Mercado de terapia génica de Asia y el Pacífico, por tipo de vector (vector viral y vector no viral), método (ex vivo e in vivo), aplicación (trastornos oncológicos, enfermedades cardiovasculares, enfermedades infecciosas , enfermedades raras, trastornos neurológicos y otras enfermedades), usuario final (institutos de cáncer, hospitales, institutos de investigación y otros): tendencias de la industria y pronóstico hasta 2030.
Análisis y perspectivas del mercado de terapia génica en Asia y el Pacífico
Se espera que el mercado de terapia génica de Asia-Pacífico crezca en el año de pronóstico debido al aumento de los participantes del mercado y la disponibilidad de servicios y productos avanzados. Junto con esto, los fabricantes están involucrados en actividades de I+D para lanzar productos novedosos al mercado.
Se espera que la creciente investigación en terapia génica y sus técnicas y desarrollo impulse aún más el crecimiento del mercado. Sin embargo, se espera que las preocupaciones éticas y de seguridad al realizar el método obstaculicen el crecimiento del mercado de terapia génica de Asia y el Pacífico en el período de pronóstico. Se espera que la creciente demanda de terapia génica, un campo emergente y avanzado en ingeniería genética y atención médica, brinde oportunidades al mercado para mejorar los enfoques de tratamiento y diagnóstico.
Se espera que la creciente demanda de una atención sanitaria de mejor calidad para el cáncer y los trastornos genéticos impulse el crecimiento del mercado. Sin embargo, se espera que el alto coste de los diagnósticos y la falta de profesionales cualificados y certificados supongan un reto para el crecimiento del mercado.
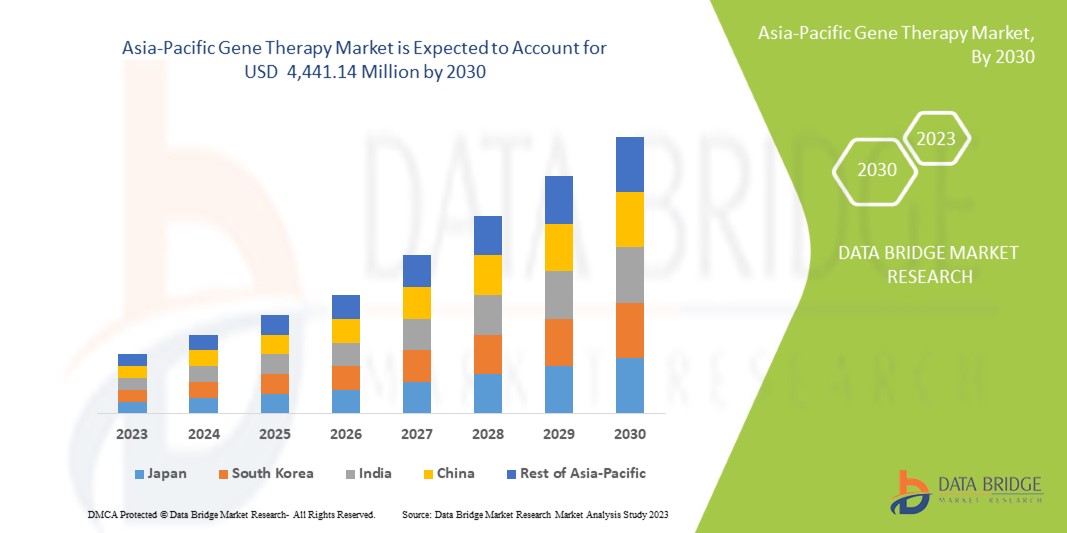
Data Bridge Market Research analiza que se espera que el mercado de terapia génica de Asia-Pacífico alcance los 4.441,14 millones de dólares en 2030, con una tasa compuesta anual del 19,0 % durante el período de pronóstico. Los productos representan el segmento de tipo más importante en el mercado debido al creciente uso de productos de investigación de exosomas en diagnósticos y terapias.
