Middle East Molecular Point Of Care Testing Using Naat Market
Tamaño del mercado en miles de millones de dólares
Tasa de crecimiento anual compuesta (CAGR) :
% 
 USD
274.00 Million
USD
484.16 Million
2024
2032
USD
274.00 Million
USD
484.16 Million
2024
2032
| 2025 –2032 | |
| USD 274.00 Million | |
| USD 484.16 Million | |
|
|
|
|
Segmentación del mercado de pruebas moleculares en el punto de atención (mediante NAAT) en Oriente Medio, por producto (instrumentos, consumibles y reactivos), indicación (pruebas de infecciones respiratorias, pruebas de infecciones de transmisión sexual (ITS), pruebas de infecciones del tracto gastrointestinal y otras), usuario final (laboratorios, hospitales, clínicas, centros ambulatorios, atención domiciliaria, residencias de ancianos y otros), modalidad de prueba (pruebas con receta y pruebas de venta libre), canal de distribución (farmacia hospitalaria, farmacia minorista y farmacia en línea): tendencias del sector y pronóstico hasta 2032.
Tamaño del mercado de pruebas moleculares en el punto de atención (mediante NAAT) en Oriente Medio
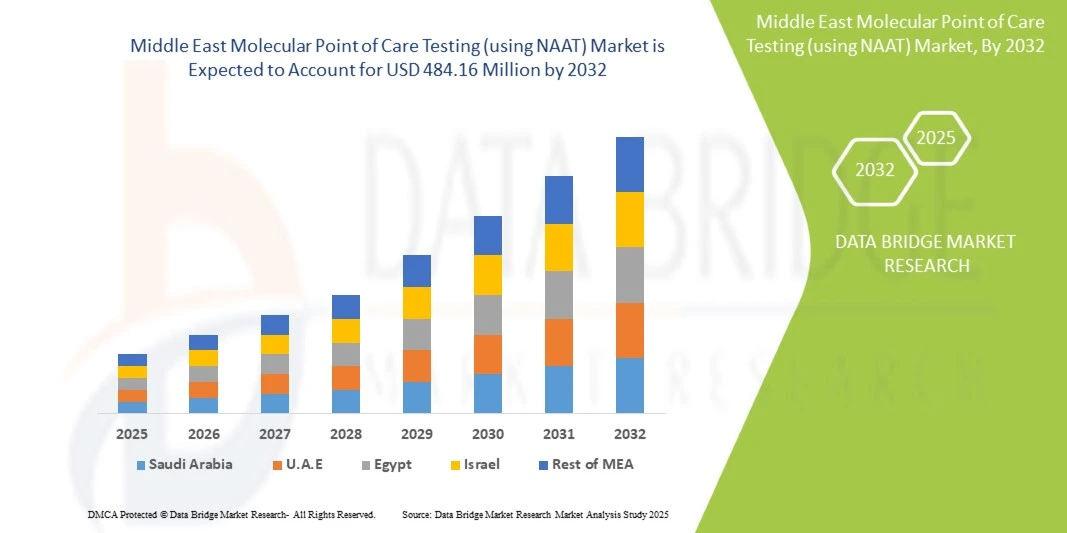
- El tamaño del mercado de pruebas moleculares en el punto de atención (utilizando NAAT) de Medio Oriente se valoró en USD 274,00 millones en 2024 y se espera que alcance los USD 484,16 millones para 2032 , con una CAGR del 7,4% durante el período de pronóstico.
- El crecimiento del mercado está impulsado en gran medida por la creciente prevalencia de enfermedades infecciosas, los avances tecnológicos en el diagnóstico molecular y la creciente adopción de pruebas en el punto de atención en hospitales, clínicas y laboratorios de diagnóstico.
- Además, el creciente gasto sanitario, las iniciativas gubernamentales para mejorar la accesibilidad al diagnóstico y la demanda de soluciones de pruebas rápidas, precisas y descentralizadas están consolidando las pruebas NAAT en el punto de atención como la herramienta de diagnóstico preferida en la región. Estos factores convergentes están acelerando la adopción de soluciones moleculares en el punto de atención, impulsando así significativamente el crecimiento del sector.
