北米の多発性硬化症治療市場
Market Size in USD Billion
CAGR :
% 
 USD
2,879.60 million
USD
5,737.78 million
2022
2030
USD
2,879.60 million
USD
5,737.78 million
2022
2030
| 2023 –2030 | |
| USD 2,879.60 million | |
| USD 5,737.78 million | |
|
|
|
北米の多発性硬化症治療市場、疾患タイプ(再発寛解型多発性硬化症(RRMS)、二次進行性多発性硬化症(SPMS)、一次進行性多発性硬化症(PPMS)、重症再発寛解型多発性硬化症(RES)、治療(予防療法、中絶療法/急性増悪の治療、対症療法)、薬剤タイプ(ブランド、ジェネリック)、エンドユーザー(病院および診療所、診断研究所、その他)– 2030年までの業界動向と予測。
北米の多発性硬化症治療市場の分析と規模
市場の成長の主な原因は、世界中の医療システムに対する多数の感染症の負担の増加です。多発性硬化症の発症率の増加、多発性硬化症のより良い治療オプションの必要性の高まり、政府の取り組みの拡大により、多発性硬化症治療の需要は前年に比べて増加しています。先進国と発展途上国の政府と非政府組織は、多発性硬化症の認知度を高めるために努力し、医薬品研究に多額の資金を提供しています。
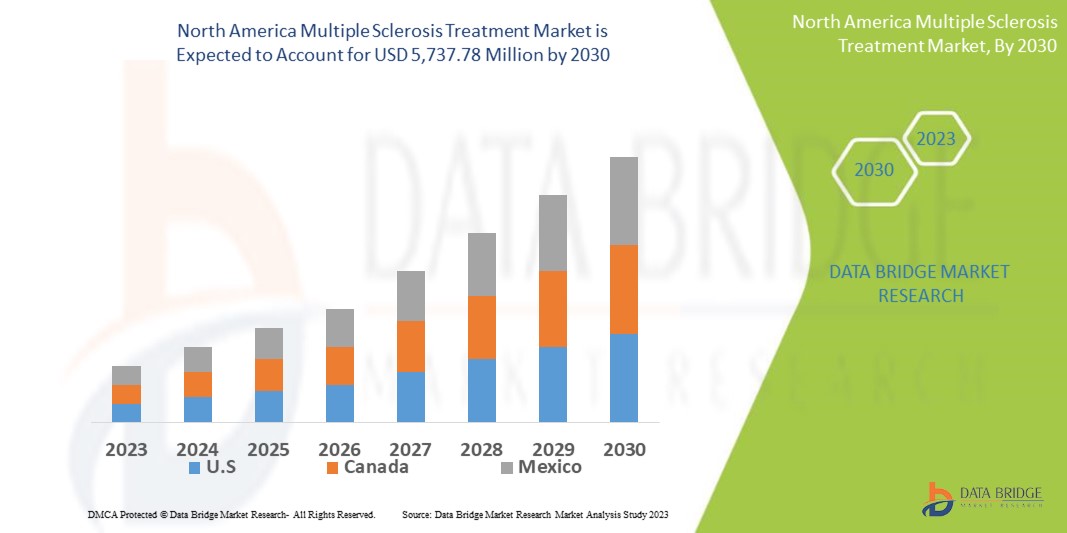
Data Bridge Market Research の分析によると、多発性硬化症治療市場は 2030 年までに 57 億 3,778 万米ドルに達し、2022 年には 28 億 7,960 万米ドルに達し、2023 年から 2030 年の予測期間中に 9% の CAGR で成長すると予想されています。市場価値、成長率、セグメンテーション、地理的範囲、主要プレーヤーなどの市場シナリオに関する洞察に加えて、Data Bridge Market Research がまとめた市場レポートには、詳細な専門家の分析、患者の疫学、パイプライン分析、価格分析、規制の枠組みも含まれています。
北米の多発性硬化症治療市場の範囲とセグメンテーション
|
レポートメトリック |
詳細 |
|
予測期間 |
2023年から2030年 |
|
基準年 |
2022 |
|
歴史的な年 |
2021 (2015 - 2020 にカスタマイズ可能) |
|
定量単位 |
売上高(百万米ドル)、販売数量(個数)、価格(米ドル) |
|
対象セグメント |
疾患タイプ(再発寛解型多発性硬化症(RRMS)、二次進行性多発性硬化症(SPMS)、一次進行性多発性硬化症(PPMS)、重症再発寛解型多発性硬化症(RES)、治療(予防療法、中絶療法/急性増悪の治療、対症療法)、薬剤タイプ(ブランド、ジェネリック)、エンドユーザー(病院、外傷センター、外来手術センター、その他) |
|
対象国 |
北米では米国、カナダ、メキシコ |
|
対象となる市場プレーヤー |
AbbVie Inc. (米国)、Bausch Health Companies Inc. (カナダ)、Biora Therapeutics, Inc. (米国)、Boehringer Ingelheim International Gmbh (ドイツ)、Amgen Inc. (米国)、Pfizer Inc (米国)、F. Hoffmann-La Roche Ltd (スイス)、Mylan NV (米国)、Novartis AG (スイス)、Bayer AG (ドイツ)、Bristol Myers Squibb Company (米国)、Biogen (米国)、Teva Pharmaceutical Industries Ltd (イスラエル)、武田薬品工業株式会社 (日本)、Jazz Pharmaceuticals, Inc (英国)、Abbott (米国)、Bio-Rad Laboratories Inc. (米国)、Mylan NV (米国) |
