ヨーロッパの静脈疾患治療市場
Market Size in USD Billion
CAGR :
% 
 USD
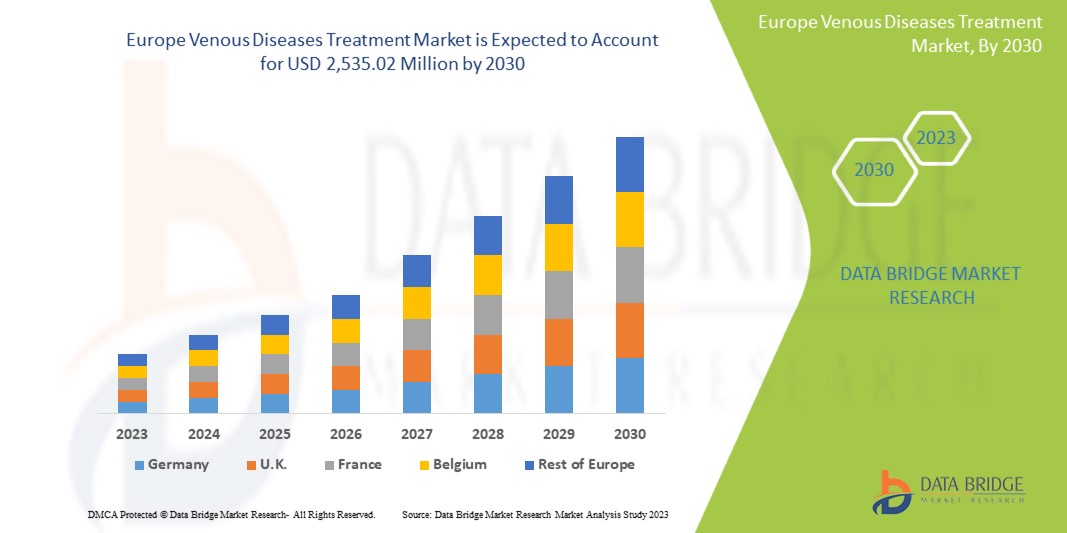
1,264.21 million
USD
2,535.02 million
2023
2030
USD
1,264.21 million
USD
2,535.02 million
2023
2030
| 2024 –2030 | |
| USD 1,264.21 million | |
| USD 2,535.02 million | |
|
|
|
ヨーロッパの静脈疾患治療市場、製品タイプ別(硬化療法注射、アブレーション装置、静脈閉鎖製品、静脈ステント、薬剤など)、疾患タイプ別(深部静脈血栓症(DVT)、慢性静脈不全症(CVI)、肺塞栓症、表在性血栓性静脈炎、静脈瘤など)、治療タイプ別(硬化療法、高周波アブレーション療法、レーザー治療、外来静脈切除術、静脈結紮および剥離術、血管形成術またはステント留置術、手術、圧迫療法、静脈活性薬剤、大静脈フィルターおよびその他の療法)、エンドユーザー別(病院、診療所、外来手術センターなど)、流通チャネル別(直接入札、小売販売など) - 2030年までの業界動向と予測。
ヨーロッパの静脈疾患治療市場の分析と洞察
静脈疾患には、脚、腕、脳、肺、または腎臓、脾臓、肝臓などの内臓の血栓、深部静脈血栓症、慢性静脈不全、静脈瘤、静脈、静脈の潰瘍が含まれます。この疾患の治療には、薬物療法、内因性レーザー焼灼術または高周波焼灼術 (RFA)、硬化療法、および手術が含まれます。
Data Bridge Market Research は、ヨーロッパの静脈疾患治療市場は、予測期間中に 6.8% の CAGR で成長し、2030 年までに 25 億 3,502 万米ドルに達すると予測しています。この市場レポートでは、価格分析、特許分析、技術進歩についても詳細に取り上げています。
|
レポートメトリック |
詳細 |
|
予測期間 |
2023年から2030年 |
|
基準年 |
2022 |
|
歴史的な年 |
2021(2020-1015にカスタマイズ可能) |
|
定量単位 |
収益は百万ドル、価格は米ドル |
|
対象セグメント |
製品タイプ別(硬化療法注射、アブレーション装置、静脈閉鎖製品、静脈ステント、薬剤など)、疾患タイプ別(深部静脈血栓症(DVT)、慢性静脈不全症(CVI)、肺塞栓症、表在性血栓性静脈炎、静脈瘤など)、治療タイプ別(硬化療法、高周波アブレーション療法、レーザー治療、外来静脈切除術、静脈結紮および剥離術、血管形成術またはステント留置術、外科手術、圧迫療法、静脈活性薬剤、大静脈フィルターおよびその他の療法)、エンドユーザー別(病院、診療所、外来手術センターなど)、流通チャネル別(直接入札、小売販売など) |
|
対象国 |
ドイツ、フランス、ロシア、イタリア、イギリス、スペイン、トルコ、オランダ、ベルギー、スイス、イスラエル、その他のヨーロッパ諸国 |
|
対象となる市場プレーヤー |
Abbott、Imricor、Baylis Medical Company、Inc.、Theraclion、Sonablate、plusmedica.de、Boston Scientific Corporation、Olympus Corporation、Smith + Nephew、Cook、Scitech、Carl Zeiss Meditec AG、Teleflex corporate、Alma Lasers、BD、B.Braun SE、Medtronic、Stryker、Koninklijke Philips NV、Varian Medical Systems、Candela Corporation、Teromo corporation、Angiodynamics、optimed Medizinische Instrumente GmbH、Merit Medical Systems、Bolitec Laser など。 |
欧州静脈疾患治療市場の定義
心臓発作や脳卒中は通常、急性の症状で、主に心臓や脳への血流を遮断する閉塞によって引き起こされます。最も一般的な原因は、心臓や脳に血液を供給する血管の内層に脂肪沈着物が蓄積することです。脳卒中は、脳の血管の出血や血栓によって引き起こされることがあります。
血管ステントの技術的進歩、低侵襲手術の需要増加、および高齢者人口の増加が、ヨーロッパ市場を牽引しています。さらに、企業は静脈疾患治療のための最良のサービスを提供するために製品ポートフォリオを拡大しています。アブレーション装置などの医療機器は、治療目的で異常な体組織を除去または切除するための低侵襲手術で使用されます。これらのシステムは、無線周波数、エネルギー、極寒、またはレーザーによって生成された熱を使用して小さな火傷を引き起こします。製品用途の拡大のためのロボット技術の採用の増加、および患者の安全性と手順の効率を向上させるためのアブレーション装置への最先端技術の統合が、市場を牽引すると予想されます。
ヨーロッパの静脈疾患治療市場の動向
このセクションでは、市場の推進要因、利点、機会、制約、課題について理解します。これらについては、以下で詳しく説明します。
ドライバー
- 静脈疾患の発生率上昇
静脈疾患は、体内の静脈に損傷を与える病気です。血管壁が損傷すると循環器系が機能しなくなり、筋肉が弛緩すると血液が溜まり、逆流します。これにより、静脈内に異常に高い圧力が蓄積します。この蓄積により、静脈が引き締まり、ねじれ、腫れが増し、弁の機能不全が進み、血流が遅くなり、血栓ができる可能性があります。最終的に、この病気は静脈疾患と呼ばれるさまざまな病気につながる可能性があります。
加齢、肥満、高血圧、静脈疾患の家族歴などのさまざまなリスク要因により、静脈疾患の患者は世界的に増加しており、重大な社会経済的問題となっています。したがって、静脈疾患の患者数の増加により、静脈疾患治療の需要が増加し、ヨーロッパの静脈疾患治療市場の推進力となっています。
- ライフスタイルの急激な変化は肥満につながり、静脈疾患を引き起こす
喫煙、不健康な食生活、運動不足などのライフスタイルの変化は、慢性疾患、特に心臓病、脳卒中、糖尿病、肥満、メタボリックシンドロームの発症につながり、最終的には静脈疾患につながる可能性があります。
身体活動は不可欠であり、多くの病気は座りがちな生活習慣の直接的な結果です。座りがちな生活習慣は、体重増加、疲労感、原因不明の痛みや苦痛を引き起こし、中程度から重度、さらには生命を脅かす心臓血管疾患や静脈疾患などの慢性疾患を引き起こす可能性があります。
ScienceDirect の記事によると、静脈疾患は非肥満グループよりも肥満の手足で臨床的に重篤です。したがって、不健康なライフスタイルの採用の増加により、静脈疾患が増加している肥満人口が急増しています。したがって、ライフスタイルの急速な変化は肥満につながり、静脈疾患を引き起こし、静脈疾患治療の需要が増加し、ヨーロッパの静脈疾患治療市場の推進力として機能します。
拘束
- 熟練した資格を持った専門家の不足
熟練した資格を持った専門家の要件は、静脈疾患治療市場にとって大きな制約となっています。ヨーロッパでは静脈疾患の症例が増加しているため、静脈疾患治療の需要は高まっていますが、医療センターに在籍する熟練した専門家の数が少ないことが市場の成長を妨げています。
慢性静脈疾患 (CVD) は、医療従事者が問題の範囲と影響を理解しておらず、一次静脈疾患と二次静脈疾患の異なる症状が十分に認識されていないため、見過ごされがちです。心血管疾患の重要性は、患者数と、より深刻な症状の社会経済的影響に関係しています。
機会
- 静脈疾患に対する意識の高まり
静脈の問題の治療には、薬物療法と生活習慣の変更の両方が利用できます。静脈は、薬物療法、運動、着圧ストッキングで改善できますが、静脈の問題によっては、静脈の健康と機能を回復するために、より徹底した治療が必要になることもあります。治療方針は、静脈疾患の種類と重症度によって決まります。過去数十年間で血管疾患の患者は劇的に増加しており、糖尿病が最も顕著なリスク要因となっています。他の健康問題とは異なり、血管疾患と血管手術は人口の約 80% に知られていません。健康意識の高まりにより、特定の病気に対する人々の態度や行動は大きく変わる可能性があります。
さまざまな団体、政府機関などによって、いくつかの啓発プログラムが実施されています。これらの取り組みにより、人々の健康に関する意識が高まり、よりよい治療と予防策のために早期診断が行われます。このため、静脈疾患に対する意識の高まりは、ヨーロッパの静脈疾患治療市場の需要拡大の機会となることが期待されています。
チャレンジ
- 静脈疾患の治療にかかる高額な費用
米国だけで 2,500 万人以上が慢性静脈不全 (CVI) を患っており、600 万人以上が進行した静脈疾患を患っています。米国の医療制度は、CVI の発症率の高さと医療費の上昇により、財政的に大きな負担を強いられています。広範囲にわたる数多くの健康問題は、静脈循環不良が原因です。脚の痛み、浮腫、重苦しさは慢性静脈疾患 (CVD) の初期症状の一部であり、一日中続くこともあれば、夕方に顕著になることもあります。患者は、静脈瘤の有無にかかわらず起こりうる症状の初期治療や、美容上の静脈瘤除去を求めることがよくあります。CVD の 2 つの主なリスク要因は、高齢化と体重過多です。
COVID-19後の欧州静脈疾患治療市場への影響
パンデミックはメーカーだけでなくユーザーにも悪影響を及ぼしました。静脈瘤の手術は緊急性が低いため、手術件数は大幅に減少しました。旅行制限により静脈瘤の治療のための手術や処置が減少したことも、メーカーの売上に影響を与えました。
メーカーは、COVID-19後の回復に向けてさまざまな戦略的決定を下しています。各社は、ペットフードのフレーバーと原料市場に関わる技術とテスト結果を改善するために、複数の研究開発活動、製品の発売、戦略的パートナーシップを行っています。
最近の動向
- 2022年7月、ヨーロッパの医療技術企業であるスミス・ネフューは、創傷ケアにおける実践のばらつきを減らすための臨床サポートアプリをリリースしました。WOUND COMPASS臨床サポートアプリは、医療従事者向けの包括的なデジタルサポートツールで、創傷の評価と意思決定を支援し、実践のばらつきを減らすのに役立ちます。これにより、同社はヘルスケア顧客を引き付け、製品ポートフォリオを拡大することができました。
- 2022年4月、カールツァイスメディテックは、ソリューションプロバイダーとしての地位をさらに強化するために、外科用器具メーカー2社(Kogent Surgical、LLCおよびKatalyst Surgical、LLC)の買収を発表しました。これにより、同社は事業を拡大することができました。
欧州の静脈疾患治療市場の範囲
ヨーロッパの静脈疾患治療市場は、製品タイプ、疾患タイプ、治療タイプ、エンドユーザー、流通チャネルに区分されています。セグメント間の成長は、ニッチな成長分野と市場へのアプローチ戦略を分析し、コアアプリケーション領域とターゲット市場の違いを決定するのに役立ちます。
欧州の静脈疾患治療市場(製品タイプ別)
- アブレーション装置