|
Métrica del informe |
Detalles |
|
Período de pronóstico |
2023 a 2030 |
|
Año base |
2022 |
|
Años históricos |
2021 (Personalizable para 2015-2020) |
|
Unidades cuantitativas |
Ingresos en millones de USD, precios en USD |
|
Segmentos cubiertos |
Por tipo de vector (vector viral y vector no viral), método (ex vivo e in vivo), aplicación (trastornos oncológicos, enfermedades cardiovasculares, enfermedades infecciosas, enfermedades raras, trastornos neurológicos y otras enfermedades), usuario final (institutos oncológicos, hospitales, institutos de investigación y otros) |
|
Países cubiertos |
China, Japón, India, Australia, Corea del Sur, Singapur, Malasia, Tailandia, Indonesia, Filipinas, Resto de Asia-Pacífico |
|
Actores del mercado cubiertos |
Novartis AG, Kite Pharma (una subsidiaria de Gilead Sciences, Inc.), uniQure NV., Oxford Biomedica, Spark Therapeutics, Inc., SIBONO, bluebird bio, Inc., Shanghai Sunway Biotech Co., Ltd., Biogen, Dendreon Pharmaceuticals LLC., Amgen Inc., AnGes, Inc. y Enzyvant Therapeutics GmbH, entre otros. |
Definición del mercado de terapia génica en Asia y el Pacífico
La terapia génica es una estrategia médica que aborda el problema genético subyacente para tratar o prevenir una enfermedad. En lugar de utilizar medicamentos o cirugía, los procedimientos de terapia génica permiten a los médicos tratar un problema modificando la composición genética de una persona. Unos pocos trastornos seleccionados, entre ellos una afección ocular llamada amaurosis congénita de Leber y una afección muscular llamada atrofia muscular espinal, se están tratando con terapia génica. Para garantizar que sean seguras y eficaces, se están estudiando muchas otras terapias génicas. Los profesionales médicos aspiran a aplicar pronto la prometedora técnica de edición genómica para curar enfermedades humanas.
La capacidad de transportar con éxito un gen terapéutico a una célula diana es el requisito previo más importante para que la terapia génica tenga éxito. Una vez transportado, ese gen debe ir al núcleo de la pared celular, donde servirá como modelo para la fabricación de moléculas de proteína. La proteína produce entonces la principal acción terapéutica. Por ejemplo, la destrucción celular podría utilizarse en el tratamiento de tumores, mientras que la conservación celular podría utilizarse en el caso de enfermedades neurodegenerativas. Sin embargo, se espera que las estrictas regulaciones y normas para la aprobación y comercialización de productos limiten el crecimiento del mercado.
Dinámica del mercado de terapia génica en Asia y el Pacífico
En esta sección se aborda la comprensión de los factores impulsores, las ventajas, las oportunidades, las limitaciones y los desafíos del mercado. Todo esto se analiza en detalle a continuación:
Conductores
- Nuevos enfoques de la terapia génica
La terapia génica ha permitido curar de forma permanente enfermedades que antes eran meros tratamientos temporales. Durante mucho tiempo, la terapia génica no funcionó; sin embargo, en los últimos años se han registrado casos de tratamiento eficaz y duradero. Se han obtenido resultados prometedores para una amplia gama de enfermedades hereditarias, incluidas anomalías sanguíneas, deficiencias inmunológicas, problemas de visión, regeneración de células nerviosas, trastornos metabólicos y diferentes tipos de cáncer.
Con mayor especificidad y menos efectos secundarios, la terapia génica tiene el potencial de ser una medicina personalizada que puede "curar" una variedad de enfermedades. La terapia génica generalmente se refiere a la transferencia de material genético para tratar una enfermedad o, al menos, para mejorar el estado clínico de un paciente. El uso de virus como vectores genéticos para entregar el gen deseado a las células objetivo es un método de cómo funciona la terapia génica. Estos vectores se clasifican como vectores virales basados en ARN o basados en ADN según el tipo de genoma que contengan.
La mayoría de los expertos coinciden en que la terapia génica tiene el potencial de ser el uso más interesante de la investigación del ADN hasta la fecha. Una simple inyección intravenosa de un agente de transferencia génica podría algún día utilizarse para administrar genes como medicina, buscando células diana para la integración cromosómica estable y específica del sitio y la posterior expresión génica. Se prevé que existirá una necesidad de terapia génica que utilice técnicas revolucionarias que están siendo probadas por investigadores de todo el mundo e incorporadas al tratamiento convencional y se espera que actúen como impulsores del crecimiento del mercado de terapia génica en Asia y el Pacífico.
- Aumento de la prevalencia de trastornos genéticos
En varios países de la región, una parte considerable de la mortalidad prenatal y neonatal se debe a enfermedades genéticas y congénitas. Muchas enfermedades multifactoriales también suelen tener su origen en factores genéticos. Las alteraciones genéticas que están presentes en casi todas las células del cuerpo causan muchas enfermedades hereditarias. Por lo tanto, estas enfermedades afectan con frecuencia a muchos sistemas corporales y la mayoría de ellas no pueden tratarse.
Por ejemplo,
El Departamento de Salud indicó que aproximadamente seis de cada diez personas se verán afectadas por una enfermedad que tiene algún vínculo genético, según el Gobierno de Australia Occidental. Los trastornos genéticos pueden variar de leves a muy graves. Entre el 3 y el 5% de los recién nacidos en Australia Occidental tienen trastornos genéticos o anomalías congénitas.
