North America Prophylaxis Of Organ Rejection Market
Tamaño del mercado en miles de millones de dólares
Tasa de crecimiento anual compuesta (CAGR) :
% 
 USD
1,092.43 Million
USD
1,560.26 Million
2021
2029
USD
1,092.43 Million
USD
1,560.26 Million
2021
2029
| 2022 –2029 | |
| USD 1,092.43 Million | |
| USD 1,560.26 Million | |
|
|
|
Mercado de profilaxis del rechazo de órganos en América del Norte, por causa (exposición epidemiológica, profilaxis antibacteriana, profilaxis contra otros patógenos y otros), tratamiento (inmunosupresores para pacientes ambulatorios, inmunosupresores para pacientes hospitalizados y otros), vía de administración (oral, intravenosa), órgano (riñón, hígado, corazón, pulmón, otros), tipo de paciente (pediatría, adultos), usuario final (hospitales, atención médica domiciliaria clínica y otros), canal de distribución (licitación directa, farmacias y otros), país (EE. UU., Canadá, México), tendencias del mercado y pronóstico hasta 2029.
Análisis y perspectivas del mercado : mercado de profilaxis del rechazo de órganos en América del Norte
Se espera que el mercado de profilaxis del rechazo de órganos de América del Norte gane crecimiento de mercado en el período de pronóstico de 2022 a 2029. Data Bridge Market Research analiza que el mercado está creciendo con una CAGR del 4,7% en el período de pronóstico de 2022 a 2029 y se espera que alcance los USD 1.560,26 millones para 2029 desde USD 1.092,43 millones en 2021. Es probable que la creciente prevalencia del trasplante de órganos y el mayor uso de inmunosupresores sean los principales impulsores que impulsan la demanda del mercado en el período de pronóstico.
El término profilaxis significa tratamiento dado o acción tomada para prevenir la enfermedad. La profilaxis del rechazo de órganos, se refiere a la prevención del rechazo de órganos mediante el uso de medicamentos. Los pacientes que se someten a un trasplante, deben mantenerse en un régimen de inmunosupresión para la profilaxis del rechazo para ayudar a asegurar la supervivencia del injerto. El rechazo del trasplante es un proceso, en el que la inmunidad de un receptor de trasplante la profilaxis del rechazo actual implementa el uso de inhibidores de la calcineurina, inhibidores de mTOR, agentes antimetabolitos y corticosteroides. Los agentes de tratamiento mejoran las consecuencias a corto plazo del trasplante de órgano, pero ciertas mejoras. Los pacientes receptores, que se han sometido a un trasplante de órgano sólido, deben tomar medicamentos antirrechazo. Esto se debe a que el sistema inmunológico destruiría el órgano trasplantado.
Los factores que impulsan el crecimiento del mercado de profilaxis del rechazo de órganos en América del Norte son la mayor prevalencia de trasplantes de órganos, el aumento de los procedimientos quirúrgicos, el aumento de la población receptora y los lanzamientos de productos. Sin embargo, los factores que se espera que limiten el mercado son el aumento del costo del procedimiento de trasplante, la falta de conocimiento sobre el trasplante de órganos y los riesgos que conlleva mientras el paciente recibe el órgano trasplantado.
- Por otra parte, las iniciativas estratégicas de los actores del mercado, el aumento de la investigación y el desarrollo y el uso de inmunosupresores pueden actuar como una oportunidad para el crecimiento del mercado de profilaxis del rechazo de órganos en América del Norte. La necesidad de conocimientos especializados y la aprobación regulatoria pueden crear desafíos para el mercado de profilaxis del rechazo de órganos en América del Norte. Hay desarrollos recientes relacionados con el mercado de profilaxis del rechazo de órganos en América del Norte.
El informe de mercado de profilaxis del rechazo de órganos en América del Norte proporciona detalles sobre la participación de mercado, los nuevos desarrollos y el impacto de los actores del mercado nacional y localizado, analiza las oportunidades en términos de nuevos segmentos de ingresos, cambios en las regulaciones del mercado, aprobaciones de productos, decisiones estratégicas, lanzamientos de productos, expansiones geográficas e innovaciones tecnológicas en el mercado. Para comprender el análisis y el escenario del mercado, contáctenos para obtener un resumen analítico; nuestro equipo lo ayudará a crear una solución de impacto en los ingresos para lograr su objetivo deseado.
Alcance y tamaño del mercado de profilaxis del rechazo de órganos en América del Norte
El mercado de profilaxis del rechazo de órganos en América del Norte se clasifica en siete segmentos: causa, tratamiento, vía de administración, órgano, tipo de paciente, usuario final y canal de distribución.