- 静脈ステント
- 静脈閉鎖製品
- 硬化療法注射
- 医薬品
- その他
製品タイプに基づいて、ヨーロッパの静脈疾患治療市場は、アブレーション装置、静脈ステント、静脈閉鎖製品、硬化療法注射、医薬品、その他に分類されます。
欧州の静脈疾患治療市場(疾患タイプ別)
- 深部静脈血栓症(DVT)
- 慢性静脈不全(CVI)
- 肺塞栓症
- 表在性血栓性静脈炎
- 静脈瘤
- その他
疾患の種類に基づいて、ヨーロッパの静脈疾患治療市場は、深部静脈血栓症(DVT)、慢性静脈不全(CVI)、肺塞栓症、表在性血栓性静脈炎、静脈瘤などに分類されます。
欧州の静脈疾患治療市場(治療タイプ別)
- 圧迫療法
- VENO ACTIVE メディケーション
- 手術
- 硬化療法
- 血管形成術またはステント留置術
- 静脈結紮術と静脈剥離術
- 大静脈フィルター
- 外来静脈切除術
- 高周波アブレーション療法
- レーザー治療
- その他の治療法
治療の種類に基づいて、ヨーロッパの静脈疾患治療市場は、圧迫療法、静脈活性薬、外科手術、硬化療法、血管形成術またはステント留置術、静脈結紮術およびストリッピング術、大静脈フィルター、外来静脈切除術、高周波アブレーション療法、レーザー治療、およびその他の療法に分類されます。
欧州の静脈疾患治療市場(エンドユーザー別)
- 病院
- クリニック
- 外来手術センター
- その他
エンドユーザーに基づいて、ヨーロッパの静脈疾患治療市場は、病院、診療所、外来手術センター、その他に分類されます。
欧州静脈疾患治療市場(流通チャネル別)
- 直接入札
- 小売販売
- その他
流通チャネルに基づいて、ヨーロッパの静脈疾患治療市場は、直接入札、小売販売、その他に分類されます。
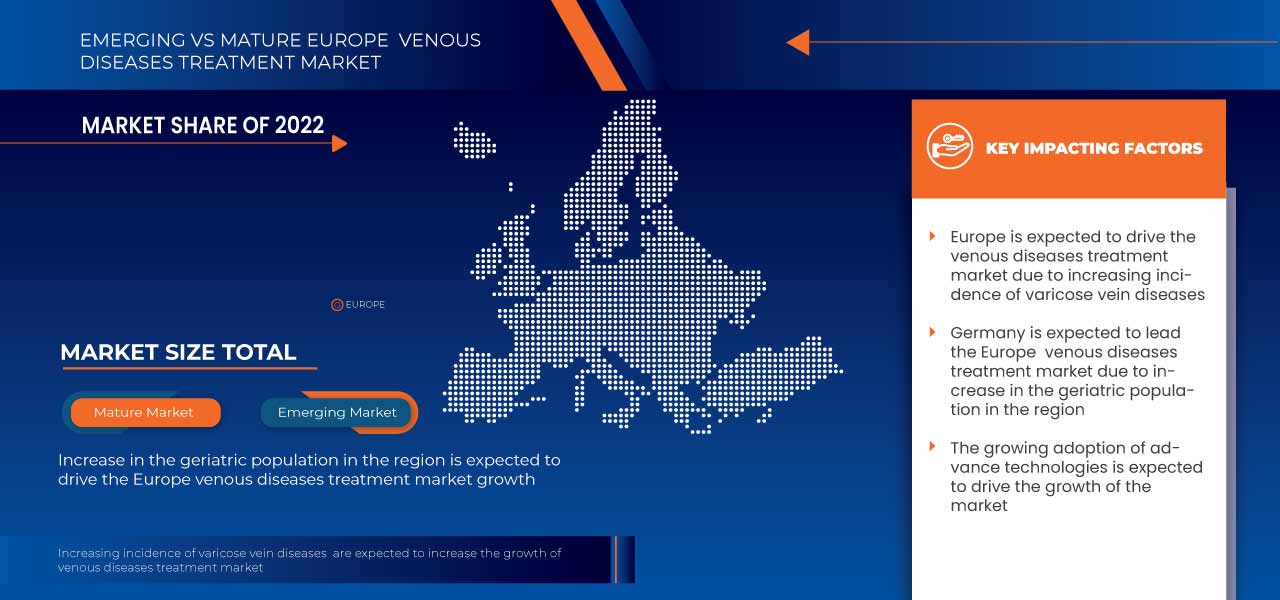
ヨーロッパの静脈疾患治療市場の地域分析/洞察
ヨーロッパの静脈疾患治療市場が分析され、製品タイプ、疾患タイプ、治療タイプ、エンドユーザー、流通チャネルに関する市場規模情報が提供されます。
この市場レポートでカバーされている国は、ドイツ、フランス、ロシア、イタリア、英国、スペイン、トルコ、オランダ、ベルギー、スイス、イスラエル、その他のヨーロッパ諸国です。
ドイツは、静脈疾患治療薬の大量生産、新興市場からの需要増加、静脈疾患治療産業の拡大により、ヨーロッパ地域で優位に立っています。
レポートの国別セクションでは、市場の現在および将来の傾向に影響を与える国内市場における個別の市場影響要因と規制の変更も提供しています。新規販売、交換販売、国の人口統計、規制行為、輸出入関税などのデータ ポイントは、各国の市場シナリオを予測するために使用される主要な指標の一部です。また、国別データの予測分析を提供する際には、ヨーロッパ ブランドの存在と可用性、地元および国内ブランドとの競争が激しいか少ないために直面する課題、販売チャネルの影響も考慮されます。
競争環境と欧州の静脈疾患治療市場シェア分析
ヨーロッパの静脈疾患治療市場の競争状況は、競合他社ごとに詳細を提供します。含まれる詳細には、会社概要、会社の財務状況、収益、市場の可能性、研究開発への投資、新しい市場への取り組み、生産拠点と施設、会社の強みと弱み、製品の発売、製品試験パイプライン、製品の承認、特許、製品の幅と幅、アプリケーションの優位性、技術ライフライン曲線などがあります。提供されている上記のデータ ポイントは、ヨーロッパの静脈疾患治療市場への会社の重点にのみ関連しています。
ヨーロッパの静脈疾患治療市場で活動している主要企業には、Abbott、Imricor、Baylis Medical Company、Inc.、Theraclion、Sonablate、plusmedica.de、Boston Scientific Corporation、Olympus Corporation、Smith + Nephew、Cook、Scitech、Carl Zeiss Meditec AG、Teleflex corporate、Alma Lasers、BD、B.Braun SE、Medtronic、Stryker、Koninklijke Philips NV、Varian Medical Systems、Candela Corporation、Teromo corporation、Angiodynamics、optimed Medizinische Instrumente GmbH、Merit Medical Systems、Bolitec Laserなどがあります。
SKU-
世界初のマーケットインテリジェンスクラウドに関するレポートにオンラインでアクセスする
- インタラクティブなデータ分析ダッシュボード
- 成長の可能性が高い機会のための企業分析ダッシュボード
- カスタマイズとクエリのためのリサーチアナリストアクセス
- インタラクティブなダッシュボードによる競合分析
- 最新ニュース、更新情報、トレンド分析
- 包括的な競合追跡のためのベンチマーク分析のパワーを活用
目次
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF THE EUROPE VENOUS DISEASES TREATMENT MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATIONS
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 DBMR TRIPOD DATA VALIDATION MODEL
2.5 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.6 MULTIVARIATE MODELLING
2.7 MARKET END USER COVERAGE GRID
2.8 INSTRUMENT BASED LIFELINE CURVE
2.9 DBMR MARKET POSITION GRID
2.1 VENDOR SHARE ANALYSIS
2.11 SECONDARY SOURCES
2.12 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PESTEL ANALYSIS
4.2 PORTER'S FIVE FORCES
5 EUROPE VENOUS DISEASES TREATMENT MARKET, INDUSTRY INSIGHTS
6 EPIDEMIOLOGY
7 REGULATORY FRAMEWORK
8 MARKET OVERVIEW
8.1 DRIVERS
8.1.1 RISING INCIDENCES OF VENOUS DISEASES
8.1.2 RAPID CHANGES IN LIFESTYLE LEAD TO OBESITY RESULTING IN VENOUS DISEASES
8.1.3 INCREASE IN THE GERIATRIC POPULATION
8.1.4 TECHNOLOGICALLY ADVANCEMENT IN THE TREATMENT OF VENOUS DISEASES
8.2 RESTRAINTS
8.2.1 LACK OF SKILLED AND CERTIFIED PROFESSIONALS
8.2.2 INADEQUATE REIMBURSEMENT COVERAGE
8.3 OPPORTUNITIES
8.3.1 RISING AWARENESS TOWARDS VENOUS DISORDERS
8.3.2 NEED FOR PROPER DIAGNOSIS AND TREATMENT OF VENOUS DISEASES
8.3.3 GROWING PREFERENCE FOR MINIMALLY INVASIVE PROCEDURES
8.3.4 OCCUPATIONAL LIFESTYLE INCREASES THE NEED FOR VENOUS DISEASE TREATMENT
8.4 CHALLENGES
8.4.1 HIGH COST ASSOCIATED WITH VENOUS DISEASE TREATMENT
8.4.2 SIDE EFFECTS AND RISK ASSOCIATED WITH DIFFERENT TREATMENT MODES
9 EUROPE VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE
9.1 OVERVIEW
9.2 SCLEROTHERAPY INJECTION
9.2.1 SCLEROTHERAPY, BY TYPE
9.2.1.1 INTRAVENOUS
9.2.1.2 INTRADERMAL
9.2.1.3 SUBCUTANEOUS
9.2.2 SUBCUTANEOUS, BY APPLICATION
9.2.2.1 MALFORMED LYMPHED VESSELS
9.2.2.2 HEMORRHOIDS
9.2.2.3 HYDROCELES
9.2.2.4 OTHERS
9.3 ABLATION DEVICES
9.3.1 THERMAL ABLATION
9.3.1.1 RADIOFREQUENCY
9.3.1.1.1 CATHETER MANIPULATION SYSTEMS
9.3.1.1.2 TEMPERATURE CONTROLLED
9.3.1.1.3 FLUID COOLED
9.3.1.2 LIGHT
9.3.1.2.1 EXCIMER LASERS
9.3.1.2.2 COLD LASERS
9.3.1.3 ULTRASOUND
9.3.1.3.1 HIGH INTENSITY FOCUSED ULTRASOUND (HIFU)
9.3.1.3.2 SHOCK WAVE LITHOTRIPSY
9.3.1.3.3 MAGNETIC RESONANCE IMAGING GUIDED FOCUSED ULTRASOUND (MRI-FUS)
9.3.1.3.4 ULTRASONIC SURGICAL SYSTEMS
9.3.1.4 RADIATION
9.3.1.4.1 STEREOTACTIC BODY RADIATION THERAPY
9.3.1.4.2 INTENSITY-MODULATED RADIATION THERAPY
9.3.1.4.3 STEREOTACTIC RADIOTHERAPY & RADIOSURGERY
9.3.1.4.4 IMAGE GUIDED RADIATION THERAPY
9.3.1.4.5 INTRAVASCULAR BRACHYTHERAPY
9.3.1.4.6 PROTON BEAM THERAPY
9.3.1.5 ELECTRICAL
9.3.1.5.1 ELECTRICAL ABLATORS
9.3.1.5.2 ELECTRONIC BRACHYTHERAPY
9.3.1.6 MICROWAVE
9.3.1.7 HYDROTHERMAL
9.3.2 NON-THERMAL ABLATION
9.3.2.1 CRYOABLATION
9.3.2.1.1 EPIDERMAL AND SUBCUTANEOUS CRYOABLATION DEVICES
