICA CARE COORDINATION MEDICAL RECORD (CCMR) IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 14 MIDDLE EAST & AFRICA OTHERS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 15 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY DELIVERY MODE, 2021-2030 (USD MILLION)
TABLE 16 MIDDLE EAST & AFRICA WEB-HOSTED (ON-DEMAND) SOLUTIONS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 17 MIDDLE EAST & AFRICA CLOUD-BASED (SAAS) SOLUTIONS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 18 MIDDLE EAST & AFRICA LICENSED ENTERPRISE (ON-PREMISES) SOLUTIONS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 19 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY CLINICAL TRIAL PHASE, 2021-2030 (USD MILLION)
TABLE 20 MIDDLE EAST & AFRICA PHASE III IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 21 MIDDLE EAST & AFRICA PHASE I IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 22 MIDDLE EAST & AFRICA PHASE II IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 23 MIDDLE EAST & AFRICA PHASE IV IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 24 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY ORGANIZATION SIZE, 2021-2030 (USD MILLION)
TABLE 25 MIDDLE EAST & AFRICA MEDIUM AND SMALL IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 26 MIDDLE EAST & AFRICA LARGE IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 27 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY USER DEVICE, 2021-2030 (USD MILLION)
TABLE 28 MIDDLE EAST & AFRICA DESKTOP IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 29 MIDDLE EAST & AFRICA TABLET IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 30 MIDDLE EAST & AFRICA SMART PHONE IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 31 MIDDLE EAST & AFRICA HANDHELD PDA DEVICE IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 32 MIDDLE EAST & AFRICA OTHERS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 33 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 34 MIDDLE EAST & AFRICA CONTRACT RESEARCH ORGANIZATIONS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 35 MIDDLE EAST & AFRICA PHARMACEUTICAL AND BIOPHARMACEUTICAL COMPANIES IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 36 MIDDLE EAST & AFRICA MEDICAL DEVICE MANUFACTURERS IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 37 MIDDLE EAST & AFRICA HOSPITAL IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 38 MIDDLE EAST & AFRICA CONSULTING SERVICE COMPANIES IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 39 MIDDLE EAST & AFRICA ACADEMIC RESEARCH INSTITUTES IN E-CLINICAL SOLUTIONS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 40 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET, BY COUNTRY, 2021-2030 (USD MILLION)
TABLE 41 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET, BY PRODUCT, 2021-2030 (USD MILLION)
TABLE 42 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET, BY DELIVERY MODE, 2021-2030 (USD MILLION)
TABLE 43 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET, BY CLINICAL TRIAL PHASE, 2021-2030 (USD MILLION)
TABLE 44 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET, BY ORGANIZATION SIZE, 2021-2030 (USD MILLION)
TABLE 45 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET, BY USER DEVICE, 2021-2030 (USD MILLION)
TABLE 46 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 47 SOUTH AFRICA E-CLINICAL SOLUTIONS MARKET, BY PRODUCT, 2021-2030 (USD MILLION)
TABLE 48 SOUTH AFRICA E-CLINICAL SOLUTIONS MARKET, BY DELIVERY MODE, 2021-2030 (USD MILLION)
TABLE 49 SOUTH AFRICA E-CLINICAL SOLUTIONS MARKET, BY CLINICAL TRIAL PHASE, 2021-2030 (USD MILLION)
TABLE 50 SOUTH AFRICA E-CLINICAL SOLUTIONS MARKET, BY ORGANIZATION SIZE, 2021-2030 (USD MILLION)
TABLE 51 SOUTH AFRICA E-CLINICAL SOLUTIONS MARKET, BY USER DEVICE, 2021-2030 (USD MILLION)
TABLE 52 SOUTH AFRICA E-CLINICAL SOLUTIONS MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 53 SAUDI ARABIA E-CLINICAL SOLUTIONS MARKET, BY PRODUCT, 2021-2030 (USD MILLION)
TABLE 54 SAUDI ARABIA E-CLINICAL SOLUTIONS MARKET, BY DELIVERY MODE, 2021-2030 (USD MILLION)
TABLE 55 SAUDI ARABIA E-CLINICAL SOLUTIONS MARKET, BY CLINICAL TRIAL PHASE, 2021-2030 (USD MILLION)