Análisis del mercado de pruebas moleculares en el punto de atención (NAAT) en Oriente Medio
- Las pruebas moleculares en el punto de atención que utilizan NAAT, que ofrecen una detección rápida y precisa de enfermedades infecciosas en el lugar de atención al paciente o cerca de él, son cada vez más vitales en los sistemas de atención médica modernos en hospitales, clínicas y laboratorios de diagnóstico debido a su alta sensibilidad, rápido tiempo de respuesta y facilidad de integración en los flujos de trabajo clínicos.
- La creciente demanda de pruebas en el punto de atención basadas en NAAT se ve impulsada principalmente por la creciente prevalencia de enfermedades infecciosas, la creciente conciencia de los diagnósticos tempranos y la preferencia por soluciones de pruebas rápidas y descentralizadas en lugar de las pruebas de laboratorio tradicionales.
- Arabia Saudita dominó el mercado de pruebas en el punto de atención basadas en NAAT en Medio Oriente con la mayor participación en los ingresos del 38,5 % en 2024, caracterizado por una infraestructura de atención médica avanzada, un alto gasto en atención médica y una fuerte presencia de actores clave de la industria, con un crecimiento sustancial impulsado por innovaciones tanto de empresas de diagnóstico establecidas como de nuevas empresas locales emergentes.
- Se espera que los Emiratos Árabes Unidos sean el país de más rápido crecimiento en el mercado durante el período de pronóstico, debido al aumento de las inversiones en atención médica, las iniciativas gubernamentales para mejorar el acceso al diagnóstico y la creciente adopción de tecnologías de diagnóstico modernas.
- El segmento de pruebas de infecciones respiratorias dominó el mercado con una participación del 50,5 % en 2024, impulsado por la alta prevalencia de infecciones virales y bacterianas y la confiabilidad clínica establecida de la tecnología NAAT para una detección rápida y precisa.
Alcance del informe y segmentación del mercado de pruebas moleculares en el punto de atención (mediante NAAT) en Oriente Medio
|
Atributos |
Análisis moleculares en el punto de atención en Oriente Medio (mediante NAAT): Perspectivas clave del mercado |
|
Segmentos cubiertos |
|
|
Países cubiertos |
|
|
Actores clave del mercado |
|
|
Oportunidades de mercado |
|
|
Conjuntos de información de datos de valor añadido |
Además de los conocimientos sobre escenarios de mercado como valor de mercado, tasa de crecimiento, segmentación, cobertura geográfica y actores principales, los informes de mercado seleccionados por Data Bridge Market Research también incluyen análisis de expertos en profundidad, análisis de precios, análisis de participación de marca, encuesta de consumidores, análisis demográfico, análisis de la cadena de suministro, análisis de la cadena de valor, descripción general de materias primas/consumibles, criterios de selección de proveedores, análisis PESTLE, análisis de Porter y marco regulatorio. |
Tendencias del mercado de pruebas moleculares en el punto de atención (mediante NAAT) en Oriente Medio
Pruebas rápidas y descentralizadas para enfermedades infecciosas
- Una tendencia significativa y en aceleración en el mercado de pruebas en el punto de atención basadas en NAAT en Medio Oriente es la creciente adopción de soluciones de diagnóstico rápidas y descentralizadas que pueden brindar resultados altamente precisos cerca del sitio de atención al paciente, lo que reduce la dependencia de los laboratorios centrales.
- Por ejemplo, la plataforma Cepheid GeneXpert se implementa ampliamente en hospitales y clínicas de Arabia Saudita y los Emiratos Árabes Unidos, proporcionando resultados casi al paciente para tuberculosis, COVID-19 e infecciones de transmisión sexual. De igual manera, los dispositivos Abbott ID NOW se están implementando en centros de salud comunitarios para la detección rápida de la influenza y la COVID-19.
- La integración con plataformas de salud digital y sistemas de información de laboratorio permite informes en tiempo real, alertas automatizadas y una mejor gestión de los pacientes. Por ejemplo, los dispositivos NAAT POCT conectados a los sistemas de Historias Clínicas Electrónicas (HCE) de hospitales de los EAU permiten a los médicos recibir notificaciones instantáneas de casos positivos, lo que facilita una intervención más rápida.