9.3.2.1.2 CRYOGEN SPRAY PROBE
9.3.2.1.3 TISSUE CONTACT PROBE
9.3.2.2 HYDROMECHANICAL ABLATION
9.4 VENOUS CLOSURE PRODUCTS
9.4.1 VENOUS CLOSURE PRODUCTS, BY PROCEDURE
9.4.1.1 INTERVENTIONAL CARDIOLOGY
9.4.1.2 INTERVENTIONAL RADIOLOGY
9.4.2 VENOUS CLOSURE PRODUCTS, BY TECHNOLOGY
9.4.2.1 FEMORAL ACCESS TECHNIQUE
9.4.2.2 RADIAL ACCESS TECHNIQUE
9.5 VENOUS STENTS
9.5.1 VENOUS STENTS, BY TECHNOLOGY
9.5.1.1 WALLSTENT TECHNOLOGY
9.5.1.2 ILIAC VEIN STENT TECHNOLOGY
9.5.2 VENOUS STENTS, BY APPLICATION
9.5.2.1 LEG
9.5.2.2 CHEST
9.5.2.3 ABDOMEN
9.5.2.4 OTHERS
9.6 MEDICATION
9.7 OTHERS
10 EUROPE VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE
10.1 OVERVIEW
10.2 VARICOSE VEINS
10.3 DEEP VEIN THROMBOSIS (DVT)
10.4 PULMONARY EMBOLISM
10.5 CHRONIC VENOUS INSUFFICIENCY (CVI)
10.6 SUPERFICIAL THROMBOPHLEBITIS
10.7 OTHERS
11 EUROPE VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE
11.1 OVERVIEW
11.2 SCLEROTHERAPY
11.3 RADIOFREQUENCY ABLATION THERAPY
11.4 LASER TREATMENT
11.5 AMBULATORY PHLEBECTOMY
11.6 VEIN LIGATION AND STRIPPING
11.7 ANGIOPLASTY OR STENTING
11.8 SURGERIES
11.9 COMPRESSION THERAPY
11.1 VEINACTIVE MEDICATION
11.11 VENA CAVA FILTER
11.12 OTHER THERAPIES
12 EUROPE VENOUS TREATMENT DISEASES MARKET, BY END USER
12.1 OVERVIEW
12.2 HOSPITALS
12.3 CLINICS
12.4 AMBULATORY SURGICAL CENTERS
12.5 OTHERS
13 EUROPE VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL
13.1 OVERVIEW
13.2 DIRECT TENDER
13.3 RETAIL SALES
13.4 OTHERS
14 EUROPE VENOUS DISEASES TREATMENT MARKET, BY REGION
14.1 EUROPE
14.1.1 GERMANY
14.1.2 U.K.
14.1.3 FRANCE
14.1.4 ITALY
14.1.5 RUSSIA
14.1.6 SPAIN
14.1.7 TURKEY
14.1.8 NETHERLANDS
14.1.9 BELGIUM
14.1.10 SWITZERLAND
14.1.11 IRELAND
14.1.12 REST OF EUROPE
15 EUROPE VENOUS DISEASES TREATMENT MARKET, COMPANY LANDSCAPE
15.1 COMPANY SHARE ANALYSIS: EUROPE
16 SWOT ANALYSIS
17 COMPANY PROFILE
17.1 MEDTRONIC
17.1.1 COMPANY SNAPSHOT
17.1.2 REVENUE ANALYSIS
17.1.3 COMPANY SHARE ANALYSIS
17.1.4 PRODUCT PORTFOLIO
17.1.5 RECENT DEVELOPMENTS
17.2 BOSTON SCIENTIFIC CORPORATION
17.2.1 COMPANY SNAPSHOT
17.2.2 REVENUE ANALYSIS
17.2.3 COMPANY SHARE ANALYSIS
17.2.4 PRODUCT PORTFOLIO
17.2.5 RECENT DEVELOPMENTS
17.3 VARIAN MEDICAL SYSTEMS, INC.
17.3.1 COMPANY SNAPSHOT
17.3.2 COMPANY SHARE ANALYSIS
17.3.3 PRODUCT PORTFOLIO
17.3.4 RECENT DEVELOPMENT
17.4 STRYKER
17.4.1 COMPANY SNAPSHOT
17.4.2 REVENUE ANALYSIS
17.4.3 COMPANY SHARE ANALYSIS
17.4.4 PRODUCT PORTFOLIO
17.4.5 RECENT DEVELOPMENT
17.5 KONINKLIJKE PHILIPS N.V.
17.5.1 COMPANY SNAPSHOT
17.5.2 REVENUE ANALYSIS
17.5.3 COMPANY SHARE ANALYSIS
17.5.4 PRODUCT PORTFOLIO
17.5.5 RECENT DEVELOPMENT
17.6 ABBOTT
17.6.1 COMPANY SNAPSHOT
17.6.2 REVENUE ANALYSIS
17.6.3 PRODUCT PORTFOLIO
17.6.4 RECENT DEVELOPMENT
17.7 ALMA LASERS
17.7.1 COMPANY SNAPSHOT
17.7.2 PRODUCT PORTFOLIO
17.7.3 RECENT DEVELOPMENT
17.8 ANGIODYNAMICS
17.8.1 COMPANY SNAPSHOT
17.8.2 REVENUE ANALYSIS
17.8.3 PRODUCT PORTFOLIO
17.8.4 RECENT DEVELOPMENT
17.9 B.BRAUN MELSUNGEN AG
17.9.1 COMPANY SNAPSHOT
17.9.2 REVENUE ANALYSIS
17.9.3 PRODUCT PORTFOLIO
17.9.4 RECENT DEVELOPMENTS
17.1 BAYLIS MEDICAL COMPANY, INC
17.10.1 COMPANY SNAPSHOT
17.10.2 PRODUCT PORTFOLIO
17.10.3 RECENT DEVELOPMENT
17.11 BD
17.11.1 COMPANY SNAPSHOT
17.11.2 REVENUE ANALYSIS
17.11.3 PRODUCT PORTFOLIO
17.11.4 RECENT DEVELOPMENT
17.12 CANDELA MEDICAL
17.12.1 COMPANY SNAPSHOT
17.12.2 PRODUCT PORTFOLIO
17.12.3 RECENT DEVELOPMENT
17.13 CARL ZEISS MEDITEC AG
17.13.1 COMPANYSNAPSHOT
17.13.2 REVENUE ANALYSIS
17.13.3 PRODUCT PORTFOLIO
17.13.4 RECENT DEVELOPMENT
17.14 COOK
17.14.1 COMPANY SNAPSHOT
17.14.2 PRODUCT PORTFOLIO
17.14.3 RECENT DEVELOPMENTS
17.15 IMRICOR
17.15.1 COMPANY SNAPSHOT
17.15.2 REVENUE ANALYSIS
17.15.3 PRODUCT PORTFOLIO
17.15.4 RECENT DEVELOPMENT
17.16 OLYMPUS CORPORATION
17.16.1 COMPANY SNAPSHOT
17.16.2 REVENUE ANALYSIS
17.16.3 PRODUCT PORTFOILIO
17.16.4 RECENT DEVELOPMENT
17.17 OPTIMED MEDIZINISCHE INSTRUMENTE GMBH
17.17.1 COMPANY SNAPSHOT
17.17.2 PRODUCT PORTFOLIO
17.17.3 RECENT DEVELOPMENTS
17.18 PLUSMEDICA.DE
17.18.1 COMPANY SNAPSHOT
17.18.2 PRODUCT PORTFOLIO
17.18.3 RECENT DEVELOPMENT
17.19 SCITECH
17.19.1 COMPANY SNAPSHOT
17.19.2 PRODUCT PORTFOLIO
17.19.3 RECENT DEVELOPMENTS
17.2 SMITH + NEPHEW
17.20.1 COMPANY SNAPSHOT
17.20.2 REVENUE ANALYSIS
17.20.3 PRODUCT PORTFOLIO
17.20.4 RECENT DEVELOPMENT
17.21 SONABLATE
17.21.1 COMPANY SNAPSHOT
17.21.2 PRODUCT PORTFOLIO
17.21.3 RECENT DEVELOPMENT
17.22 THERACLION
17.22.1 COMPANY SNAPSHOT
17.22.2 REVENUE ANALYSIS
17.22.3 PRODUCT PORTFOLIO
17.22.4 RECENT DEVELOPMENT
17.23 TELEFLEX INCORPORATED
17.23.1 COMPANY SNAPSHOT
17.23.2 REVENUE ANALYSIS
17.23.3 PRODUCT PORTFOLIO
17.23.4 RECENT DEVELOPMENTS
17.24 TERUMO CORPORATION
17.24.1 COMPANY SNAPSHOT
17.24.2 REVENUE ANALYSIS
17.24.3 PRODUCT PORTFOLIO
17.24.4 RECENT DEVELOPMENTS
18 QUESTIONNAIRE
19 RELATED REPORTS
表のリスト
