Middle East and Africa Stem Cell Therapy Market, By Product Type (Bone Marrow Derived Mesenchymal Cells, Placental or Umbilical Stem Cell, Adipose Tissue Derived Mesenchymal Stem Cells, and Others), Type (Allogenic Stem Cell Therapy and Autologous Stem Cell Therapy), Application (Musculoskeletal Disorders, Acute Graft-Versus-Host Disease (AGVHD), Wounds and Injuries, Cardiovascular Diseases, Surgeries, Gastrointestinal Diseases, and Others), End User (Hospitals and Surgical Centers, Therapeutic Companies, Services Companies, and Others), Distribution Channel (Direct Tender, Third Party Distributors) Industry Trends and Forecast to 2029
Market Analysis and Insights
Chronic diseases-including cancer, musculoskeletal disorders, neurology disorders, chronic injuries, cardiovascular and gastrointestinal -can lead to hospitalization, long-term disability, reduced quality of life, and death.
The mesenchymal stem cells penetrate and integrate into multiple organs, repair cardiovascular, lung, and spinal cord injuries, and improve the state of autoimmune diseases, liver, and bone and cartilage diseases. Stems cells are a potent tool for the treatment of infections caused by inflammation, immune system failure, and, or tissue degeneration
The drivers responsible for the growth of the Middle East and Africa stem cell therapy market are the increased incidence of chronic diseases, rise in GMP-certification approvals for cell therapy production facilities, growing biotechnology sector and rise in clinical trials for stem-cell-based therapies. However, factors that are expected to restrain the market growth are the rise in the cost of stem cell-based research, and the risks faced while undergoing stem cell therapy, and the availability of alternatives.
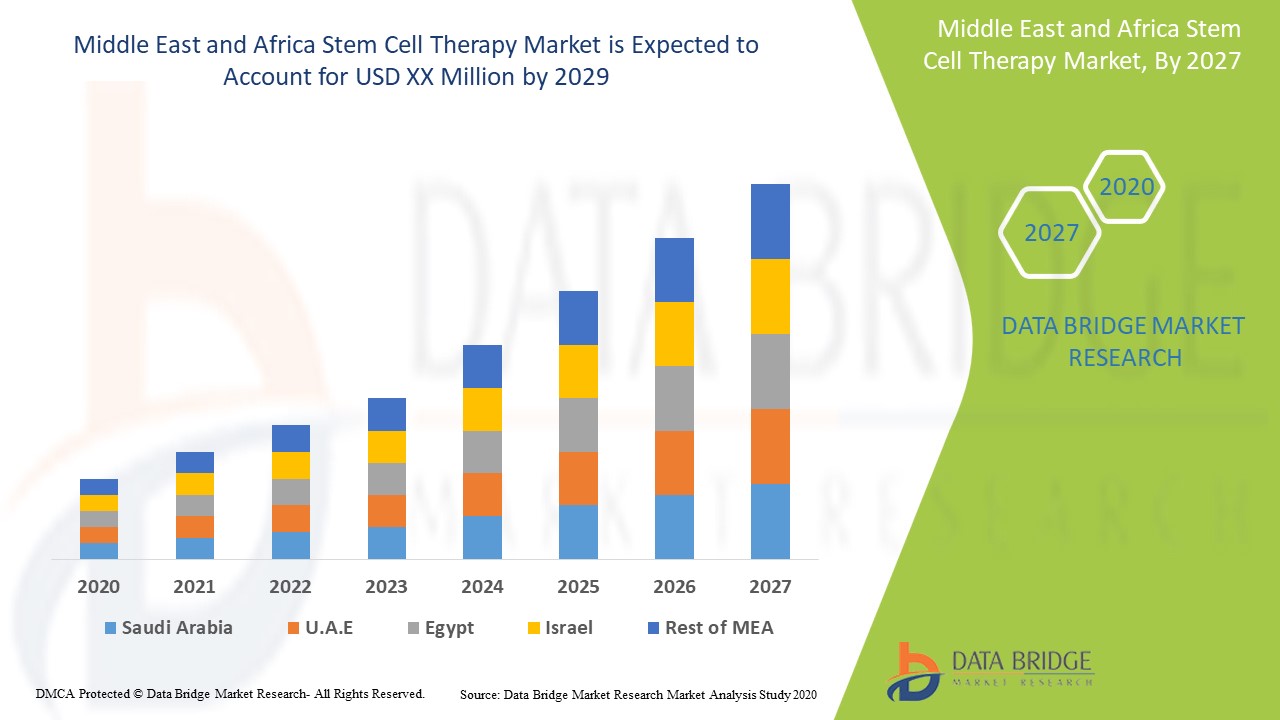
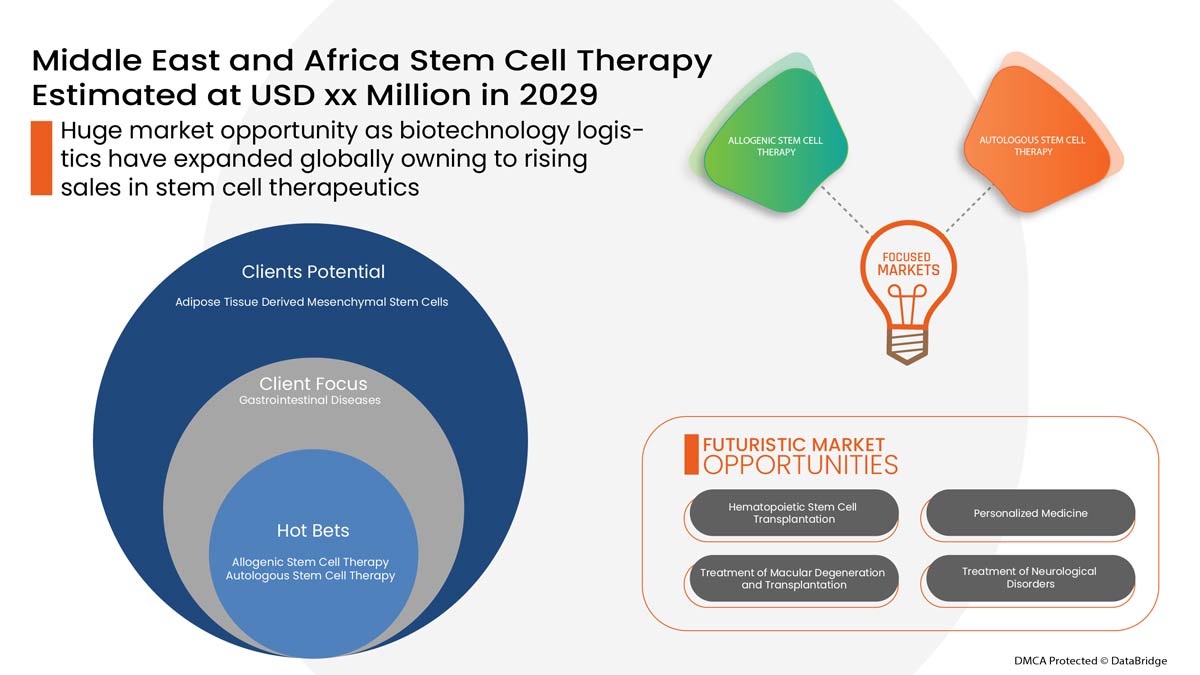
The Middle East and Africa stem cell therapy is supportive and aims to reduce the severity of the symptoms. Data Bridge Market Research analyses that the Middle East and Africa stem cell therapy market is expected to reach the value of USD 1.56 million and grow at a CAGR of 4.1% during the forecast period of 2022 to 2029.