- La tendencia hacia dispositivos de prueba NAAT portátiles, fáciles de usar y multiplex está impulsando una accesibilidad más amplia, lo que permite que las clínicas e incluso los centros de pruebas remotos realicen diagnósticos rápidos de múltiples enfermedades infecciosas.
- Esta transición hacia pruebas rápidas, precisas y descentralizadas está transformando las expectativas de diagnóstico en la región. Empresas como bioMérieux y QIAGEN están desarrollando dispositivos compactos de NAAT para el punto de atención con funciones integradas de generación de informes y monitorización remota, lo que mejora la velocidad y la fiabilidad de la detección de enfermedades infecciosas.
- La demanda de soluciones POCT moleculares rápidas, descentralizadas e integradas digitalmente está creciendo en hospitales, clínicas y centros de pruebas comunitarios, a medida que los proveedores de atención médica priorizan cada vez más un diagnóstico más rápido y una mejor gestión del paciente.
Dinámica del mercado de pruebas moleculares en el punto de atención (mediante NAAT) en Oriente Medio
Conductor
Aumento de la prevalencia de enfermedades infecciosas e inversiones en atención médica
- La creciente carga de enfermedades infecciosas como la COVID-19, la gripe, la tuberculosis y las infecciones de transmisión sexual en los países de Oriente Medio, junto con las crecientes inversiones en infraestructura sanitaria, es un factor importante para la creciente adopción de pruebas en el punto de atención basadas en NAAT.
- Por ejemplo, en 2024, el Ministerio de Salud saudí amplió sus programas de pruebas rápidas en hospitales y centros de atención primaria, implementando dispositivos NAAT para la detección temprana de COVID-19 e infecciones respiratorias. Se espera que estas iniciativas de los gobiernos y las organizaciones sanitarias impulsen el crecimiento del mercado durante el período de pronóstico.
- A medida que los médicos y las autoridades de salud pública priorizan el diagnóstico oportuno y el manejo eficaz de los pacientes, las soluciones NAAT POCT ofrecen una respuesta rápida, alta sensibilidad y detección precisa, lo que proporciona una alternativa convincente a las pruebas de laboratorio centralizadas tradicionales.
- Además, la creciente adopción de registros médicos electrónicos, telemedicina y plataformas de salud digital está creando un entorno favorable para soluciones POCT integradas, lo que permite una gestión de datos fluida y la generación de informes en tiempo real.
- La necesidad de soluciones de diagnóstico rápidas, precisas y descentralizadas, junto con el apoyo gubernamental y las inversiones en infraestructura de atención médica, está impulsando la adopción de pruebas en el punto de atención basadas en NAAT en los sectores de atención médica públicos y privados.
Restricción/Desafío
Costos elevados y mano de obra calificada limitada
- El costo relativamente alto de los dispositivos, consumibles y reactivos POCT basados en NAAT sigue siendo un desafío importante para una mayor penetración en el mercado, especialmente en clínicas pequeñas y hospitales con presupuestos limitados. Si bien los precios están disminuyendo gradualmente, la inversión inicial en dispositivos avanzados de pruebas moleculares puede dificultar su adopción.
- Por ejemplo, hospitales más pequeños en Egipto y Qatar han informado limitaciones presupuestarias al adquirir plataformas NAAT avanzadas de empresas como Cepheid y Abbott, lo que limita su implementación generalizada.
- Otro desafío clave es la escasez de personal clínico y de laboratorio capacitado para operar con precisión los dispositivos NAAT e interpretar los resultados. La mala gestión o el manejo inadecuado pueden afectar la fiabilidad de las pruebas y los resultados de los pacientes.
- Abordar estos desafíos mediante programas de capacitación, subsidios gubernamentales y plataformas NAAT rentables será vital para el crecimiento sostenido del mercado. Empresas como bioMérieux y QIAGEN priorizan interfaces intuitivas, capacitación remota y soporte para impulsar la adopción en entornos con recursos limitados.