TABLE 1 EUROPE VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 2 EUROPE SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 3 EUROPE SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 4 EUROPE SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 5 EUROPE ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 6 EUROPE ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 7 EUROPE THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 8 EUROPE RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 9 EUROPE LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 10 EUROPE ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 11 EUROPE RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 12 EUROPE ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 13 EUROPE NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 14 EUROPE CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 15 EUROPE VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 16 EUROPE VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 17 EUROPE VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 18 EUROPE VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 19 EUROPE VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 20 EUROPE ILIAC VEIN STENT TECHNOLOGY IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 21 EUROPE MEDICATION IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 22 EUROPE OTHERS IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 23 EUROPE VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 24 EUROPE VARICOSE VEINS IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 25 EUROPE DEEP VEIN THROMBOSIS (DVT) IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 26 EUROPE PULMONARY EMBOLISM IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 27 EUROPE CHRONIC VENOUS INSUFFICIENCY (CVI) IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 28 EUROPE SUPERFICIAL THROMBOPHLEBITIS IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 29 EUROPE OTHERS IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 30 EUROPE VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 31 EUROPE SCELEROTHERAPY IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 32 EUROPE RADIOFREQUENCY ABLATION THERAPY IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 33 EUROPE LASER TREATMENT IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 34 EUROPE AMBULATORY PHELEBECTOMY IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 35 EUROPE VEIN LIGATION AND STRIPPING IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 36 EUROPE ANGIOPLASTY OR STENTING IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 37 EUROPE SURGERIES IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 38 EUROPE COMPRESSION THERAPY IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 39 EUROPE VEINACTIVE MEDICATION IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 40 EUROPE VENA CAVA FILTER IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 41 EUROPE OTHER THERAPIES IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 42 EUROPE VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 43 EUROPE HOSPITALS IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 44 EUROPE CLINICS IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 45 EUROPE AMBULATORY SURGICAL CENTERS IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 46 EUROPE OTHERS IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 47 EUROPE VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 48 EUROPE DIRECT TENDER IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 49 EUROPE RETAIL SALES IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 50 EUROPE OTHERS IN VENOUS DISEASES TREATMENT MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 51 EUROPE VENOUS DISEASES TREATMENT MARKET, BY COUNTRY, 2021-2030 (USD MILLION)
TABLE 52 EUROPE VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 53 EUROPE SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 54 EUROPE SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 55 EUROPE ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 56 EUROPE THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 57 EUROPE RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 58 EUROPE LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 59 EUROPE ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 60 EUROPE RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 61 EUROPE ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 62 EUROPE NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 63 EUROPE CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 64 EUROPE VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 65 EUROPE VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 66 EUROPE VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 67 EUROPE VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 68 EUROPE VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 69 EUROPE VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 