El crecimiento entre segmentos le ayuda a analizar nichos de crecimiento y estrategias para abordar el mercado y determinar sus áreas de aplicación principales y la diferencia en sus mercados objetivo.
- En función de la causa, el mercado de profilaxis del rechazo de órganos de América del Norte se segmenta en exposición epidemiológica, profilaxis antibacteriana, profilaxis contra otros patógenos y otros. En 2022, se espera que el segmento de exposición epidemiológica domine el mercado de profilaxis del rechazo de órganos de América del Norte debido a la disponibilidad de información directa y la presencia de patógenos relacionados con la profilaxis del rechazo de órganos en los EE. UU.
- En función del tratamiento, el mercado de profilaxis del rechazo de órganos en América del Norte se segmenta en inmunosupresores para pacientes ambulatorios, inmunosupresores para pacientes hospitalizados y otros. En 2022, se espera que el segmento de inmunosupresores para pacientes ambulatorios domine el mercado de profilaxis del rechazo de órganos en América del Norte, ya que puede lograr una respuesta inmunitaria sostenida y específica contra las células dañadas y la disponibilidad de ciclosporina en los EE. UU. y Canadá.
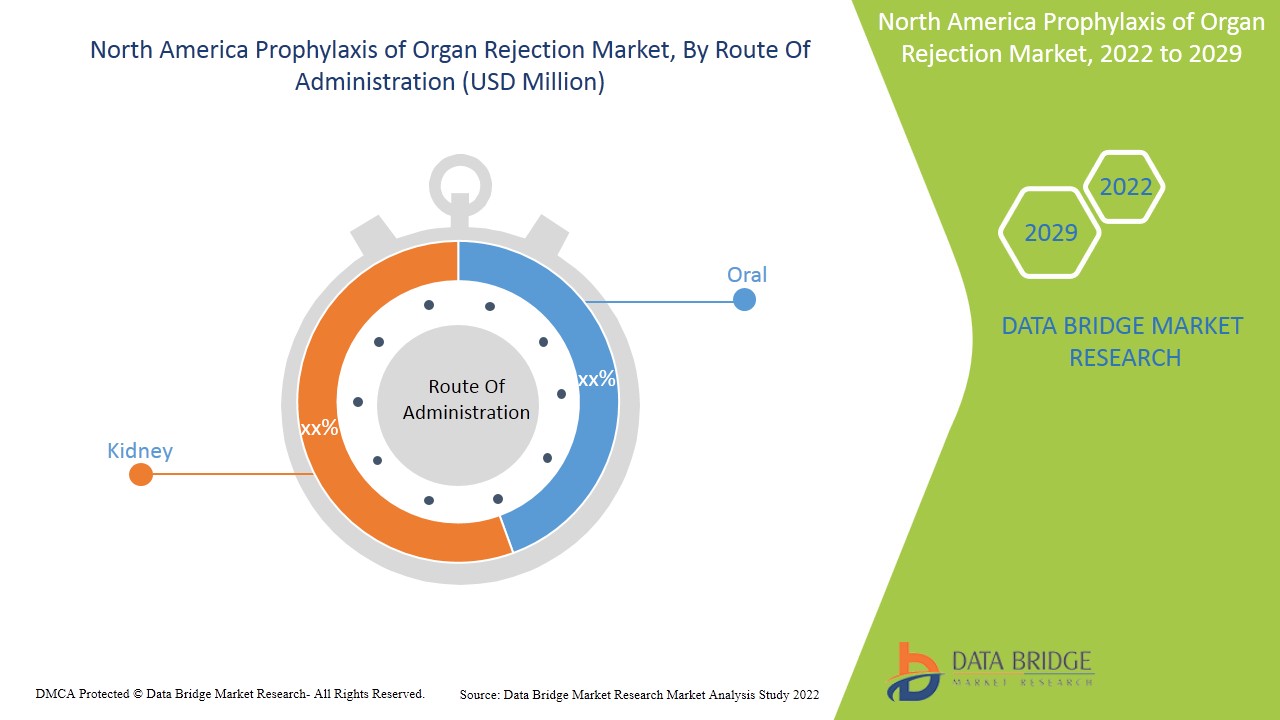
- Según la vía de administración, el mercado de profilaxis del rechazo de órganos de América del Norte se segmenta en oral e intravenosa . En 2022, se espera que el segmento oral domine la profilaxis del rechazo de órganos de América del Norte debido a la mayor disponibilidad y consumo de comprimidos y cápsulas orales.