- Superar los altos costos y las limitaciones de la fuerza laboral mediante soluciones NAAT innovadoras, fáciles de usar y asequibles será fundamental para ampliar el acceso e impulsar el crecimiento del mercado a largo plazo en el Medio Oriente.
Alcance del mercado de pruebas moleculares en el punto de atención (mediante NAAT) en Oriente Medio
El mercado está segmentado según el producto, la indicación, el usuario final, el modo de prueba y el canal de distribución.
- Por producto
Según el producto, el mercado se segmenta en instrumentos, consumibles y reactivos. Los instrumentos dominaron el mercado en 2024, con la mayor participación en los ingresos debido a su papel crucial en las pruebas basadas en NAAT y su alto costo inicial. Hospitales, clínicas y laboratorios de referencia prefieren invertir en instrumentos avanzados para garantizar resultados precisos y confiables. Los instrumentos admiten múltiples ensayos y pruebas multiplex, lo que permite una gestión eficiente del diagnóstico respiratorio, gastrointestinal y de ITS. Los gobiernos y los proveedores de atención médica en Arabia Saudita y los Emiratos Árabes Unidos están implementando estos dispositivos en programas de salud pública para optimizar la respuesta diagnóstica. Su durabilidad, automatización e integración con plataformas de salud digital los hacen indispensables en los flujos de trabajo hospitalarios. Grandes empresas como Cepheid, Abbott y bioMérieux ofrecen instrumentos compactos y automatizados diseñados para pruebas en persona, lo que impulsa aún más su adopción.
Se prevé que los consumibles y reactivos experimenten el mayor crecimiento entre 2025 y 2033, impulsado por la demanda recurrente de cartuchos de prueba, kits de extracción y reactivos con cada prueba realizada. La creciente prevalencia de enfermedades infecciosas y la expansión de los centros de pruebas comunitarios impulsan la demanda. Los reactivos multiplex, capaces de detectar múltiples patógenos simultáneamente, mejoran la rentabilidad y resultan más atractivos para los profesionales sanitarios. Las clínicas emergentes y los hospitales más pequeños prefieren estos consumibles para realizar pruebas flexibles y rápidas sin invertir en múltiples instrumentos. El aumento de las campañas de cribado impulsadas por el gobierno y la integración de la telesalud impulsan aún más su adopción. El crecimiento de estos consumibles garantiza un flujo de ingresos estable para las empresas de diagnóstico.
- Por indicación
Según las indicaciones, el mercado se segmenta en pruebas de infecciones respiratorias, pruebas de infecciones de transmisión sexual (ITS), pruebas de infecciones del tracto gastrointestinal y otras. Las pruebas de infecciones respiratorias dominaron el mercado en 2024 debido a la alta prevalencia de enfermedades como la COVID-19, la influenza y el VSR en Oriente Medio. Los hospitales y las clínicas dependen de las pruebas rápidas de NAAT para el manejo oportuno de los pacientes y el control de infecciones. Los gobiernos de Arabia Saudita y los Emiratos Árabes Unidos han lanzado iniciativas de detección masiva, lo que ha impulsado aún más la demanda. La alta sensibilidad y la rápida respuesta de la NAAT la hacen preferible a los métodos basados en antígenos o cultivos. El dominio del segmento se ve reforzado por los brotes estacionales y las medidas de preparación para pandemias. Los proveedores de atención médica priorizan las pruebas respiratorias, ya que impactan directamente en las decisiones de tratamiento y la eficiencia del flujo de trabajo hospitalario.
Se espera que las pruebas de infecciones de transmisión sexual (ITS) experimenten el crecimiento más rápido entre 2025 y 2033 debido a la creciente concienciación y a los programas de detección respaldados por el gobierno. Las pruebas de ITS basadas en NAAT ofrecen alta sensibilidad y especificidad para clamidia, gonorrea y otras ITS, lo que las hace ideales para el diagnóstico cercano al paciente. La creciente adopción en clínicas y centros ambulatorios para pruebas discretas y rápidas impulsa la expansión. La creciente prevalencia de ITS en poblaciones urbanas y la creciente demanda de soluciones de pruebas privadas respaldan el rápido crecimiento de este segmento. Los paneles de NAAT multiplex para la detección de ITS mejoran aún más la eficiencia y la conveniencia. La expansión de la telesalud y la recolección de muestras en el hogar también impulsa la adopción de NAAT para ITS.