|
Report Metric |
Details |
|
Forecast Period |
2022 to 2029 |
|
Base Year |
2021 |
|
Historic Year |
2020 (Customizable to 2019 - 2014) |
|
Quantitative Units |
Revenue in USD Million |
|
Segments Covered |
By Product Type (Bone Marrow Derived Mesenchymal Cells, Placental or Umbilical Stem Cell, Adipose Tissue Derived Mesenchymal Stem Cells, and Others), Type (Allogenic Stem Cell Therapy and Autologous Stem Cell Therapy), Application (Musculoskeletal Disorders, Acute Graft-Versus-Host Disease (AGVHD), Wounds and Injuries, Cardiovascular Diseases, Surgeries, Gastrointestinal Diseases, and Others), End User (Hospitals and Surgical Centers, Therapeutic Companies, Services Companies, and Others), Distribution Channel (Direct Tender, Third Party Distributors) |
|
적용 국가 |
이스라엘 |
|
시장 참여자 포함 |
US Stem Cell, Inc., STEMPEUTICS RESEARCH PVT LTD, Pluristem Inc. 등 |
시장 정의
줄기세포는 신체의 초기 물질로, 특수 기능을 가진 다른 모든 세포가 생성됩니다. 신체 또는 실험실에서 적절한 조건 하에 줄기세포는 분열하여 더 많은 딸세포라고 불리는 세포를 형성합니다. 딸세포는 혈액 세포, 뇌 세포, 심장 근육 세포 또는 뼈 세포와 같이 더 구체적인 기능을 가진 새로운 줄기세포 또는 특수 세포(분화)가 됩니다. 줄기세포에 대한 많은 관심은 연구 과학자들 사이에서 관심을 불러일으켰습니다. 줄기세포를 사용하여 질병이 어떻게 발병하고 발생하는지 이해하고, 세포를 대체할 건강한 세포를 생성하고, 신약의 안전성과 효능을 테스트하는 것이 줄기세포 치료제가 사용되는 과학적 이유입니다.
줄기세포 치료는 줄기세포 또는 파생물을 사용하여 기능 장애 또는 손상된 조직 복구 반응을 촉진합니다. 장기 이식의 다음 장이며 공급이 제한적인 기증 장기 대신 세포를 사용합니다. 지방 조직 유래 중간엽 줄기세포, 골수 유래 중간엽 줄기세포, 태반 또는 탯줄 줄기세포와 같은 성인 줄기세포는 대부분 조직에서 적은 수로 발견됩니다. 배아 줄기세포는 3~5일 된 배아에서 유래합니다. 새로운 징후는 성인 줄기세포가 다양한 유형의 세포를 생성할 수 있음을 나타냅니다.
중동 및 아프리카 줄기세포 치료 시장 동향
운전자
- 만성질환의 유병률 및 발생률 증가
만성 질환은 전 세계적으로 흔한 건강 문제입니다. 중동과 아프리카에서는 성인 3명 중 1명이 만성 질환을 앓고 있습니다. 만성 질환은 많은 시민의 건강과 삶의 질에 영향을 미쳤습니다. 암, 근골격계 질환, 신경계 질환, 만성 손상, 심혈관 및 위장관 질환을 포함한 만성 질환은 입원, 장기 장애, 삶의 질 저하 및 사망으로 이어질 수 있습니다.
중간엽 줄기 세포는 여러 장기에 침투하여 통합되고, 심혈관, 폐 및 척수 손상을 치료하고, 자가면역 질환, 간, 뼈 및 연골 질환의 상태를 개선합니다. 줄기 세포는 염증, 면역 체계 부전 및 조직 변성으로 인한 질병을 치료하는 강력한 도구입니다.
예를 들어,
- 2021년 세계보건기구(WHO) 데이터에 따르면 전 세계적으로 근골격계 질환을 앓고 있는 사람은 약 17억 명입니다. 허리 통증은 근골격계 질환의 높은 부담을 초래합니다.