- En función del órgano, el mercado de profilaxis del rechazo de órganos en América del Norte se segmenta en riñón, hígado, corazón, pulmón y otros. En 2022, se espera que el segmento renal domine la profilaxis del rechazo de órganos en América del Norte debido a la mayor incidencia de insuficiencia renal y la presencia de políticas de reembolso como Medicare, para el seguro del receptor.
- En función del tipo de paciente, el mercado de profilaxis del rechazo de órganos de América del Norte se segmenta en pediátrico y adulto. En 2022, se espera que el segmento de adultos domine el mercado de profilaxis del rechazo de órganos de América del Norte, debido a la mayor prevalencia de enfermedades crónicas y afecciones genéticas de los pacientes adultos, el sistema inmunológico débil en los adultos y la lista de espera de receptores adultos más larga que la lista de espera de niños y mujeres en Nuevo México y los EE. UU.
- En función del usuario final, el mercado de profilaxis del rechazo de órganos de América del Norte se segmenta en hospitales, clínicas, atención médica domiciliaria y otros. En 2022, se espera que el segmento de hospitales domine el mercado de profilaxis del rechazo de órganos de América del Norte debido al aumento de la atención médica y se prevé que la colaboración con otros hospitales de América del Norte para la entrega de órganos de donantes a los pacientes receptores domine el mercado.
- Sobre la base del canal de distribución, el mercado de profilaxis del rechazo de órganos de América del Norte se segmenta en licitaciones directas, farmacias y otros. En 2022, se espera que el segmento de licitaciones directas domine el mercado de profilaxis del rechazo de órganos de América del Norte debido al aumento de la demanda de inmunosupresores ambulatorios por parte de las compañías farmacéuticas y se prevé que el pago garantizado domine el mercado.
Análisis a nivel de país del mercado de profilaxis del rechazo de órganos en América del Norte
El mercado de profilaxis del rechazo de órganos en América del Norte se clasifica en siete segmentos: causa, tratamiento, vía de administración, órgano, tipo de paciente, usuario final y canal de distribución.
Los países cubiertos en el informe del mercado de profilaxis del rechazo de órganos en América del Norte son Estados Unidos, Canadá y México.
- En 2021, Estados Unidos domina debido a la presencia del mayor mercado de consumo con un alto PIB. Además, Estados Unidos tiene el mayor gasto familiar del mundo y ofrece acuerdos comerciales con varios países, lo que lo convierte en el mayor mercado de productos de consumo, incluidos los productos para la profilaxis del rechazo de órganos, debido a la presencia de importantes actores del mercado y al mayor avance tecnológico en la región.
La sección de países del informe también proporciona factores de impacto individuales en el mercado y cambios en la regulación en el mercado a nivel nacional que afectan las tendencias actuales y futuras del mercado. Los puntos de datos como nuevas ventas, ventas de reemplazo, demografía del país, leyes regulatorias y aranceles de importación y exportación son algunos de los principales indicadores utilizados para pronosticar el escenario del mercado para países individuales. Además, se considera la presencia y disponibilidad de marcas de América del Norte y los desafíos que enfrentan debido a la competencia grande o escasa de las marcas locales y nacionales, el impacto de los canales de venta al proporcionar un análisis de pronóstico de los datos del país.
El potencial de crecimiento de la profilaxis del rechazo de órganos en las economías emergentes y las iniciativas estratégicas de los actores del mercado están creando nuevas oportunidades en el mercado de profilaxis del rechazo de órganos en América del Norte
El mercado de profilaxis del rechazo de órganos de América del Norte también le proporciona un análisis detallado del mercado para el crecimiento de cada país en una industria en particular con las ventas de profilaxis del rechazo de órganos. El impacto del avance en la profilaxis del rechazo de órganos y los cambios en los escenarios regulatorios con su apoyo al mercado de profilaxis del rechazo de órganos. Los datos están disponibles para el período histórico de 2020 a 2021.
Análisis del panorama competitivo y de la cuota de mercado de la profilaxis del rechazo de órganos en América del Norte
El panorama competitivo del mercado de profilaxis del rechazo de órganos en América del Norte proporciona detalles por competidor. Los detalles incluidos son una descripción general de la empresa, las finanzas de la empresa, los ingresos generados, el potencial de mercado, la inversión en investigación y desarrollo, las nuevas iniciativas de mercado, los sitios e instalaciones de producción, las fortalezas y debilidades de la empresa, el lanzamiento de productos, las líneas de prueba de productos, las aprobaciones de productos, las patentes, la amplitud y amplitud de los productos, el dominio de las aplicaciones y la curva de la línea de vida de la tecnología. Los puntos de datos anteriores proporcionados solo están relacionados con el enfoque de la empresa relacionado con el mercado de profilaxis del rechazo de órganos en América del Norte.