- Por el usuario final
Según el usuario final, el mercado se segmenta en laboratorios, hospitales, clínicas, centros ambulatorios, atención domiciliaria, residencias de ancianos y otros. Los hospitales dominaron el mercado en 2024 debido al alto volumen de pacientes y a la necesidad de diagnósticos rápidos y fiables. Los hospitales se benefician de los instrumentos NAAT internos que reducen los retrasos en el diagnóstico y mejoran el control de infecciones. Su implementación en servicios de urgencias, hospitalización y consultas externas garantiza una demanda continua. Los hospitales públicos y privados de Arabia Saudita y los Emiratos Árabes Unidos adoptan NAAT POCT para sus programas nacionales de cribado. La combinación de la cantidad de pacientes, las necesidades clínicas y la inversión en infraestructura convierte a los hospitales en la principal fuente de ingresos. Los hospitales también utilizan sistemas integrados de informes para el seguimiento en tiempo real de las tendencias de las enfermedades infecciosas.
Se prevé que las clínicas sean el segmento de usuarios finales de mayor crecimiento entre 2025 y 2033, impulsado por la descentralización de las pruebas y la demanda de diagnósticos cercanos. Las clínicas pueden proporcionar resultados el mismo día, lo que reduce las derivaciones a laboratorios centrales y mejora la satisfacción del paciente. El crecimiento de los centros de atención primaria y las clínicas comunitarias en los Emiratos Árabes Unidos, Egipto y Catar está impulsando su adopción. Las clínicas prefieren dispositivos de NAAT compactos y fáciles de usar que requieren una mínima capacitación del personal. La tendencia de los chequeos médicos preventivos y las pruebas de detección rutinarias de enfermedades infecciosas acelera aún más el crecimiento. La creciente integración con plataformas de telemedicina mejora la comodidad y la escalabilidad de las pruebas de diagnóstico en el punto de atención (POCT) en las clínicas.
- Por modo de prueba
Según el método de prueba, el mercado se segmenta en pruebas con receta y pruebas de venta libre. Las pruebas con receta dominaron el mercado en 2024 debido a los requisitos de supervisión médica para las pruebas NAAT y al cumplimiento normativo. Los hospitales y clínicas ofrecen pruebas con receta para garantizar un manejo e interpretación precisos. Las políticas regulatorias en Arabia Saudita, Emiratos Árabes Unidos y Egipto exigen la supervisión profesional para el diagnóstico molecular. El alto costo y la capacitación especializada refuerzan aún más el dominio de las pruebas con receta. Este enfoque garantiza el control de calidad y reduce el riesgo de diagnósticos erróneos. La mayoría de los programas de cribado a gran escala y las implementaciones hospitalarias se basan en soluciones NAAT con receta.
Se prevé que las pruebas de venta libre experimenten el mayor crecimiento entre 2025 y 2033 debido a la creciente demanda de diagnósticos domiciliarios y la integración de la telesalud. Los pacientes pueden realizar pruebas NAAT para COVID-19, influenza y otras infecciones en casa y compartir los resultados con los profesionales de la salud de forma remota. Este enfoque aumenta la accesibilidad y la comodidad, especialmente en zonas urbanas con un ritmo de vida ajetreado. Las plataformas digitales permiten la orientación y la elaboración de informes a distancia, lo que impulsa aún más su adopción. La creciente concienciación sobre la atención médica preventiva y el autodiagnóstico impulsa la expansión de este segmento. Las pruebas NAAT de venta libre ofrecen flexibilidad a los pacientes y alivian la presión sobre hospitales y clínicas.