- 연구개발 투자 증가
줄기세포 연구는 국립보건원(NIH) 예산으로 지원됩니다. 민간 부문도 줄기세포 연구에 자금을 지원하지만, 이러한 투자는 일반적으로 나중에, 테스트 및 개발 단계에서, 그 다음 초기 기초 연구에서 이루어집니다. 줄기세포 치료법이 새로운 분야이기 때문에 편견 없는 정부 기관이 이를 감독해야 합니다. FDA는 신중하고 철저하지만, 그들은 끊임없이 자금 조달에 어려움을 겪고 있으며, 지불을 잠재적 미래 수혜자와 일치시키는 장기 투자를 하고 있습니다.
- 성장하는 생명공학 분야
사람들은 건강에 대해 더 많이 알게 되고 있으며 예방적 건강 관리에 대한 경각심도 높아지고 있습니다. 건강 관리에 대한 강조가 인기를 얻고 있습니다. 질병과 증상에 대한 인식은 의료 시스템에서 사용 가능한 첨단 기술(예: 의료 제공자가 생존율을 높이는 데 도움이 되는 줄기 세포 기술)로 인해 감염을 선별하고 조기에 감지하는 데 필수적입니다. 줄기 세포 기반 치료법 개발에 중점을 둔 많은 생명 공학 회사가 시장 성장을 주도할 것으로 예상됩니다.
예를 들어,
- 2018년, Medicare 및 Medicaid 서비스 센터에 따르면 NHE는 4.6% 증가하여 3조 6,000억 달러(1인당 11,172.00달러)에 달했으며, GDP의 17.7%를 차지했습니다. 국가 의료 지출은 2028년까지 6조 2,000억 달러에 도달할 것이며, 2019-2028년의 연평균 성장률은 5.4%입니다.
기회
- 의료비 지출 증가
게다가 정부와 민간 기관의 연구 개발 활동이 늘어나고 투자도 늘어나면서 시장 성장률에 새로운 기회가 생길 것입니다.
예를 들어,
• 2019년 Knoema에 따르면 이스라엘의 1인당 의료비는 3,456달러였습니다. 이스라엘의 1인당 의료비는 2000년 1,514달러에서 2019년 3,456달러로 증가했으며 연평균 4.66%의 성장률을 기록했습니다.
- 시장 참여자들의 전략적 이니셔티브
만성 질환의 시기적절한 치료로 인해 줄기세포 치료에 대한 수요가 증가했습니다. 이러한 유리한 요인은 약물에 대한 필요성을 높이고 시장 수요를 달성하기 위해 소규모 및 대규모 시장 참여자는 다양한 전략을 활용하고 있습니다.
주요 기업들은 또한 사업을 원활하게 운영하고, 위험을 피하고, 시장 판매의 장기적 성장을 높이기 위해 제품 출시, 인수, 승인, 확장, 파트너십 등의 구체적인 전략을 고안하려고 노력하고 있습니다.
예를 들어,
- 2022년 3월 Pluristem Inc.는 PLX-R18에 대한 긍정적인 1상 임상 시험을 발표했습니다. 미국 식품의약국(FDA)은 HCT 후 이식 실패 및 불완전한 조혈 회복을 치료하고 급성 방사선 증후군(ARS)을 예방 및 치료하기 위한 희귀 의약품 지정을 부여했습니다. 완료된 1상 임상 시험은 시장 참여자가 2상 및 3상 임상 시험과 시판 후 승인을 수행할 자격을 갖추게 됩니다.
인수, 컨퍼런스, 집중된 세그먼트 제품 출시를 포함한 시장 참여자들의 이러한 전략적 이니셔티브는 회사가 성장하고 회사의 제품 포트폴리오를 개선하는 데 도움이 되며 궁극적으로 더 많은 수익 창출로 이어집니다. 따라서 시장 참여자들의 이러한 전략적 이니셔티브는 시장 성장을 촉진하는 데 도움이 되는 기회를 제공합니다.
제약/도전
- 줄기세포 기반 치료 연구 비용 증가
줄기세포 치료는 여러 질환을 치료하기 위한 개발되고 새로운 치료 옵션입니다. 때때로 치료 비용은 여러 질환에 대한 우려 사항입니다. 줄기세포 치료 치료 절차. 줄기세포 분야는 여전히 매우 전문화되어 있으며 주류 및 보험 회사에서 채택하지 않았습니다. 줄기세포 치료 기반 연구 치료 비용은 의료 보험에서 보장되지 않습니다. 이러한 비용은 환자에게 부담이 됩니다. 따라서 현재의 높은 비용은 감소 추세를 보일 것으로 예상됩니다.
예를 들어,
- Bioinformant의 데이터에 따르면 2022년 미국에서 자가줄기세포 치료 비용은 약 15만 달러입니다.
- 숙련된 전문가 부족
숙련된 전문성의 부족이나 부족은 한 곳에서 회복과 성장의 속도에 도전할 것입니다. 종종 한 곳의 실업자는 다른 곳에서는 부족한 기술을 가지고 있습니다. 게다가 이 분야의 급속한 기술 발전은 전문성 부족으로 이어집니다.
줄기세포 치료는 손상된 세포를 건강한 새로운 세포로 대체하는 현대 기술을 포괄합니다. 오늘날 줄기세포 치료는 신경 질환, 당뇨병, 뇌 손상 및 척수 손상과 같은 광범위한 질병으로 고통받는 환자에게 상당한 이점을 제공합니다.