TABLE 56 SAUDI ARABIA E-CLINICAL SOLUTIONS MARKET, BY ORGANIZATION SIZE, 2021-2030 (USD MILLION)
TABLE 57 SAUDI ARABIA E-CLINICAL SOLUTIONS MARKET, BY USER DEVICE, 2021-2030 (USD MILLION)
TABLE 58 SAUDI ARABIA E-CLINICAL SOLUTIONS MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 59 U.A.E. E-CLINICAL SOLUTIONS MARKET, BY PRODUCT, 2021-2030 (USD MILLION)
TABLE 60 U.A.E. E-CLINICAL SOLUTIONS MARKET, BY DELIVERY MODE, 2021-2030 (USD MILLION)
TABLE 61 U.A.E. E-CLINICAL SOLUTIONS MARKET, BY CLINICAL TRIAL PHASE, 2021-2030 (USD MILLION)
TABLE 62 U.A.E. E-CLINICAL SOLUTIONS MARKET, BY ORGANIZATION SIZE, 2021-2030 (USD MILLION)
TABLE 63 U.A.E. E-CLINICAL SOLUTIONS MARKET, BY USER DEVICE, 2021-2030 (USD MILLION)
TABLE 64 U.A.E. E-CLINICAL SOLUTIONS MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 65 EGYPT E-CLINICAL SOLUTIONS MARKET, BY PRODUCT, 2021-2030 (USD MILLION)
TABLE 66 EGYPT E-CLINICAL SOLUTIONS MARKET, BY DELIVERY MODE, 2021-2030 (USD MILLION)
TABLE 67 EGYPT E-CLINICAL SOLUTIONS MARKET, BY CLINICAL TRIAL PHASE, 2021-2030 (USD MILLION)
TABLE 68 EGYPT E-CLINICAL SOLUTIONS MARKET, BY ORGANIZATION SIZE, 2021-2030 (USD MILLION)
TABLE 69 EGYPT E-CLINICAL SOLUTIONS MARKET, BY USER DEVICE, 2021-2030 (USD MILLION)
TABLE 70 EGYPT E-CLINICAL SOLUTIONS MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 71 ISRAEL E-CLINICAL SOLUTIONS MARKET, BY PRODUCT, 2021-2030 (USD MILLION)
TABLE 72 ISRAEL E-CLINICAL SOLUTIONS MARKET, BY DELIVERY MODE, 2021-2030 (USD MILLION)
TABLE 73 ISRAEL E-CLINICAL SOLUTIONS MARKET, BY CLINICAL TRIAL PHASE, 2021-2030 (USD MILLION)
TABLE 74 ISRAEL E-CLINICAL SOLUTIONS MARKET, BY ORGANIZATION SIZE, 2021-2030 (USD MILLION)
TABLE 75 ISRAEL E-CLINICAL SOLUTIONS MARKET, BY USER DEVICE, 2021-2030 (USD MILLION)
TABLE 76 ISRAEL E-CLINICAL SOLUTIONS MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 77 REST OF MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET, BY PRODUCT, 2021-2030 (USD MILLION)
Lista de Figura
FIGURE 1 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: SEGMENTATION
FIGURE 2 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: DATA TRIANGULATION
FIGURE 3 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: DROC ANALYSIS
FIGURE 4 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: MIDDLE EAST & AFRICA VS REGIONAL MARKET ANALYSIS
FIGURE 5 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: MARKET END USER COVERAGE GRID
FIGURE 8 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: DBMR MARKET POSITION GRID
FIGURE 9 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: VENDOR SHARE ANALYSIS
FIGURE 10 MIDDLE EAST & AFRICA E- CLINICAL SOLUTIONS MARKET: SEGMENTATION
FIGURE 11 THE GROWING USAGE OF E-CLINICAL SOLUTIONS IN CLINICAL TRIALS AS WELL AS INCREASING R&D ACTIVITIES AND CORRESPONDING EXPENDITURE, IS EXPECTED TO DRIVE THE GROWTH OF THE MIDDLE EAST & AFRICA E- CLINICAL SOLUTIONS MARKET FROM 2023 TO 2030
FIGURE 12 PRODUCT SEGMENT IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET IN 2023 AND 2030
FIGURE 13 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF THE MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET
FIGURE 14 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY PRODUCT, 2022
FIGURE 15 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY PRODUCT, 2023-2030 (USD MILLION)
FIGURE 16 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY PRODUCT, CAGR (2023-2030)
FIGURE 17 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY PRODUCT, LIFELINE CURVE
FIGURE 18 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY DELIVERY MODE, 2022
FIGURE 19 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY DELIVERY MODE, 2023-2030 (USD MILLION)
FIGURE 20 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY DELIVERY MODE, CAGR (2023-2030)
FIGURE 21 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY DELIVERY MODE, LIFELINE CURVE
FIGURE 22 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY CLINICAL TRIAL PHASE, 2022
FIGURE 23 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY CLINICAL TRIAL PHASE, 2023-2030 (USD MILLION)
FIGURE 24 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY CLINICAL TRIAL PHASE, CAGR (2023-2030)