70 EUROPE VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 71 EUROPE VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 72 GERMANY VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 73 GERMANY SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 74 GERMANY SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 75 GERMANY ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 76 GERMANY THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 77 GERMANY RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 78 GERMANY LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 79 GERMANY ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 80 GERMANY RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 81 GERMANY ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 82 GERMANY NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 83 GERMANY CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 84 GERMANY VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 85 GERMANY VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 86 GERMANY VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 87 GERMANY VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 88 GERMANY VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 89 GERMANY VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 90 GERMANY VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 91 GERMANY VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 92 U.K. VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 93 U.K. SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 94 U.K. SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 95 U.K. ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 96 U.K. THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 97 U.K. RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 98 U.K. LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 99 U.K. ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 100 U.K. RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 101 U.K. ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 102 U.K. NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 103 U.K. CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 104 U.K. VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 105 U.K. VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 106 U.K. VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 107 U.K. VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 108 U.K. VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 109 U.K. VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 110 U.K. VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 111 U.K. VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 112 FRANCE VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 113 FRANCE SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 114 FRANCE SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 115 FRANCE ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 116 FRANCE THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 117 FRANCE RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 118 FRANCE LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 119 FRANCE ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 120 FRANCE RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 121 FRANCE ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 122 FRANCE NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 123 FRANCE CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 124 FRANCE VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 125 FRANCE VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 126 FRANCE VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 127 FRANCE VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 128 FRANCE VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 129 FRANCE VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 130 FRANCE VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 131 FRANCE VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 132 ITALY VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 133 ITALY SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 134 SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 135 ITALY ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 136 ITALY THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 137 ITALY RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 138 ITALY LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 139 ITALY ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 140 ITALY RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 141 ITALY ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 142 ITALY NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 143 ITALY CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 144 ITALY VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 