줄기세포 치료를 다루는 혈액학자는 적절한 줄기세포 치료 투여 훈련을 받았어야 합니다. 그들은 특정 응급 치료를 위한 현장의 잘 조직된 시스템에 익숙해야 합니다. 줄기세포 치료 코디네이터는 환자가 치료를 받을 때 모든 관련 인력에게 알려야 함을 보장합니다. 코디네이터는 주 등록 간호사를 포함한 의료 전문가가 될 수 있습니다.
중동 및 아프리카 줄기세포 치료 시장 보고서는 최근의 새로운 개발, 무역 규정, 수출입 분석, 생산 분석, 가치 사슬 최적화, 시장 점유율, 국내 및 지역 시장 참여자의 영향, 새로운 수익 주머니, 시장 규정의 변화, 전략적 시장 성장 분석, 시장 규모, 범주 시장 성장, 응용 분야 틈새 시장 및 지배력, 제품 승인, 제품 출시, 지리적 확장, 시장의 기술 혁신에 대한 분석 기회를 제공합니다. 중동 및 아프리카 줄기세포 치료 시장에 대한 자세한 정보를 얻으려면 Data Bridge Market Research에 연락하여 분석가 브리핑을 받으세요. 저희 팀은 시장 성장을 달성하기 위한 정보에 입각한 시장 결정을 내리는 데 도움을 드립니다.
환자 역학 분석
2017년 중동아프리카 질병부담(GBD) 자료에 따르면 허리통증이 가장 많은 발병률을 보였으며, 그 다음으로 근골격계 질환(21.5%) 순이었다.
중동 및 아프리카 줄기세포 치료 시장은 또한 환자 분석, 예후 및 치료법에 대한 자세한 시장 분석을 제공합니다. 유병률, 발생률, 사망률 및 준수율은 보고서에서 사용할 수 있는 일부 데이터 변수입니다. 시장 성장에 대한 역학의 직접 또는 간접 영향 분석을 분석하여 성장 기간 동안 시장을 예측하기 위한 보다 견고하고 코호트 다변량 통계 모델을 만듭니다.
COVID-19가 중동 및 아프리카 줄기세포 치료 시장 에 미치는 영향
팬데믹 동안 줄기세포 치료는 COVID-19 환자의 사망률과 이환율을 줄이는 데 놀라운 효과가 있었습니다. 이러한 결과를 승인하려면 더 많은 대규모 연구가 필요합니다. 최상의 임상 결과를 달성하기 위해 COVID-19 감염에서 줄기세포 치료에 대한 프로토콜을 정의해야 합니다. COVID-19 동안 임상 시험이 수행되었습니다.
최근 개발
- 2022년 3월 Pluristem Inc.는 PLX-R18에 대한 긍정적인 1상 임상 시험을 발표했습니다. 미국 식품의약국(FDA)은 HCT 후 이식 실패 및 불완전한 조혈 회복을 치료하고 급성 방사선 증후군(ARS)을 예방 및 치료하기 위한 희귀 의약품 지정을 부여했습니다. 완료된 1상 임상 시험은 시장 참여자가 2상 및 3상 임상 시험을 실시하고 시판 후 승인을 받을 자격을 갖추게 됩니다.
중동 및 아프리카 줄기세포 치료 시장 범위
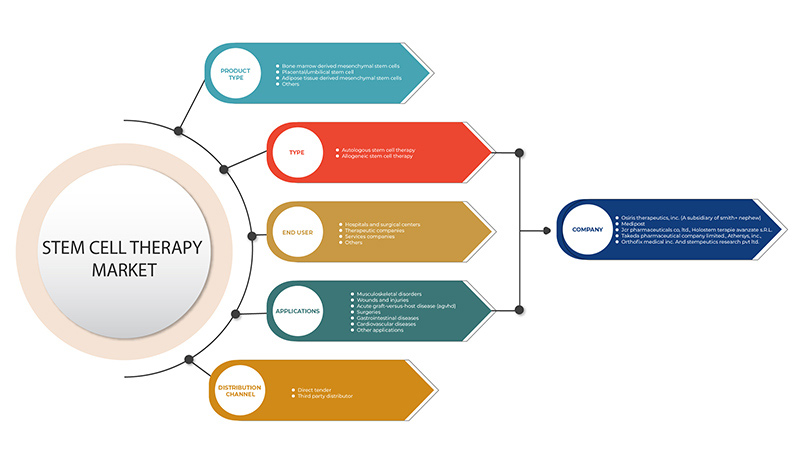
중동 및 아프리카 줄기세포 치료 시장은 제품 유형, 유형, 응용 분야, 최종 사용자 및 유통 채널을 기준으로 5개 세그먼트로 구분됩니다. 이러한 세그먼트 간의 성장은 산업의 빈약한 성장 세그먼트를 분석하고 사용자에게 핵심 시장 응용 분야를 식별하기 위한 전략적 결정을 내리는 데 도움이 되는 귀중한 시장 개요와 시장 통찰력을 제공하는 데 도움이 됩니다.
제품 유형
- 골수유래 중간엽줄기세포
- 태반 또는 탯줄 줄기 세포
- 지방조직 유래 중간엽 줄기세포
- 기타
중동 및 아프리카 줄기세포 치료 시장은 제품 유형을 기준으로 골수 유래 중간엽 줄기세포, 태반 또는 태아줄기세포, 지방 조직 유래 중간엽 줄기세포 및 기타로 구분됩니다.
유형
- 동종 줄기 세포 치료
- 자가줄기세포 치료
중동 및 아프리카 줄기세포 치료 시장은 유형을 기준으로 동종 줄기세포 치료와 자가 줄기세포 치료로 구분됩니다.
애플리케이션
- 근골격계 질환
- 상처와 부상
- 급성 이식편대숙주병(AGVHD)
- 수술
- 위장관 질환
- 심혈관 질환
- 기타
중동 및 아프리카 줄기세포 치료 시장은 응용 분야별로 근골격계 질환, 상처 및 부상, 급성 이식편대숙주병(AGVHD), 수술, 위장관 질환, 심혈관 질환 등으로 구분됩니다.