|
市場機会 |
|
市場の定義
多発性硬化症 (MS) は、脊髄と脳神経細胞の軸索切断、脱髄、神経変性を引き起こす可能性がある、慢性で炎症性の免疫介在性疾患です。症状は、損傷した神経の量、影響を受けた神経の形状と位置によって異なります。多発性硬化症は、痛み、疲労、視力低下、協調運動障害など、さまざまな症状を引き起こします。この病気の期間、症状、重症度は人によって異なります。
北米の多発性硬化症治療市場の動向
ドライバー
- 複数の市場プレーヤーによる医薬品開発プロセスの増加
いくつかの製薬会社も、医薬品開発プロセスに多額の投資を行っています。彼らは、多くの顧客を引き付けるために、さまざまな適応症をターゲットにすることを計画しています。たとえば、多発性硬化症用に開発された薬には、二次進行型MS、一次進行型MS、再発寛解型MS、およびミエリン修復または神経保護が含まれます。たとえば、ノバルティスは、臨床的または画像的特徴によって定義される活動性疾患のある成人の再発型多発性硬化症(RMS)の治療薬として、2021年3月に欧州委員会からオファツムマブの承認を取得しました。したがって、この要因は市場の成長を後押しします。
- ブランド医薬品の需要増加
ブランド薬の需要の高まりは、市場の成長を加速させます。多数の企業が随時多数の製品を発売しているため、需要が増加しており、ジェネリック医薬品がそれを打ち負かすのは困難です。さらに、安定性、有効性、剤形、製造、安全性、ラベル、化学、パッケージの証拠とともに新薬申請が提出されると、FDA はブランド薬を承認するため、需要が大幅に増加します。したがって、市場の成長が促進されます。
機会
- 製品発売の増加
多発性硬化症の治療薬の発売が増加しており、市場の成長を促進しています。たとえば、マイランNVは、再発性多発性硬化症(MS)の治療に使用される、FDA承認のジェネリック医薬品ジメチルフマル酸塩遅延放出カプセル120 mgおよび240 mgの発売を発表しました。新しく発売された薬は、バイオジェンのテクフィデラカプセルと治療上ほぼ同様です。COVID-19パンデミック中に同社が発売したこの新しいFDA承認薬は、市場での需要を高めています。
- 経口投与の需要の高まり
多発性硬化症の治療薬として最近承認されたいくつかの新しい経口薬は、治療における大きな進歩を表しています。経口投与は患者の満足度を高め、治療コンプライアンスを高めます。たとえば、欧州委員会(EC)は、2021年6月に、再発寛解型多発性硬化症(RRMS)を患う10〜17歳の小児患者の治療薬としてAubagio(テリフルノミド)を承認しました。Aubagioは、欧州連合でMSを患う小児および成人の第一選択治療に適用できる初の経口多発性硬化症(MS)治療薬です。さらに、ブリストル・マイヤーズスクイブ社は、2020年3月に、再発型多発性硬化症(RMS)の成人、再発寛解型疾患、臨床的に孤立した症候群、および活動性二次進行性疾患の治療薬として、ZEPOSIA(オザニモド)0.92 mgの米国FDA承認を取得しました。したがって、この要因は市場の成長率を高めます。
制約/課題
- 多発性硬化症治療薬の副作用
多発性硬化症には、市場の成長を妨げるさまざまな副作用があります。インフルエンザのような症状、胸痛、心拍数の変動、まれな脳感染症、化学療法のような効果などの影響により、患者は多発性硬化症治療薬を服用しにくくなります。この要因により、市場の成長が制限されると予想されます。