Las principales empresas que brindan profilaxis para el rechazo de órganos son Ossium Health, Inc, Altavant Sciences, Inc, Concord Biotech, WOCKHARDT, SEBELA PHARMACEUTICALS, Veloxis Pharmaceuticals, Inc, Astellas Pharma Inc, Novartis AG, Bristol-Myers Squibb Company, Dr Reddy's Laboratories, Ltd, Viatris Inc, Strides Pharma Science Limited, Glenmark, Biocon, Pfizer Inc, Mayne Pharma Group Limited, Hikma Pharmaceuticals PLC, AbbVie Inc, Apotex Inc, Teva Pharmaceutical USA Inc, Amneal Pharmaceuticals LLC, Zydus Pharmaceuticals, Inc., Novitium Pharma, ZHEJIANG HISUN PHARMACEUTICAL Co., LTD., CSL Behring, entre otras.
Los analistas de DBMR comprenden las fortalezas competitivas y brindan un análisis competitivo para cada competidor por separado.
Las iniciativas estratégicas de los actores del mercado junto con los nuevos avances tecnológicos para la profilaxis del rechazo de órganos en América del Norte están cerrando la brecha en el uso de medicamentos para evitar el rechazo de órganos.
Por ejemplo,
- En enero de 2020, Altavant Sciences, Inc. adquirió Onspira Therapeutics. Altavant Sciences Inc. había desarrollado OSP-101 para el tratamiento del síndrome de bronquiolitis obliterante, la principal causa de morbilidad y mortalidad en pacientes con trasplante de pulmón. La adquisición ampliaría la cartera de productos e incluiría OSP-101, para su aprobación posterior a la comercialización.
La colaboración, las empresas conjuntas y otras estrategias por parte de los actores del mercado están mejorando el mercado de la empresa en el mercado de profilaxis del rechazo de órganos, lo que también proporciona el beneficio para que la organización mejore su oferta para el mercado de profilaxis del rechazo de órganos de América del Norte.
SKU-
Obtenga acceso en línea al informe sobre la primera nube de inteligencia de mercado del mundo
- Panel de análisis de datos interactivo
- Panel de análisis de empresas para oportunidades con alto potencial de crecimiento
- Acceso de analista de investigación para personalización y consultas
- Análisis de la competencia con panel interactivo
- Últimas noticias, actualizaciones y análisis de tendencias
- Aproveche el poder del análisis de referencia para un seguimiento integral de la competencia
Tabla de contenido
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATIONS
1.6 MARKETS COVERED
2 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 DBMR TRIPOD DATA VALIDATION MODEL
2.5 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.6 MULTIVARIATE MODELLING
2.7 BY CAUSE SEGMENT LIFELINE CURVE
2.8 DBMR MARKET POSITION GRID
2.9 VENDOR SHARE ANALYSIS
2.1 MARKET END USER COVERAGE GRID
2.11 SECONDARY SOURCES
3 EXECUTIVE SUMMARY
4 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION: EPIDEMIOLOGY SNAPSHOT