145 ITALY VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 146 ITALY VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 147 ITALY VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 148 ITALY VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 149 ITALY VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 150 ITALY VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 151 ITALY VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 152 RUSSIA VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 153 RUSSIA SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 154 RUSSIA SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 155 RUSSIA ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 156 RUSSIA THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 157 RUSSIA RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 158 RUSSIA LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 159 RUSSIA ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 160 RUSSIA RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 161 RUSSIA ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 162 RUSSIA NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 163 RUSSIA CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 164 RUSSIA VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 165 RUSSIA VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 166 RUSSIA VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 167 RUSSIA VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 168 RUSSIA VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 169 RUSSIA VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 170 RUSSIA VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 171 RUSSIA VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 172 SPAIN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 173 SPAIN SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 174 SPAIN SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 175 SPAIN ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 176 SPAIN THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 177 SPAIN RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 178 SPAIN LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 179 SPAIN ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 180 SPAIN RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 181 SPAIN ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 182 SPAIN NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 183 SPAIN CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 184 SPAIN VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 185 SPAIN VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 186 SPAIN VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 187 SPAIN VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 188 SPAIN VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 189 SPAIN VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 190 SPAIN VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 191 SPAIN VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 192 TURKEY VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 193 TURKEY SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 194 TURKEY SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 195 TURKEY ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 196 TURKEY THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 197 TURKEY RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 198 TURKEY LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 199 TURKEY ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 200 TURKEY RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 201 TURKEY ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 202 TURKEY NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 203 TURKEY CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 204 TURKEY VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 205 TURKEY VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 206 TURKEY VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 207 TURKEY VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 208 TURKEY VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 209 TURKEY VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 210 TURKEY VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 211 TURKEY VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 212 NETHERLANDS VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 213 NETHERLANDS SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 214 NETHERLANDS SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 215 NETHERLANDS ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 216 NETHERLANDS THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 217 NETHERLANDS RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 218 NETHERLANDS LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 