FIGURE 25 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY CLINICAL TRIAL PHASE, LIFELINE CURVE
FIGURE 26 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY ORGANIZATION SIZE, 2022
FIGURE 27 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY ORGANIZATION SIZE, 2023-2030 (USD MILLION)
FIGURE 28 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY ORGANIZATION SIZE, CAGR (2023-2030)
FIGURE 29 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY ORGANIZATION SIZE, LIFELINE CURVE
FIGURE 30 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY USER DEVICE, 2022
FIGURE 31 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY USER DEVICE, 2023-2030 (USD MILLION)
FIGURE 32 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY USER DEVICE, CAGR (2023-2030)
FIGURE 33 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY USER DEVICE, LIFELINE CURVE
FIGURE 34 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY END USER, 2022
FIGURE 35 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY END USER, 2023-2030 (USD MILLION)
FIGURE 36 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY END USER, CAGR (2023-2030)
FIGURE 37 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: BY END USER, LIFELINE CURVE
FIGURE 38 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET: SNAPSHOT (2022)
FIGURE 39 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET: BY COUNTRY (2022)
FIGURE 40 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET: BY COUNTRY (2023 & 2030)
FIGURE 41 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET: BY COUNTRY (2022 & 2030)
FIGURE 42 MIDDLE EAST AND AFRICA E-CLINICAL SOLUTIONS MARKET: PRODUCT (2023-2030)
FIGURE 43 MIDDLE EAST & AFRICA E-CLINICAL SOLUTIONS MARKET: COMPANY SHARE 2022 (%)

Metodologia de Investigação
A recolha de dados e a análise do ano base são feitas através de módulos de recolha de dados com amostras grandes. A etapa inclui a obtenção de informações de mercado ou dados relacionados através de diversas fontes e estratégias. Inclui examinar e planear antecipadamente todos os dados adquiridos no passado. Da mesma forma, envolve o exame de inconsistências de informação observadas em diferentes fontes de informação. Os dados de mercado são analisados e estimados utilizando modelos estatísticos e coerentes de mercado. Além disso, a análise da quota de mercado e a análise das principais tendências são os principais fatores de sucesso no relatório de mercado. Para saber mais, solicite uma chamada de analista ou abra a sua consulta.
A principal metodologia de investigação utilizada pela equipa de investigação do DBMR é a triangulação de dados que envolve a mineração de dados, a análise do impacto das variáveis de dados no mercado e a validação primária (especialista do setor). Os modelos de dados incluem grelha de posicionamento de fornecedores, análise da linha de tempo do mercado, visão geral e guia de mercado, grelha de posicionamento da empresa, análise de patentes, análise de preços, análise da quota de mercado da empresa, normas de medição, análise global versus regional e de participação dos fornecedores. Para saber mais sobre a metodologia de investigação, faça uma consulta para falar com os nossos especialistas do setor.
Personalização disponível
A Data Bridge Market Research é líder em investigação formativa avançada. Orgulhamo-nos de servir os nossos clientes novos e existentes com dados e análises que correspondem e atendem aos seus objetivos. O relatório pode ser personalizado para incluir análise de tendências de preços de marcas-alvo, compreensão do mercado para países adicionais (solicite a lista de países), dados de resultados de ensaios clínicos, revisão de literatura, mercado remodelado e análise de base de produtos . A análise de mercado dos concorrentes-alvo pode ser analisada desde análises baseadas em tecnologia até estratégias de carteira de mercado. Podemos adicionar quantos concorrentes necessitar de dados no formato e estilo de dados que procura. A nossa equipa de analistas também pode fornecer dados em tabelas dinâmicas de ficheiros Excel em bruto (livro de factos) ou pode ajudá-lo a criar apresentações a partir dos conjuntos de dados disponíveis no relatório.