5 MARKET OVERVIEW
5.1 DRIVERS
5.1.1 RISE IN PREVALENCE OF ORGAN TRANSPLANTATIONS
5.1.2 RISE IN CHRONIC DISEASES
5.1.3 RISE IN CLINICAL TRIALS
5.1.4 RISE IN RECIPIENT PATIENTS
5.1.5 PRODUCT APPROVALS
5.2 RESTRAINTS
5.2.1 RISE IN COST OF TRANSPLANTATION PROCEDURE
5.2.2 LACK OF AWARENESS ABOUT ORGAN TRANSPLANTATION
5.2.3 RISKS INCLUDED WITH THE PATIENT RECEIVES TRANSPLANTED ORGAN
5.2.4 ETHICAL ISSUES RELATED TO ORGAN TRANSPLANTATION
5.3 OPPORTUNITIES
5.3.1 RISE IN RESEARCH AND DEVELOPMENTS
5.3.2 STRATEGIC INITIATIVES BY MARKET PLAYERS
5.3.3 RISE IN USE OF IMMUNOSUPPRESSANT
5.4 CHALLENGES
5.4.1 SHORTAGE OF SKILLED PROFESSIONALS REQUIRED FOR THE USE OF PROPHYLAXIS OF ORGAN REJECTION
5.4.2 STRINGENT REGULATIONS
6 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY CAUSE
6.1 OVERVIEW
6.2 EPIDEMIOLOGIC EXPOSURE
6.2.1 HOSPITAL EXPOSURE
6.2.2 COMMUNITY EXPOSURE
6.3 ANTIBACTERIAL PROPHYLAXIS
6.3.1 POST-TRANSPLANT PROPHYLAXIS
6.3.2 PERIOPERATIVE ANTIBACTERIAL PROPHYLAXIS
6.4 PROPHYLAXIS AGAINST OTHER PATHOGENS
6.5 OTHERS
7 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT
7.1 OVERVIEW
7.2 OUTPATIENT IMMUNOSUPPRESSANT
7.2.1 CALCINEURIN INHIBITORS
7.2.1.1 CYCLOSPORINE
7.2.1.2 TACROLIMUS
7.2.1.2.1 IMMEDIATE RELEASE
7.2.1.2.2 EXTENDED RELEASE
7.2.2 CORTICOSTEROIDS
7.2.2.1 METHYLPREDNISOLONE
7.2.2.2 PREDNISONE
7.2.2.3 OTHERS
7.2.3 MYCOPHENOLIC ACIDS
7.2.3.1 MYCOPHENOLATE MOFETIL
7.2.3.2 MYCOPHENOLATE SODIUM
7.2.4 MTOR INHIBITORS
7.2.4.1 SIROLIMUS
7.2.4.2 EVEROLIMUS
7.2.5 AZATHIOPRINE
7.2.6 OTHERS
7.3 INPATIENT IMMUNOSUPPRESSANT
7.3.1 BELATACEPT
7.3.2 BASILIXIMAB
7.3.3 OTHERS
7.4 OTHERS
7.4.1 ERYTHROPOIESIS-STIMULATING AGENTS
7.4.1.1 ERYTHROPOIETIN
7.4.1.2 DARBEPOETIN
7.4.2 ANTI-VIRAL AGENTS
7.4.2.1 VALGANCICLOVIR
7.4.2.2 LAMIVUDINE
7.4.2.3 ADEFOVIR
7.4.2.4 OTHERS
7.4.3 OTHERS
8 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY ROUTE OF ADMINISTRATION
8.1 OVERVIEW
8.2 ORAL
8.2.1 TABLET
8.2.1.1 IMMEDIATE RELEASE
8.2.1.2 EXTENDED-RELEASE
8.2.2 ORAL SOLUTION
8.2.3 OTHERS
8.3 INTRAVENOUS
9 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY ORGAN
9.1 OVERVIEW
9.2 KIDNEY
9.3 LIVER
9.4 HEART
9.5 LUNG
9.6 OTHERS
10 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY PATIENT TYPE
10.1 OVERVIEW
10.2 ADULTS
10.3 PEDIATRIC
11 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY END USER
11.1 OVERVIEW
11.2 HOSPITALS
11.3 CLINICS
11.4 HOME HEALTHCARE
11.5 OTHERS
12 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY DISTRIBUTION CHANNEL
12.1 OVERVIEW
12.2 DIRECT TENDERS
12.3 PHARMACY STORES
12.3.1 HOSPITAL PHARMACY
12.3.2 RETAIL PHARMACY
12.3.3 ONLINE PHARMACY
12.4 OTHERS
13 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION
13.1 NORTH AMERICA
13.1.1 U.S.
13.1.2 CANADA
13.1.3 MEXICO
14 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: COMPANY LANDSCAPE
14.1 COMPANY SHARE ANALYSIS: NORTH AMERICA
15 QUESTIONNAIRE
16 RELATED REPORTS
Lista de Tablas
TABLE 1 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 2 NORTH AMERICA EPIDEMIOLOGIC EXPOSURE IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 3 NORTH AMERICA EPIDEMIOLOGIC EXPOSURE IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 4 NORTH AMERICA ANTIBACTERIAL PROPHYLAXIS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 5 NORTH AMERICA ANTIBACTERIAL PROPHYLAXIS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 6 NORTH AMERICA PROPHYLAXIS AGAINST OTHER PATHOGENS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 7 NORTH AMERICA OTHERS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 8 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 9 NORTH AMERICA OUTPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 10 NORTH AMERICA OUTPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 11 NORTH AMERICA CALCINEURIN INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 12 NORTH AMERICA TACROLIMUS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 13 NORTH AMERICA CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 14 NORTH AMERICA MYCOPHENOLIC ACIDS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 15 NORTH AMERICA MTOR INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 16 NORTH AMERICA INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 17 NORTH AMERICA INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 18 NORTH AMERICA OTHERS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 19 NORTH AMERICA OTHERS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 20 NORTH AMERICA ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 21 NORTH AMERICA ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 22 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 23 NORTH AMERICA ORAL IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 24 NORTH AMERICA ORAL IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 25 NORTH AMERICA TABLET IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 26 NORTH AMERICA INTRAVENOUS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 27 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY ORGAN, 2020-2029 (USD MILLION)