- Por canal de distribución
Según el canal de distribución, el mercado se segmenta en farmacia hospitalaria, farmacia minorista y farmacia en línea. La farmacia hospitalaria dominó el mercado en 2024 gracias a la centralización de las compras para hospitales y clínicas. Las farmacias hospitalarias gestionan eficientemente el suministro de instrumental, kits de prueba y consumibles en múltiples departamentos. La adquisición a granel garantiza la rentabilidad y un suministro constante de reactivos. Los hospitales dependen de la logística gestionada por las farmacias para mantener servicios de prueba ininterrumpidos. La integración con los sistemas informáticos del hospital permite la gestión del inventario y la reposición en tiempo real. Las farmacias hospitalarias siguen siendo la principal fuente de ingresos para las empresas de NAAT POCT en la región.
Se prevé que la farmacia en línea experimente el mayor crecimiento entre 2025 y 2033, a medida que se expande la adopción de la salud digital en Oriente Medio. Los pacientes pueden solicitar kits de NAAT para pruebas a domicilio con servicio de entrega a domicilio, beneficiándose de la comodidad y el acceso discreto. Las plataformas de telesalud facilitan la orientación y el envío de resultados a distancia. El crecimiento del comercio electrónico y las aplicaciones móviles de salud acelera la adopción de este canal. Las pruebas a domicilio y la monitorización remota mejoran la participación del paciente. Las farmacias en línea brindan accesibilidad tanto a poblaciones urbanas como remotas, lo que las convierte en un segmento de distribución en rápida expansión.
Análisis regional del mercado de pruebas moleculares en el punto de atención (NAAT) en Oriente Medio
- Arabia Saudita dominó el mercado de pruebas en el punto de atención basadas en NAAT en Medio Oriente con la mayor participación en los ingresos del 38,5 % en 2024, caracterizado por una infraestructura de atención médica avanzada, un alto gasto en atención médica y una fuerte presencia de actores clave de la industria, con un crecimiento sustancial impulsado por innovaciones tanto de empresas de diagnóstico establecidas como de nuevas empresas locales emergentes.
- Los profesionales sanitarios y las autoridades de salud pública de Arabia Saudita valoran enormemente la precisión, la velocidad y la fiabilidad de las pruebas de función pulmonar (POCT) basadas en NAAT para el tratamiento de infecciones respiratorias, ITS y otras enfermedades infecciosas. El diagnóstico rápido facilita el tratamiento oportuno, la contención de brotes y una mejor evolución de los pacientes.
- Esta adopción generalizada se ve respaldada además por el alto gasto sanitario, un personal médico tecnológicamente avanzado y sólidas alianzas con empresas líderes mundiales en diagnóstico. Hospitales, clínicas y laboratorios integran cada vez más dispositivos NAAT POCT con los sistemas de información hospitalaria, lo que permite la generación de informes en tiempo real y una gestión eficiente de los pacientes.
Análisis del mercado de pruebas moleculares en el punto de atención (NAAT) en Arabia Saudita
El mercado de pruebas moleculares en el punto de atención (mediante NAAT) de Arabia Saudita capturó la mayor participación en los ingresos, con un 38,5 %, en 2024, impulsado por la alta prevalencia de enfermedades infecciosas y una infraestructura sanitaria avanzada. Hospitales, clínicas y laboratorios están adoptando cada vez más el diagnóstico basado en NAAT para la detección rápida y precisa de infecciones respiratorias, ITS e infecciones gastrointestinales. Los programas nacionales de cribado del gobierno y las iniciativas de preparación ante pandemias impulsan aún más su adopción. El aumento de las inversiones en tecnología sanitaria y la integración de dispositivos NAAT con plataformas de salud digital, como los sistemas de información hospitalaria y la telemedicina, están ampliando el alcance del mercado. El creciente enfoque en la atención médica preventiva y las medidas de control de infecciones continúa impulsando el crecimiento del mercado.
Análisis del mercado de pruebas moleculares en el punto de atención (NAAT) en Emiratos Árabes Unidos
El mercado de pruebas moleculares en el punto de atención de los EAU se perfila como el país de mayor crecimiento de la región gracias al aumento de la inversión en salud y la expansión de las redes hospitalarias. La adopción de pruebas basadas en NAAT cuenta con el apoyo de programas gubernamentales para la vigilancia de enfermedades infecciosas y la integración de la telesalud. Clínicas y hospitales están adoptando pruebas rápidas en el lugar del paciente para un diagnóstico oportuno y una mejor atención. El sistema de salud tecnológicamente avanzado de los EAU, junto con la alta concienciación de los pacientes sobre el diagnóstico temprano, está impulsando el crecimiento del mercado. La creciente demanda de pruebas NAAT multiplex y soluciones de atención domiciliaria también contribuye a su adopción.