この多発性硬化症治療市場レポートでは、最近の新しい開発、貿易規制、輸出入分析、生産分析、バリュー チェーンの最適化、市場シェア、国内および現地の市場プレーヤーの影響、新たな収益源の観点から見た機会の分析、市場規制の変更、戦略的市場成長分析、市場規模、カテゴリ市場の成長、アプリケーションのニッチと優位性、製品の承認、製品の発売、地理的拡大、市場における技術革新などの詳細が提供されます。多発性硬化症治療市場に関する詳細情報を取得するには、アナリスト ブリーフについて Data Bridge Market Research にお問い合わせください。当社のチームが、情報に基づいた市場決定を行い、市場の成長を達成できるようお手伝いします。
最近の動向:
- 2020年、ジェンザイム社はプリンシピア・バイオファーマ社との買収契約を締結しました。この買収の主な目的は、多発性硬化症やその他の免疫介在性疾患の分野における研究活動を改善することでした。
- ノバルティスは2020年、成人の臨床的に孤立した症候群、再発寛解型疾患、活動性二次進行性疾患を含む再発型多発性硬化症(RMS)の治療薬として、皮下注射剤の形でオキサプタを米国FDAが承認したことを発表しました。
北米の多発性硬化症治療市場の範囲
多発性硬化症治療市場は、疾患の種類、治療、薬剤の種類、エンドユーザーに基づいてセグメント化されています。これらのセグメントの成長は、業界におけるわずかな成長セグメントの分析に役立ち、ユーザーに貴重な市場概要と市場洞察を提供して、コア市場アプリケーションを特定するための戦略的決定を下すのに役立ちます。
病気の種類
- 再発寛解型多発性硬化症(RRMS)
- 二次進行性多発性硬化症(SPMS)
- 原発性進行性多発性硬化症(PPMS)
- 重症再発寛解型多発性硬化症(RES)
処理
- 予防療法
- 急性増悪の阻止療法/治療
- 対症療法
薬剤の種類
- ブランド
- ジェネリック
投与経路
- オーラル
- 非経口
エンドユーザー
- 病院と診療所
- 診断検査室
- その他
多発性硬化症治療の地域分析/洞察
多発性硬化症治療市場が分析され、上記のように病気の種類、治療、薬剤の種類、エンドユーザー別に市場規模の洞察と傾向が提供されます。
多発性硬化症治療市場レポートで取り上げられている国は、北米では米国、カナダ、メキシコです。
米国は、この地域で多発性硬化症の最新の治療法が導入されるため、2023年から2030年の予測期間に市場をリードすると予想されています。この地域での医薬品開発のための研究開発の増加も、市場の成長を加速させています。多発性硬化症とその治療に関連する人々の意識の高まりは、市場の成長を牽引するのに役立っています。
レポートの国別セクションでは、市場の現在および将来の傾向に影響を与える国内市場における個別の市場影響要因と規制の変更も提供しています。下流および上流のバリュー チェーン分析、技術動向、ポーターの 5 つの力の分析、ケース スタディなどのデータ ポイントは、各国の市場シナリオを予測するために使用される指標の一部です。また、国別データの予測分析を提供する際には、北米ブランドの存在と可用性、および地元および国内ブランドとの競争が激しいか少ないために直面する課題、国内関税と貿易ルートの影響も考慮されます。
競争環境と北米の多発性硬化症治療市場シェア分析
多発性硬化症治療市場の競争状況は、競合他社ごとに詳細を提供します。詳細には、会社概要、会社の財務状況、収益、市場の可能性、研究開発への投資、新しい市場への取り組み、北米でのプレゼンス、生産拠点と施設、生産能力、会社の強みと弱み、製品の発売、製品の幅と広さ、アプリケーションの優位性が含まれます。提供されている上記のデータ ポイントは、多発性硬化症治療市場に関連する会社の焦点にのみ関連しています。