TABLE 28 NORTH AMERICA KIDNEY IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 29 NORTH AMERICA LIVER IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 30 NORTH AMERICA HEART IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 31 NORTH AMERICA LUNG IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 32 NORTH AMERICA OTHERS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 33 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 34 NORTH AMERICA ADULTS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 35 NORTH AMERICA PEDIATRIC IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 36 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 37 NORTH AMERICA HOSPITALS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 38 NORTH AMERICA CLINICS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 39 NORTH AMERICA HOME HEALTHCARE IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 40 NORTH AMERICA OTHERS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 41 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 42 NORTH AMERICA DIRECT TENDERS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 43 NORTH AMERICA PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 44 NORTH AMERICA PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 45 NORTH AMERICA OTHERS IN PROPHYLAXIS OF ORGAN REJECTION MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 46 NORTH AMERICA PROPHYLAXIS OF ORGAN TRANSPLANT MARKET, BY COUNTRY, 2020-2029 (USD MILLION)
TABLE 47 NORTH AMERICA PROPHYLAXIS OF ORGAN TRANSPLANT, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 48 NORTH AMERICA EPIDEMIOLOGIC EXPOSURE IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 49 NORTH AMERICA ANTIBACTERIAL PROPHYLAXIS IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 50 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 51 NORTH AMERICA OUTPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 52 NORTH AMERICA CALCINEURIN INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 53 NORTH AMERICA TACROLIMUS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 54 NORTH AMERICA MYCOPHENOLIC ACIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 55 NORTH AMERICA MTOR INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 56 NORTH AMERICA CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 57 NORTH AMERICA INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 58 NORTH AMERICA OTHERS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 59 NORTH AMERICA ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 60 NORTH AMERICA ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 61 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 62 NORTH AMERICA ORAL IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 63 NORTH AMERICA TABLET IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 64 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION, BY ORGAN, 2020-2029 (USD MILLION)
TABLE 65 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 66 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION, BY END USER, 2020-2029 (USD MILLION)
TABLE 67 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 68 NORTH AMERICA PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 69 U.S. PROPHYLAXIS OF ORGAN TRANSPLANT, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 70 U.S. EPIDEMIOLOGIC EXPOSURE IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 71 U.S. ANTIBACTERIAL PROPHYLAXIS IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 72 U.S. PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 73 U.S. OUTPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 74 U.S. CALCINEURIN INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 75 U.S. TACROLIMUS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 76 U.S. MYCOPHENOLIC ACIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 77 U.S. MTOR INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 78 U.S. CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 79 U.S. INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 80 U.S. OTHERS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 81 U.S. ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 82 U.S. ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 83 U.S. PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 84 U.S. ORAL IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 85 U.S. TABLET IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 86 U.S. PROPHYLAXIS OF ORGAN REJECTION, BY ORGAN, 2020-2029 (USD MILLION)
TABLE 87 U.S. PROPHYLAXIS OF ORGAN REJECTION, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 88 U.S. PROPHYLAXIS OF ORGAN REJECTION, BY END USER, 2020-2029 (USD MILLION)
TABLE 89 U.S. PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 90 U.S. PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 91 CANADA PROPHYLAXIS OF ORGAN TRANSPLANT, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 92 CANADA EPIDEMIOLOGIC EXPOSURE IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 93 CANADA ANTIBACTERIAL PROPHYLAXIS IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 94 CANADA PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 95 CANADA OUTPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 96 CANADA CALCINEURIN INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 97 CANADA TACROLIMUS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 98 CANADA MYCOPHENOLIC ACIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 99 CANADA MTOR INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 100 CANADA CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 101 CANADA INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 102 CANADA OTHERS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 103 CANADA ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 104 CANADA ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 105 CANADA PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 106 CANADA ORAL IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 107 CANADA TABLET IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 108 CANADA PROPHYLAXIS OF ORGAN REJECTION, BY ORGAN, 2020-2029 (USD MILLION)