Análisis del mercado de pruebas moleculares en el punto de atención en Egipto (mediante NAAT)
El mercado egipcio de pruebas de diagnóstico por angiotensina (PAA) basadas en pruebas de actividad de la sangre (NAAT) representó una participación significativa en 2024, impulsado por la mejora de la infraestructura sanitaria y la creciente concienciación sobre el diagnóstico molecular rápido. Los hospitales y centros de atención primaria adoptan cada vez más las pruebas NAAT para infecciones respiratorias e ITS. Las iniciativas gubernamentales que apoyan las pruebas descentralizadas y los programas de cribado comunitarios impulsan el crecimiento del mercado. La expansión de las plataformas de telemedicina y los sistemas de informes digitales mejora la accesibilidad y la comodidad. La creciente prevalencia de enfermedades infecciosas y la creciente demanda de diagnósticos más rápidos por parte de los pacientes impulsan el mercado. La asequibilidad y las colaboraciones locales con empresas de diagnóstico impulsan aún más su adopción.
Análisis del mercado de pruebas moleculares en el punto de atención (NAAT) en Qatar
El mercado catarí de pruebas NAAT en el punto de atención experimenta un crecimiento constante gracias a los programas de salud respaldados por el gobierno y al creciente énfasis en el diagnóstico preventivo. Hospitales y clínicas están adoptando dispositivos NAAT para la detección de enfermedades infecciosas y el monitoreo rutinario. El elevado gasto sanitario per cápita, sumado a una sólida infraestructura sanitaria, impulsa la adopción de diagnósticos moleculares avanzados. El uso de NAAT para el diagnóstico rápido en urgencias y consultas externas impulsa aún más la expansión del mercado. La integración con los sistemas informáticos hospitalarios permite la generación de informes en tiempo real y una gestión eficiente de los pacientes. La creciente concienciación sobre el diagnóstico temprano entre los pacientes fomenta su uso tanto en el sector sanitario público como en el privado.
Cuota de mercado de pruebas moleculares en el punto de atención (mediante NAAT) en Oriente Medio
La industria de pruebas moleculares en el punto de atención (mediante NAAT) en Oriente Medio está liderada principalmente por empresas bien establecidas, entre las que se incluyen:
- Abbott (EE. UU.)
- F. Hoffmann-La Roche Ltd (Suiza)
- BIOMÉRIEUX (Francia)
- QIAGEN (Países Bajos)
- Danaher (Estados Unidos)
- Thermo Fisher Scientific Inc. (EE. UU.)
- BD (EE. UU.)
- Illumina, Inc. (EE. UU.)
- Siemens Healthineers AG (Alemania)
- Grifols, SA (España)
- QuidelOrtho Corporation (EE. UU.)
- DiaSorin SpA (Italia)
- Corporación Sysmex (Japón)
- Sekisui Diagnostics, LLC (EE. UU.)
- Hologic, Inc. (EE. UU.)
- Medtronic (Irlanda)
- Atlas Medical GmbH (Alemania)
- Nova Biomedical (EE. UU.)
- Werfen (España)
¿Cuáles son los últimos avances en el mercado de pruebas moleculares en el punto de atención (utilizando NAAT) en Oriente Medio?
- En febrero de 2025, Aptitude recibió la Autorización de Uso de Emergencia (EUA) de la FDA de EE. UU. para su prueba multiplex Metrix COVID/Flu. Esta prueba proporciona una precisión de grado PCR para la detección y diferenciación del SARS-CoV-2, la influenza A y la influenza B en aproximadamente 20 minutos en el punto de atención.