최종 사용자
- 병원 및 수술 센터
- 치료 회사
- 서비스 회사
- 기타
중동 및 아프리카 줄기세포 치료 시장은 최종 사용자를 기준으로 병원 및 수술 센터, 치료 회사, 서비스 회사 및 기타로 구분됩니다.
유통 채널
- 직접 입찰
- 제3자 유통업체
중동 및 아프리카 줄기세포 치료 시장은 유통 채널을 기준으로 직접 입찰과 제3자 유통업체로 구분됩니다.
중동 및 아프리카 줄기세포 치료 시장 지역 분석/통찰력
중동 및 아프리카 줄기세포 치료 시장을 분석하고, 위에 언급된 대로 지역, 제품 유형, 유형, 응용 분야, 최종 사용자 및 유통 채널별로 시장 규모에 대한 통찰력과 추세를 제공합니다.
중동 및 아프리카 줄기세포 치료 시장 보고서에서 다루는 국가 중 하나는 이스라엘입니다. 이스라엘은 만성 질환의 증가와 의료비 지출 증가로 인해 시장을 지배할 것으로 예상됩니다.
보고서의 국가 섹션은 또한 개별 시장 영향 요인과 국내 시장의 현재 및 미래 트렌드에 영향을 미치는 규제 변경 사항을 제공합니다. 신규 판매, 교체 판매, 국가 인구 통계, 질병 역학 및 수출입 관세와 같은 데이터 포인트는 개별 국가의 시장 시나리오를 예측하는 데 사용되는 주요 포인터 중 일부입니다. 또한 중동 및 아프리카 브랜드의 존재 및 가용성과 지역 및 국내 브랜드와의 대규모 또는 희소한 경쟁으로 인해 직면한 과제, 판매 채널의 영향이 국가 데이터에 대한 예측 분석을 제공하는 동안 고려됩니다.
경쟁 환경 및 중동 및 아프리카 줄기세포 치료 시장 점유율 분석
중동 및 아프리카 줄기세포 치료 시장 경쟁 구도는 경쟁업체의 세부 정보를 제공합니다. 포함된 세부 정보는 회사 개요, 회사 재무, 창출된 수익, 시장 잠재력, 연구 개발 투자, 새로운 시장 이니셔티브, 중동 및 아프리카의 입지, 생산 현장 및 시설, 생산 용량, 회사의 강점과 약점, 제품 출시, 제품 폭과 범위, 응용 분야 우세입니다. 위에 제공된 데이터 포인트는 중동 및 아프리카 줄기세포 치료 시장과 관련된 회사의 초점에만 관련이 있습니다.
중동 및 아프리카 줄기세포 치료 시장의 주요 기업으로는 US Stem Cell, Inc., STEMPEUTICS RESEARCH PVT LTD, Pluristem Inc. 등이 있습니다.
연구 방법론
Data collection and base year analysis are done using data collection modules with large sample sizes. The market data is analyzed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or can drop down your inquiry. The key research methodology used by the DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market, and primary (industry expert) validation. Apart from this, data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Company Market Share Analysis, Standards of Measurement, Middle East and Africa versus Regional, and Vendor Share Analysis. To know more about the research methodology, drop an inquiry to speak to our industry experts.
SKU-
세계 최초의 시장 정보 클라우드 보고서에 온라인으로 접속하세요
- 대화형 데이터 분석 대시보드
- 높은 성장 잠재력 기회를 위한 회사 분석 대시보드
- 사용자 정의 및 질의를 위한 리서치 분석가 액세스
- 대화형 대시보드를 통한 경쟁자 분석
- 최신 뉴스, 업데이트 및 추세 분석
- 포괄적인 경쟁자 추적을 위한 벤치마크 분석의 힘 활용
목차
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF THE MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATIONS
1.6 MARKETS COVERED
2 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 DBMR TRIPOD DATA VALIDATION MODEL
2.5 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.6 MULTIVARIATE MODELLING
2.7 PRODUCT SEGMENT LIFELINE CURVE
2.8 DBMR MARKET POSITION GRID
2.9 VENDOR SHARE ANALYSIS
2.1 MARKET END USER COVERAGE GRID
2.11 SECONDARY SOURCES
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PESTEL
4.2 PORTER'S FIVE FORCES MODEL
5 EPIDEMIOLOGY
6 PIPELINE ANALYSIS FOR THE MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET
7 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: REGULATIONS
8 MARKET OVERVIEW
8.1 DRIVERS
8.1.1 THE RISE IN PREVALENCE AND INCIDENCE OF CHRONIC DISEASES
8.1.2 RISE IN INVESTMENT IN RESEARCH AND DEVELOPMENT AND AVAILABILITY OF FUNDING FOR STEM CELL RESEARCH
8.1.3 GROWING BIOTECHNOLOGY SECTOR
8.1.4 RISE IN GMP CERTIFICATION APPROVALS FOR CELL THERAPY PRODUCTION FACILITIES
8.1.5 RISE IN CLINICAL TRIALS FOR STEM-CELL-BASED THERAPIES
8.2 RESTRAINTS
8.2.1 THE RISE IN COST OF STEM-CELL-BASED THERAPY RESEARCH
8.2.2 THE RISKS FACED WHILE UNDERGOING STEM CELL THERAPY
8.2.3 ETHICAL CONCERNS RELATED TO STEM CELL THERAPY RESEARCH
8.2.4 AVAILABILITY OF ALTERNATIVES
8.3 OPPORTUNITIES
8.3.1 STRATEGIC INITIATIVE BY MARKET PLAYERS
8.3.2 RISE IN HEALTHCARE EXPENDITURE
8.3.3 THE EMERGENCE OF INDUCED PLURIPOTENT STEM CELLS (IPSCS)
8.4 CHALLENGES
8.4.1 THE LACK OF SKILLED PROFESSIONALS REQUIRED FOR STEM CELL THERAPY
8.4.2 STRINGENT REGULATIONS
9 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET, BY PRODUCT TYPE
9.1 OVERVIEW
9.2 BONE MARROW DERIVED MESENCHYMAL STEM CELLS
9.3 PLACENTAL/UMBILICAL STEM CELL
9.4 ADIPOSE TISSUE DERIVED MESENCHYMAL STEM CELLS
9.5 OTHERS
10 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET, BY TYPE
10.1 OVERVIEW
10.2 ALLOGENEIC STEM CELL THERAPY
10.2.1 MUSCULOSKELETAL DISORDERS
10.2.2 WOUNDS AND INJURIES
10.2.3 ACUTE GRAFT-VERSUS-HOST DISEASE (AGVHD)
10.2.4 SURGERIES
10.2.5 GASTROINTESTINAL DISEASES
10.2.6 OTHER APPLICATION