TABLE 109 CANADA PROPHYLAXIS OF ORGAN REJECTION, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 110 CANADA PROPHYLAXIS OF ORGAN REJECTION, BY END USER, 2020-2029 (USD MILLION)
TABLE 111 CANADA PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 112 CANADA PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 113 MEXICO PROPHYLAXIS OF ORGAN TRANSPLANT, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 114 MEXICO EPIDEMIOLOGIC EXPOSURE IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 115 MEXICO ANTIBACTERIAL PROPHYLAXIS IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 116 MEXICO PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 117 MEXICO OUTPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 118 MEXICO CALCINEURIN INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 119 MEXICO TACROLIMUS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 120 MEXICO MYCOPHENOLIC ACIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 121 MEXICO MTOR INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 122 MEXICO CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 123 MEXICO INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 124 MEXICO OTHERS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 125 MEXICO ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 126 MEXICO ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 127 MEXICO PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 128 MEXICO ORAL IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 129 MEXICO TABLET IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 130 MEXICO PROPHYLAXIS OF ORGAN REJECTION, BY ORGAN, 2020-2029 (USD MILLION)
TABLE 131 MEXICO PROPHYLAXIS OF ORGAN REJECTION, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 132 MEXICO PROPHYLAXIS OF ORGAN REJECTION, BY END USER, 2020-2029 (USD MILLION)
TABLE 133 MEXICO PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 134 MEXICO PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
Lista de figuras
FIGURE 1 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: SEGMENTATION
FIGURE 2 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: DATA TRIANGULATION
FIGURE 3 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: DROC ANALYSIS
FIGURE 4 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: NORTH AMERICA VS REGIONAL MARKET ANALYSIS
FIGURE 5 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: DBMR POSITION GRID
FIGURE 8 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: VENDOR SHARE ANALYSIS
FIGURE 9 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: END USER COVERAGE GRID
FIGURE 10 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: SEGMENTATION
FIGURE 11 NORTH AMERICA IS EXPECTED TO DOMINATE THE NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET AND ASIA-PACIFIC IS EXPECTED TO GROW WITH THE HIGHEST CAGR IN THE FORECAST PERIOD OF 2022 TO 2029
FIGURE 12 INCREASED PREVALENCE OF ORGAN TRANSPLANTATION, RISE IN RECIPIENT PATIENTS, AND PRODUCT APPPROVALS ARE EXPECTED TO DRIVE NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET FROM 2022 TO 2029
FIGURE 13 EPIDEMIOLOGIC EXPOSURE SEGMENT IS EXPECTED TO HAVE THE LARGEST SHARE OF THE NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET FROM 2022 & 2029
FIGURE 14 ASIA-PACIFIC IS THE FASTEST-GROWING MARKET FOR PROPHYLAXIS OF ORGAN REJECTION MANUFACTURERS IN THE FORECAST PERIOD OF 2022 TO 2029
FIGURE 15 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION: INCIDENCE, 2021
FIGURE 16 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION: INCIDENCE REGIONAL LEVEL, 2021
FIGURE 17 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION: PREVALENCE (2018-2021) AT REGIONAL LEVEL SNAPSHOT
FIGURE 18 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION: SURVIVAL OF PATIENT SNAPSHOT
FIGURE 19 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET
FIGURE 20 INCREASE COUNT OF KIDNEY TRANSPLANTATIONS IN THE U.S., 2020
FIGURE 21 TOTAL NUMBER OF ORGAN TRANSPLANTATION IN THE NORTH AMERICA SCENARIO, 2020
FIGURE 22 TOTAL NUMBER OF PATIENTS ON WAITING LIST VS TRANSPLANTS PERFORMED BY ORGAN IN 2020
FIGURE 23 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY CAUSE, 2021
FIGURE 24 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY CAUSE, 2020-2029 (USD MILLION)
FIGURE 25 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY CAUSE, CAGR (2022-2029)
FIGURE 26 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY CAUSE, LIFELINE CURVE
FIGURE 27 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY TREATMENT, 2021