- En enero de 2025, Roche recibió la autorización de la FDA con exención CLIA para sus pruebas moleculares cobas liat, que facilitan el diagnóstico de infecciones de transmisión sexual (ITS) en el punto de atención. Estas pruebas, que ofrecen resultados en 20 minutos, permiten a los profesionales sanitarios diagnosticar y diferenciar múltiples ITS con una sola muestra mediante la tecnología PCR de referencia.
- En octubre de 2024, Access Genetics (que opera como OralDNA® Labs) adquirió Sensible Diagnostics Inc. El objetivo de la adquisición es comercializar una nueva plataforma de diagnóstico molecular en el punto de atención que busca brindar resultados de pruebas con calidad de laboratorio central a partir de saliva o hisopos en menos de 10 minutos, mejorando la velocidad y la asequibilidad de los diagnósticos.
- En septiembre de 2024, QIAGEN amplió su alianza estratégica con Bio-Manguinhos/Fiocruz de Brasil. La colaboración se centra en proporcionar plataformas avanzadas de cribado molecular basadas en PCR para enfermedades como la malaria, el VIH y las hepatitis B y C, dentro del programa nacional de donación de sangre de Brasil.
- En mayo de 2024, bioMérieux anunció una alianza estratégica con AnaBioTec, proveedor belga de servicios analíticos. La colaboración se centra en transformar las pruebas de micoplasma para las industrias biofarmacéutica y de terapia celular y génica mediante el uso del sistema de PCR totalmente automatizado de bioMérieux para ofrecer una solución interna más rápida y precisa.
SKU-
Obtenga acceso en línea al informe sobre la primera nube de inteligencia de mercado del mundo
- Panel de análisis de datos interactivo
- Panel de análisis de empresas para oportunidades con alto potencial de crecimiento
- Acceso de analista de investigación para personalización y consultas
- Análisis de la competencia con panel interactivo
- Últimas noticias, actualizaciones y análisis de tendencias
- Aproveche el poder del análisis de referencia para un seguimiento integral de la competencia
Metodología de investigación
La recopilación de datos y el análisis del año base se realizan utilizando módulos de recopilación de datos con muestras de gran tamaño. La etapa incluye la obtención de información de mercado o datos relacionados a través de varias fuentes y estrategias. Incluye el examen y la planificación de todos los datos adquiridos del pasado con antelación. Asimismo, abarca el examen de las inconsistencias de información observadas en diferentes fuentes de información. Los datos de mercado se analizan y estiman utilizando modelos estadísticos y coherentes de mercado. Además, el análisis de la participación de mercado y el análisis de tendencias clave son los principales factores de éxito en el informe de mercado. Para obtener más información, solicite una llamada de un analista o envíe su consulta.
La metodología de investigación clave utilizada por el equipo de investigación de DBMR es la triangulación de datos, que implica la extracción de datos, el análisis del impacto de las variables de datos en el mercado y la validación primaria (experto en la industria). Los modelos de datos incluyen cuadrícula de posicionamiento de proveedores, análisis de línea de tiempo de mercado, descripción general y guía del mercado, cuadrícula de posicionamiento de la empresa, análisis de patentes, análisis de precios, análisis de participación de mercado de la empresa, estándares de medición, análisis global versus regional y de participación de proveedores. Para obtener más información sobre la metodología de investigación, envíe una consulta para hablar con nuestros expertos de la industria.
Personalización disponible
Data Bridge Market Research es líder en investigación formativa avanzada. Nos enorgullecemos de brindar servicios a nuestros clientes existentes y nuevos con datos y análisis que coinciden y se adaptan a sus objetivos. El informe se puede personalizar para incluir análisis de tendencias de precios de marcas objetivo, comprensión del mercado de países adicionales (solicite la lista de países), datos de resultados de ensayos clínicos, revisión de literatura, análisis de mercado renovado y base de productos. El análisis de mercado de competidores objetivo se puede analizar desde análisis basados en tecnología hasta estrategias de cartera de mercado. Podemos agregar tantos competidores sobre los que necesite datos en el formato y estilo de datos que esté buscando. Nuestro equipo de analistas también puede proporcionarle datos en archivos de Excel sin procesar, tablas dinámicas (libro de datos) o puede ayudarlo a crear presentaciones a partir de los conjuntos de datos disponibles en el informe.