10.3 AUTOLOGOUS STEM CELL THERAPY
10.3.1 CARDIOVASCULAR DISEASES
10.3.2 GASTROINTESTINAL DISEASES
10.3.3 OTHER APPLICATION
11 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET, BY APPLICATION
11.1 OVERVIEW
11.2 MUSCULOSKELETAL DISORDERS
11.3 WOUNDS AND INJURIES
11.4 ACUTE GRAFT-VERSUS-HOST DISEASE (AGVHD)
11.5 SURGERIES
11.6 GASTROINTESTINAL DISEASES
11.7 CARDIOVASCULAR DISEASES
11.8 OTHER APPLICATION
12 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET, BY END USER
12.1 OVERVIEW
12.2 HOSPITAL AND SURGICAL CENTERS
12.3 THERAPEUTIC COMPANIES
12.4 SERVICES COMPANIES
12.5 OTHERS
13 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET, BY DISTRIBUTION CHANNEL
13.1 OVERVIEW
13.2 DIRECT TENDER
13.3 THIRD PARTY DISTRIBUTORS
14 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET, BY REGION
14.1 MIDDLE EAST AND AFRICA
14.1.1 ISRAEL
15 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: COMPANY LANDSCAPE
15.1 COMPANY SHARE ANALYSIS: MIDDLE EAST & AFRICA
16 SWOT ANALYSIS
17 COMPANY PROFILE
17.1 OSIRIS THERAPEUTICS, INC. (A SUBSIDIARY OF SMITH+NEPHEW) (2021)
17.1.1 COMPANY SNAPSHOT
17.1.2 REVENUE ANALYSIS
17.1.3 COMPANY SHARE ANALYSIS
17.1.4 PRODUCT PORTFOLIO
17.1.5 RECENT DEVELOPMENT
17.2 JCR PHARMACEUTICALS CO., LTD ( (2021)
17.2.1 COMPANY SNAPSHOT
17.2.2 REVENUE ANALYSIS
17.2.3 COMPANY SHARE ANALYSIS
17.2.4 PRODUCT PORTFOLIO
17.2.5 RECENT DEVELOPMENTS
17.3 ORTHOFIX MEDICAL INC. (2021)
17.3.1 COMPANY SNAPSHOT
17.3.2 REVENUE ANALYSIS
17.3.3 COMPANY SHARE ANALYSIS
17.3.4 PRODUCT PORTFOLIO
17.3.5 RECENT DEVELOPMENTS
17.4 MEDIPOST (2021)
17.4.1 COMPANY SNAPSHOT
17.4.2 REVENUE ANALYSIS
17.4.3 COMPANY SHARE ANALYSIS
17.4.4 PRODUCT PORTFOLIO
17.4.5 RECENT DEVELOPMENTS
17.5 TAKEDA PHARMACEUTICAL COMPANY LIMITED (2021)
17.5.1 COMPANY SNAPSHOT
17.5.2 REVENUE ANALYSIS
17.5.3 COMPANY SHARE ANALYSIS
17.5.4 PRODUCT PORTFOLIO
17.5.5 RECENT DEVELOPMENT
17.6 CORESTEM, INC. (2021)
17.6.1 COMPANY SNAPSHOT
17.6.2 REVENUE ANALYSIS
17.6.3 PRODUCT PORTFOLIO
17.6.4 RECENT DEVELOPMENT
17.7 PHARMICELL CO., LTD. (2021)
17.7.1 COMPANY SNAPSHOT
17.7.2 PRODUCT PORTFOLIO
17.7.3 RECENT DEVELOPMENTS
17.8 ANTEROGEN.CO., LTD (2021)
17.8.1 COMPANY SNAPSHOT
17.8.2 PRODUCT PORTFOLIO
17.8.3 RECENT DEVELOPMENTS
17.9 ATHERSYS, INC.(2021)
17.9.1 COMPANY SNAPSHOT
17.9.2 REVENUE ANALYSIS
17.9.3 PRODUCT PORTFOLIO
17.9.4 RECENT DEVELOPMENTS
17.1 BRAINSTORM CELL LIMITED (2021)
17.10.1 COMPANY SNAPSHOT
17.10.2 PRODUCT PORTFOLIO
17.10.3 RECENT DEVELOPMENTS
17.11 BIORESTORATIVE THERAPIES, INC. (2021)
17.11.1 COMPANY SNAPSHOT
17.11.2 REVENUE ANALYSIS
17.11.3 PRODUCT PORTFOLIO
17.11.4 RECENT DEVELOPMENTS
17.12 HOLOSTEM TERAPIE AVANZATE S.R.L. (2021)
17.12.1 COMPANY SNAPSHOT
17.12.2 PRODUCT PORTFOLIO
17.12.3 RECENT DEVELOPMENTS
17.13 INTERNATIONAL STEMCELL CORPORATION (2021)
17.13.1 COMPANY SNAPSHOT
17.13.2 REVENUE ANALYSIS
17.13.3 PRODUCT PORTFOLIO
17.13.4 RECENT DEVELOPMENT
17.14 MESOBLAST LTD (2021)
17.14.1 COMPANY SNAPSHOT
17.14.2 REVENUE ANALYSIS
17.14.3 PRODUCT PORTFOLIO
17.14.4 RECENT DEVELOPMENTS
17.15 PLURISTEM INC.(2021)
17.15.1 COMPANY SNAPSHOT
17.15.2 REVENUE ANALYSIS
17.15.3 PRODUCT PORTFOLIO
17.15.4 RECENT DEVELOPMENT
17.16 STEMPEUTICS RESEARCH PVT LTD
17.16.1 COMPANY SNAPSHOT
17.16.2 PRODUCT PORTFOLIO
17.16.3 RECENT DEVELOPMENTS
17.17 U.S. STEM CELL, INC. (2021)
17.17.1 COMPANY SNAPSHOT
17.17.2 REVENUE ANALYSIS
17.17.3 PRODUCT PORTFOLIO
17.17.4 RECENT DEVELOPMENT
18 QUESTIONNAIRE
19 RELATED REPORTS
표 목록
TABLE 1 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 2 MIDDLE EAST & AFRICA BONE MARROW DERIVED MESENCHYMAL STEM CELLS IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 3 MIDDLE EAST & AFRICA PLACENTAL/UMBILICAL STEM CELL IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 4 MIDDLE EAST & AFRICA ADIPOSE TISSUE DERIVED MESENCHYMAL STEM CELLS IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 5 MIDDLE EAST & AFRICA OTHERS IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 6 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 7 MIDDLE EAST & AFRICA ALLOGENEIC STEM CELL THERAPY IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 8 MIDDLE EAST & AFRICA ALLOGENEIC STEM CELL THERAPY IN STEM CELL THERAPY MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 9 MIDDLE EAST & AFRICA AUTOLOGOUS STEM CELL THERAPY IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 10 MIDDLE EAST & AFRICA AUTOLOGOUS STEM CELL THERAPY IN STEM CELL THERAPY MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 11 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 12 MIDDLE EAST & AFRICA MUSCULOSKELETAL DISORDERS IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 13 MIDDLE EAST & AFRICA WOUNDS AND INJURIES IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 14 MIDDLE EAST & AFRICA ACUTE GRAFT-VERSUS-HOST DISEASE (AGVHD) IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 15 MIDDLE EAST & AFRICA SURGERIES IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 16 MIDDLE EAST & AFRICA GASTROINTESTINAL DISEASES IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 17 MIDDLE EAST & AFRICA CARDIOVASCULAR DISEASES IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 18 MIDDLE EAST & AFRICA OTHER APPLICATION IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 19 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 20 MIDDLE EAST & AFRICA HOSPITAL AND SURGICAL CENTERS IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 21 MIDDLE EAST & AFRICA THERAPEUTIC COMPANIES IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 22 MIDDLE EAST & AFRICA SERVICES COMPANIES IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 23 MIDDLE EAST & AFRICA OTHERS IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 24 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 25 MIDDLE EAST & AFRICA DIRECT TENDER IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 26 MIDDLE EAST & AFRICA THIRD PARTY DISTRIBUTORS IN STEM CELL THERAPY MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 27 MIDDLE EAST AND AFRICA STEM CELL THERAPY MARKET, BY COUNTRY, 2020-2029 (USD MILLION)
TABLE 28 MIDDLE EAST AND AFRICA STEM CELL THERAPY MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 29 MIDDLE EAST AND AFRICA STEM CELL THERAPY MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 30 MIDDLE EAST AND AFRICA ALLOGENEIC STEM CELL THERAPY IN STEM CELL THERAPY MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 31 MIDDLE EAST AND AFRICA AUTOLOGOUS STEM CELL THERAPY IN STEM CELL THERAPY MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 32 MIDDLE EAST AND AFRICA STEM CELL THERAPY MARKET, BY APPLICATIONS, 2020-2029 (USD MILLION)
TABLE 33 MIDDLE EAST AND AFRICA STEM CELL THERAPY MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 34 MIDDLE EAST AND AFRICA STEM CELL THERAPY MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 35 ISRAEL STEM CELL THERAPY MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 36 ISRAEL STEM CELL THERAPY MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 37 ISRAEL ALLOGENEIC STEM CELL THERAPY IN STEM CELL THERAPY MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 38 ISRAEL AUTOLOGOUS STEM CELL THERAPY IN STEM CELL THERAPY MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 39 ISRAEL STEM CELL THERAPY MARKET, BY APPLICATIONS, 2020-2029 (USD MILLION)
TABLE 40 ISRAEL STEM CELL THERAPY MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 41 ISRAEL STEM CELL THERAPY MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
그림 목록
FIGURE 1 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET : SEGMENTATION
FIGURE 2 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: DATA TRIANGULATION
FIGURE 3 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: DROC ANALYSIS
FIGURE 4 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: MIDDLE EAST & AFRICA VS REGIONAL MARKET ANALYSIS
FIGURE 5 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: DBMR POSITION GRID
FIGURE 8 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: VENDOR SHARE ANALYSIS
FIGURE 9 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: END USER COVERAGE GRID
FIGURE 10 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: SEGMENTATION