219 NETHERLANDS ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 220 NETHERLANDS RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 221 NETHERLANDS ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 222 NETHERLANDS NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 223 NETHERLANDS CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 224 NETHERLANDS VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 225 NETHERLANDS VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 226 NETHERLANDS VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 227 NETHERLANDS VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 228 NETHERLANDS VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 229 NETHERLANDS VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 230 NETHERLANDS VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 231 NETHERLANDS VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 232 BELGIUM VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 233 BELGIUM SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 234 BELGIUM SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 235 BELGIUM ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 236 BELGIUM THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 237 BELGIUM RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 238 BELGIUM LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 239 BELGIUM ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 240 BELGIUM RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 241 BELGIUM ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 242 BELGIUM NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 243 BELGIUM CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 244 BELGIUM VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 245 BELGIUM VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 246 BELGIUM VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 247 BELGIUM VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 248 BELGIUM VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 249 BELGIUM VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 250 BELGIUM VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 251 BELGIUM VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 252 SWITZERLAND VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 253 SWITZERLAND SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 254 SWITZERLAND SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 255 SWITZERLAND ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 256 SWITZERLAND THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 257 SWITZERLAND RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 258 SWITZERLAND LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 259 SWITZERLAND ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 260 SWITZERLAND RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 261 SWITZERLAND ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 262 SWITZERLAND NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 263 SWITZERLAND CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 264 SWITZERLAND VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 265 SWITZERLAND VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 266 SWITZERLAND VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 267 SWITZERLAND VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 268 SWITZERLAND VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 269 SWITZERLAND VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 270 SWITZERLAND VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 271 SWITZERLAND VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 272 IRELAND VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 273 IRELAND SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 274 IRELAND SCLEROTHERAPY INJECTION IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 275 IRELAND ABLATION DEVICES IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 276 IRELAND THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 277 IRELAND RADIOFREQUENCY IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 278 IRELAND LIGHT IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 279 IRELAND ULTRASOUND IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 280 IRELAND RADIATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 281 IRELAND ELECTRICAL IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 282 IRELAND NON-THERMAL ABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 283 IRELAND CRYOABLATION IN VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 284 IRELAND VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY PROCEDURE, 2021-2030 (USD MILLION)
TABLE 285 IRELAND VENOUS CLOSURE PRODUCTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 286 IRELAND VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY TECHNOLOGY, 2021-2030 (USD MILLION)