FIGURE 28 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY TREATMENT, 2020-2029 (USD MILLION)
FIGURE 29 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY TREATMENT, CAGR (2022-2029)
FIGURE 30 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY TREATMENT, LIFELINE CURVE
FIGURE 31 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ROUTE OF ADMINISTRATION, 2021
FIGURE 32 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
FIGURE 33 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ROUTE OF ADMINISTRATION, CAGR (2022-2029)
FIGURE 34 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ROUTE OF ADMINISTRATION, LIFELINE CURVE
FIGURE 35 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ORGAN, 2021
FIGURE 36 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ORGAN, 2020-2029 (USD MILLION)
FIGURE 37 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ORGAN, CAGR (2022-2029)
FIGURE 38 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ORGAN, LIFELINE CURVE
FIGURE 39 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY PATIENT TYPE, 2021
FIGURE 40 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY PATIENT TYPE, 2020-2029 (USD MILLION)
FIGURE 41 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY PATIENT TYPE, CAGR (2022-2029)
FIGURE 42 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY PATIENT TYPE, LIFELINE CURVE
FIGURE 43 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY END USER, 2021
FIGURE 44 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY END USER, 2020-2029 (USD MILLION)
FIGURE 45 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY END USER, CAGR (2022-2029)
FIGURE 46 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY END USER, LIFELINE CURVE
FIGURE 47 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY DISTRIBUTION CHANNEL, 2021
FIGURE 48 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
FIGURE 49 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY DISTRIBUTION CHANNEL, CAGR (2022-2029)
FIGURE 50 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 51 NORTH AMERICA PROPHYLAXIS OF ORGAN TRANSPLANT MARKET: SNAPSHOT (2021)
FIGURE 52 NORTH AMERICA PROPHYLAXIS OF ORGAN TRANSPLANT MARKET: BY COUNTRY (2021)
FIGURE 53 NORTH AMERICA PROPHYLAXIS OF ORGAN TRANSPLANT MARKET: BY COUNTRY (2022 & 2029)
FIGURE 54 NORTH AMERICA PROPHYLAXIS OF ORGAN TRANSPLANT MARKET: BY COUNTRY (2021 & 2029)
FIGURE 55 NORTH AMERICA PROPHYLAXIS OF ORGAN TRANSPLANT MARKET: BY CAUSE (2022-2029)
FIGURE 56 NORTH AMERICA PROPHYLAXIS OF ORGAN REJECTION MARKET: COMPANY SHARE 2021 (%)

Metodología de investigación
La recopilación de datos y el análisis del año base se realizan utilizando módulos de recopilación de datos con muestras de gran tamaño. La etapa incluye la obtención de información de mercado o datos relacionados a través de varias fuentes y estrategias. Incluye el examen y la planificación de todos los datos adquiridos del pasado con antelación. Asimismo, abarca el examen de las inconsistencias de información observadas en diferentes fuentes de información. Los datos de mercado se analizan y estiman utilizando modelos estadísticos y coherentes de mercado. Además, el análisis de la participación de mercado y el análisis de tendencias clave son los principales factores de éxito en el informe de mercado. Para obtener más información, solicite una llamada de un analista o envíe su consulta.
La metodología de investigación clave utilizada por el equipo de investigación de DBMR es la triangulación de datos, que implica la extracción de datos, el análisis del impacto de las variables de datos en el mercado y la validación primaria (experto en la industria). Los modelos de datos incluyen cuadrícula de posicionamiento de proveedores, análisis de línea de tiempo de mercado, descripción general y guía del mercado, cuadrícula de posicionamiento de la empresa, análisis de patentes, análisis de precios, análisis de participación de mercado de la empresa, estándares de medición, análisis global versus regional y de participación de proveedores. Para obtener más información sobre la metodología de investigación, envíe una consulta para hablar con nuestros expertos de la industria.
Personalización disponible
Data Bridge Market Research es líder en investigación formativa avanzada. Nos enorgullecemos de brindar servicios a nuestros clientes existentes y nuevos con datos y análisis que coinciden y se adaptan a sus objetivos. El informe se puede personalizar para incluir análisis de tendencias de precios de marcas objetivo, comprensión del mercado de países adicionales (solicite la lista de países), datos de resultados de ensayos clínicos, revisión de literatura, análisis de mercado renovado y base de productos. El análisis de mercado de competidores objetivo se puede analizar desde análisis basados en tecnología hasta estrategias de cartera de mercado. Podemos agregar tantos competidores sobre los que necesite datos en el formato y estilo de datos que esté buscando. Nuestro equipo de analistas también puede proporcionarle datos en archivos de Excel sin procesar, tablas dinámicas (libro de datos) o puede ayudarlo a crear presentaciones a partir de los conjuntos de datos disponibles en el informe.