FIGURE 11 NORTH AMERICA IS ANTICIPATED TO DOMINATE THE MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET AND ASIA-PACIFIC IS ESTIMATED TO BE GROWING WITH THE HIGHEST CAGR IN THE FORECAST PERIOD OF 2022 TO 2029
FIGURE 12 INCREASED INCIDENCE OF CHRONIC DISEASES, RISE IN CLINICAL TRIALS, GMP CERTIFICATION AND PRODUCT APPPROVALS IS EXPECTED TO DRIVE THE MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET FROM 2022 TO 2029
FIGURE 13 PRODUCT TYPE SEGMENT IS EXPECTED TO HAVE THE LARGEST SHARE OF THE MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET FROM 2022 & 2029
FIGURE 14 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF THE MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET
FIGURE 15 NUMBER AND AGES OF PEOPLE 65 OR OLDER WITH ALZHEIMER'S DEMENTIA IN 2022
FIGURE 16 INCIDENCE OF VARIOUS TYPES OF CANCER IN 2020
FIGURE 17 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY PRODUCT TYPE, 2021
FIGURE 18 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY PRODUCT TYPE, 2022-2029 (USD MILLION)
FIGURE 19 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY PRODUCT TYPE, CAGR (2022-2029)
FIGURE 20 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY PRODUCT TYPE, LIFELINE CURVE
FIGURE 21 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY TYPE, 2021
FIGURE 22 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY TYPE, 2022-2029 (USD MILLION)
FIGURE 23 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY TYPE, CAGR (2022-2029)
FIGURE 24 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY TYPE, LIFELINE CURVE
FIGURE 25 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY APPLICATION, 2021
FIGURE 26 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY APPLICATION, 2022-2029 (USD MILLION)
FIGURE 27 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY APPLICATION, CAGR (2022-2029)
FIGURE 28 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY APPLICATION, LIFELINE CURVE
FIGURE 29 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY END USER, 2021
FIGURE 30 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY END USER, 2022-2029 (USD MILLION)
FIGURE 31 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY END USER, CAGR (2022-2029)
FIGURE 32 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY END USER, LIFELINE CURVE
FIGURE 33 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY DISTRIBUTION CHANNEL, 2021
FIGURE 34 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY DISTRIBUTION CHANNEL, 2022-2029 (USD MILLION)
FIGURE 35 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY DISTRIBUTION CHANNEL, CAGR (2022-2029)
FIGURE 36 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 37 MIDDLE EAST AND AFRICA STEM CELL THERAPY MARKET: SNAPSHOT (2021)
FIGURE 38 MIDDLE EAST AND AFRICA STEM CELL THERAPY MARKET: BY COUNTRY (2021)
FIGURE 39 MIDDLE EAST AND AFRICA STEM CELL THERAPY MARKET: BY COUNTRY (2022 & 2029)
FIGURE 40 MIDDLE EAST AND AFRICA STEM CELL THERAPY MARKET: BY COUNTRY (2021 & 2029)
FIGURE 41 MIDDLE EAST AND AFRICA STEM CELL THERAPY MARKET: BY PRODUCT TYPE (2022-2029)
FIGURE 42 MIDDLE EAST & AFRICA STEM CELL THERAPY MARKET: COMPANY SHARE 2021 (%)

연구 방법론
데이터 수집 및 기준 연도 분석은 대규모 샘플 크기의 데이터 수집 모듈을 사용하여 수행됩니다. 이 단계에는 다양한 소스와 전략을 통해 시장 정보 또는 관련 데이터를 얻는 것이 포함됩니다. 여기에는 과거에 수집한 모든 데이터를 미리 검토하고 계획하는 것이 포함됩니다. 또한 다양한 정보 소스에서 발견되는 정보 불일치를 검토하는 것도 포함됩니다. 시장 데이터는 시장 통계 및 일관된 모델을 사용하여 분석하고 추정합니다. 또한 시장 점유율 분석 및 주요 추세 분석은 시장 보고서의 주요 성공 요인입니다. 자세한 내용은 분석가에게 전화를 요청하거나 문의 사항을 드롭하세요.
DBMR 연구팀에서 사용하는 주요 연구 방법론은 데이터 마이닝, 시장에 대한 데이터 변수의 영향 분석 및 주요(산업 전문가) 검증을 포함하는 데이터 삼각 측량입니다. 데이터 모델에는 공급업체 포지셔닝 그리드, 시장 타임라인 분석, 시장 개요 및 가이드, 회사 포지셔닝 그리드, 특허 분석, 가격 분석, 회사 시장 점유율 분석, 측정 기준, 글로벌 대 지역 및 공급업체 점유율 분석이 포함됩니다. 연구 방법론에 대해 자세히 알아보려면 문의를 통해 업계 전문가에게 문의하세요.
사용자 정의 가능
Data Bridge Market Research는 고급 형성 연구 분야의 선두 주자입니다. 저희는 기존 및 신규 고객에게 목표에 맞는 데이터와 분석을 제공하는 데 자부심을 느낍니다. 보고서는 추가 국가에 대한 시장 이해(국가 목록 요청), 임상 시험 결과 데이터, 문헌 검토, 재생 시장 및 제품 기반 분석을 포함하도록 사용자 정의할 수 있습니다. 기술 기반 분석에서 시장 포트폴리오 전략에 이르기까지 타겟 경쟁업체의 시장 분석을 분석할 수 있습니다. 귀하가 원하는 형식과 데이터 스타일로 필요한 만큼 많은 경쟁자를 추가할 수 있습니다. 저희 분석가 팀은 또한 원시 엑셀 파일 피벗 테이블(팩트북)로 데이터를 제공하거나 보고서에서 사용 가능한 데이터 세트에서 프레젠테이션을 만드는 데 도움을 줄 수 있습니다.