TABLE 287 IRELAND VENOUS STENTS IN VENOUS DISEASES TREATMENT MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 288 IRELAND VENOUS DISEASES TREATMENT MARKET, BY DISEASE TYPE, 2021-2030 (USD MILLION)
TABLE 289 IRELAND VENOUS DISEASES TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD MILLION)
TABLE 290 IRELAND VENOUS DISEASES TREATMENT MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 291 IRELAND VENOUS DISEASES TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 292 REST OF EUROPE VENOUS DISEASES TREATMENT MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
図表一覧
FIGURE 1 EUROPE VENOUS DISEASES TREATMENT MARKET: SEGMENTATION
FIGURE 2 EUROPE VENOUS DISEASES TREATMENT MARKET: DATA TRIANGULATION
FIGURE 3 EUROPE VENOUS DISEASES TREATMENT MARKET: DROC ANALYSIS
FIGURE 4 EUROPE VENOUS DISEASES TREATMENT MARKET: EUROPE VS REGIONAL MARKET ANALYSIS
FIGURE 5 EUROPE VENOUS DISEASES TREATMENT MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 EUROPE VENOUS DISEASES TREATMENT MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 EUROPE VENOUS DISEASES TREATMENT MARKET: MARKET END USER COVERAGE GRID
FIGURE 8 EUROPE VENOUS DISEASES TREATMENT MARKET: DBMR MARKET POSITION GRID
FIGURE 9 EUROPE VENOUS DISEASES TREATMENT MARKET: VENDOR SHARE ANALYSIS
FIGURE 10 EUROPE VENOUS DISEASES TREATMENT MARKET: SEGMENTATION
FIGURE 11 GROWING AWARENESS ABOUT VENOUS DISEASES TREATMENTS AND INCREASING HEALTHCARE EXPENDITURE IS EXPECTED TO DRIVE THE GROWTH OF THE EUROPE VENOUS DISEASES TREATMENT MARKET FROM 2023 TO 2030
FIGURE 12 THE ABLATION DEVICES SEGMENT IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE EUROPE VENOUS DISEASES TREATMENT MARKET IN 2023 & 2030
FIGURE 13 DRIVERS, RESTRAINTS, OPPORTUNITIES AND CHALLENGES OF THE EUROPE VENOUS DISEASES TREATMENT MARKET
FIGURE 14 EUROPE VENOUS DISEASES TREATMENT MARKET : BY PRODUCT TYPE, 2022
FIGURE 15 EUROPE VENOUS DISEASES TREATMENT MARKET : BY PRODUCT TYPE, 2023-2030 (USD MILLION)
FIGURE 16 EUROPE VENOUS DISEASES TREATMENT MARKET : BY PRODUCT TYPE, CAGR (2023-2030)
FIGURE 17 EUROPE VENOUS DISEASES TREATMENT MARKET : BY PRODUCT TYPE, LIFELINE CURVE
FIGURE 18 EUROPE VENOUS DISEASES TREATMENT MARKET: BY DISEASE TYPE, 2022
FIGURE 19 EUROPE VENOUS DISEASES TREATMENT MARKET: BY DISEASE TYPE, 2023-2030 (USD MILLION)
FIGURE 20 EUROPE VENOUS DISEASES TREATMENT MARKET: BY DISEASE TYPE, CAGR (2023-2030)
FIGURE 21 EUROPE VENOUS DISEASES TREATMENT MARKET: BY DISEASE TYPE, LIFELINE CURVE
FIGURE 22 EUROPE VENOUS DISEASES TREATMENT MARKET: BY TREATMENT TYPE, 2022
FIGURE 23 EUROPE VENOUS DISEASES TREATMENT MARKET: BY TREATMENT TYPE, 2023-2030 (USD MILLION)
FIGURE 24 EUROPE VENOUS DISEASES TREATMENT MARKET: BY TREATMENT TYPE, CAGR (2023-2030)
FIGURE 25 EUROPE VENOUS DISEASES TREATMENT MARKET: BY TREATMENT TYPE, LIFELINE CURVE
FIGURE 26 EUROPE VENOUS DISEASES TREATMENT MARKET: BY END USER, 2022
FIGURE 27 EUROPE VENOUS DISEASES TREATMENT MARKET: BY END USER, 2023-2030 (USD MILLION)
FIGURE 28 EUROPE VENOUS DISEASES TREATMENT MARKET: BY END USER, CAGR (2023-2030)
FIGURE 29 EUROPE VENOUS DISEASES TREATMENT MARKET: BY END USER, LIFELINE CURVE
FIGURE 30 EUROPE VENOUS DISEASES TREATMENT MARKET : BY DISTRIBUTION CHANNEL, 2022
FIGURE 31 EUROPE VENOUS DISEASES TREATMENT MARKET : BY DISTRIBUTION CHANNEL, 2023-2030 (USD MILLION)
FIGURE 32 EUROPE VENOUS DISEASES TREATMENT MARKET : BY DISTRIBUTION CHANNEL, CAGR (2023-2030)
FIGURE 33 EUROPE VENOUS DISEASES TREATMENT MARKET : BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 34 EUROPE VENOUS DISEASES TREATMENT MARKET: SNAPSHOT (2022)
FIGURE 35 EUROPE VENOUS DISEASES TREATMENT MARKET: BY COUNTRY (2022)
FIGURE 36 EUROPE VENOUS DISEASES TREATMENT MARKET: BY COUNTRY (2023 & 2030)
FIGURE 37 EUROPE VENOUS DISEASES TREATMENT MARKET: BY COUNTRY (2022 & 2030)
FIGURE 38 EUROPE VENOUS DISEASES TREATMENT MARKET: PRODUCT TYPE (2023-2030)
FIGURE 39 EUROPE VENOUS DISEASES TREATMENT MARKET: COMPANY SHARE 2022 (%)

調査方法
データ収集と基準年分析は、大規模なサンプル サイズのデータ収集モジュールを使用して行われます。この段階では、さまざまなソースと戦略を通じて市場情報または関連データを取得します。過去に取得したすべてのデータを事前に調査および計画することも含まれます。また、さまざまな情報ソース間で見られる情報の不一致の調査も含まれます。市場データは、市場統計モデルと一貫性モデルを使用して分析および推定されます。また、市場シェア分析と主要トレンド分析は、市場レポートの主要な成功要因です。詳細については、アナリストへの電話をリクエストするか、お問い合わせをドロップダウンしてください。
DBMR 調査チームが使用する主要な調査方法は、データ マイニング、データ変数が市場に与える影響の分析、および一次 (業界の専門家) 検証を含むデータ三角測量です。データ モデルには、ベンダー ポジショニング グリッド、市場タイムライン分析、市場概要とガイド、企業ポジショニング グリッド、特許分析、価格分析、企業市場シェア分析、測定基準、グローバルと地域、ベンダー シェア分析が含まれます。調査方法について詳しくは、お問い合わせフォームから当社の業界専門家にご相談ください。
カスタマイズ可能
Data Bridge Market Research は、高度な形成的調査のリーダーです。当社は、既存および新規のお客様に、お客様の目標に合致し、それに適したデータと分析を提供することに誇りを持っています。レポートは、対象ブランドの価格動向分析、追加国の市場理解 (国のリストをお問い合わせください)、臨床試験結果データ、文献レビュー、リファービッシュ市場および製品ベース分析を含めるようにカスタマイズできます。対象競合他社の市場分析は、技術ベースの分析から市場ポートフォリオ戦略まで分析できます。必要な競合他社のデータを、必要な形式とデータ スタイルでいくつでも追加できます。当社のアナリスト チームは、粗い生の Excel ファイル ピボット テーブル (ファクト ブック) でデータを提供したり、レポートで利用可能なデータ セットからプレゼンテーションを作成するお手伝いをしたりすることもできます。