多発性硬化症治療市場で活動している主要企業は次のとおりです。
- アッヴィ社(米国)
- バウシュ・ヘルス・カンパニーズ(カナダ)
- バイオラ・セラピューティクス社(米国)
- ベーリンガーインゲルハイムインターナショナルGmbh(ドイツ)
- アムジェン社(米国)
- ファイザー社(米国)
- F. ホフマン・ラ・ロシュ社(スイス)
- マイランNV(米国)
- ノバルティスAG(スイス)
- バイエルAG(ドイツ)
- ブリストル マイヤーズ スクイブ カンパニー (米国)
- バイオジェン(米国)
- テバ製薬工業株式会社(イスラエル)
- 武田薬品工業株式会社(日本)
- ジャズ・ファーマシューティカルズ社(英国)
- アボット(米国)
- バイオ・ラッド・ラボラトリーズ社(米国)
- マイランNV(米国)
SKU-
世界初のマーケットインテリジェンスクラウドに関するレポートにオンラインでアクセスする
- インタラクティブなデータ分析ダッシュボード
- 成長の可能性が高い機会のための企業分析ダッシュボード
- カスタマイズとクエリのためのリサーチアナリストアクセス
- インタラクティブなダッシュボードによる競合分析
- 最新ニュース、更新情報、トレンド分析
- 包括的な競合追跡のためのベンチマーク分析のパワーを活用
調査方法
データ収集と基準年分析は、大規模なサンプル サイズのデータ収集モジュールを使用して行われます。この段階では、さまざまなソースと戦略を通じて市場情報または関連データを取得します。過去に取得したすべてのデータを事前に調査および計画することも含まれます。また、さまざまな情報ソース間で見られる情報の不一致の調査も含まれます。市場データは、市場統計モデルと一貫性モデルを使用して分析および推定されます。また、市場シェア分析と主要トレンド分析は、市場レポートの主要な成功要因です。詳細については、アナリストへの電話をリクエストするか、お問い合わせをドロップダウンしてください。
DBMR 調査チームが使用する主要な調査方法は、データ マイニング、データ変数が市場に与える影響の分析、および一次 (業界の専門家) 検証を含むデータ三角測量です。データ モデルには、ベンダー ポジショニング グリッド、市場タイムライン分析、市場概要とガイド、企業ポジショニング グリッド、特許分析、価格分析、企業市場シェア分析、測定基準、グローバルと地域、ベンダー シェア分析が含まれます。調査方法について詳しくは、お問い合わせフォームから当社の業界専門家にご相談ください。
カスタマイズ可能
Data Bridge Market Research は、高度な形成的調査のリーダーです。当社は、既存および新規のお客様に、お客様の目標に合致し、それに適したデータと分析を提供することに誇りを持っています。レポートは、対象ブランドの価格動向分析、追加国の市場理解 (国のリストをお問い合わせください)、臨床試験結果データ、文献レビュー、リファービッシュ市場および製品ベース分析を含めるようにカスタマイズできます。対象競合他社の市場分析は、技術ベースの分析から市場ポートフォリオ戦略まで分析できます。必要な競合他社のデータを、必要な形式とデータ スタイルでいくつでも追加できます。当社のアナリスト チームは、粗い生の Excel ファイル ピボット テーブル (ファクト ブック) でデータを提供したり、レポートで利用可能なデータ セットからプレゼンテーションを作成するお手伝いをしたりすることもできます。