Las mutaciones, la exposición a sustancias químicas y la radiación, entre otras cosas, pueden provocar trastornos genéticos. Aunque algunas enfermedades se han tratado con terapia génica, la mayoría de los planes de tratamiento para trastornos genéticos no modifican la anomalía genética subyacente. Por ello, la prevalencia de anomalías genéticas está aumentando significativamente en todos los grupos de edad y se espera que prácticamente todas las áreas geográficas actúen como impulsoras del crecimiento del mercado de terapia génica en Asia y el Pacífico.
Restricción
- El alto coste de la terapia genética
Una nueva línea de tratamientos médicos llamada terapia génica implica reemplazar, eliminar o introducir información genética en el genoma de un paciente para tratar una enfermedad. Aunque todavía está en sus inicios, la terapia génica ya ha demostrado ser muy prometedora para el tratamiento e incluso la cura de enfermedades que antes eran intratables. El precio de la terapia génica todavía está muy descontrolado y se determina caso por caso en muchos países, centrándose con frecuencia en un único pago inicial.
Por ejemplo,
- Según un artículo de noticias publicado en Web MD en febrero de 2023, Hemgenix está batiendo récords, pero no es una anomalía. En septiembre de 2022, Skysona, un medicamento para una enfermedad neurológica poco común, salió a la venta por 3 millones de dólares. Tan solo un mes antes, Zynteglo, un tratamiento genético para una enfermedad sanguínea genética, debutó en el mercado por 2,8 millones de dólares. Una cura para la atrofia muscular espinal, una enfermedad genética que mata a bebés y niños pequeños, llamada Zolgensma, costó 2,1 millones de dólares en 2019.
Aunque no siempre sean totalmente curativas, las terapias génicas pueden ser realmente transformadoras. La principal barrera para acceder a la terapia génica es el costo. El panorama de tratamiento de muchas enfermedades genéticas raras experimentará cambios significativos a medida que estén disponibles más productos de terapia génica, brindando a los pacientes alternativas potencialmente curativas por primera vez. La dificultad de garantizar que todos los pacientes, no solo un pequeño grupo con medios financieros y acceso privilegiado a la tecnología, puedan beneficiarse de estas terapias de vanguardia debe ser abordada por los sistemas de atención médica de todo el mundo. Sin embargo, debido al alto costo de los tratamientos,
Oportunidad
- Aumento de las adquisiciones estratégicas y las asociaciones entre organizaciones
Recientemente, diferentes organizaciones están dando un paso adelante para asociarse y colaborar con el fin de desarrollar diversos productos de terapia génica que son esenciales para detectar trastornos genéticos. No solo eso, con la ayuda de asociaciones y acuerdos, ambas empresas pueden desarrollar un nuevo conjunto de tecnologías y plataformas que ayudarán a detectar enfermedades.
Gracias a un acuerdo a largo plazo, ambas empresas pueden ofrecer precios dimensionales para los productos de terapia génica en respuesta a la demanda de los consumidores en el mercado. Esta asociación y este acuerdo mutuo no solo benefician a ambas empresas, sino que también crean muchas oportunidades para que el mercado crezca.
Por ejemplo,
- En julio de 2022, Novartis Pharmaceuticals UK anuncia el lanzamiento de Novartis Biome UK Heart Health Catalyst 2022, en una asociación de inversores pionera a nivel mundial con Medtronic Ltd, RYSE Asset Management y Chelsea and Westminster Hospital NHS Foundation Trust y su organización benéfica oficial CW+.
Por lo tanto, se espera que un aumento en la colaboración y las asociaciones genere muchas oportunidades para que el mercado crezca.
Desafío
- Normas estrictas para los productos de terapia genética
El uso de la terapia génica está aumentando rápidamente en todo el mundo, debido al aumento de la población envejecida y a varias enfermedades crónicas que se pueden prevenir mediante un diagnóstico temprano y tratamientos oportunos. Al mismo tiempo, los actores de la terapia génica en el mercado deben cumplir ciertas regulaciones para obtener la aprobación de las autoridades superiores para el lanzamiento del producto en una región. Estas estrictas pautas deben cumplirse y esta es una de las tareas más difíciles entre todos los pasos. La aprobación previa a la comercialización de varios productos de terapia génica varía de un país a otro.
Por ejemplo,
- En 2022, según la información proporcionada por la Administración de Alimentos y Medicamentos (FDA), el Centro de Evaluación e Investigación Biológica (CBER) regula los productos de terapia celular, los productos de terapia génica humana y ciertos dispositivos relacionados con la terapia celular y génica. El CBER utiliza tanto la Ley del Servicio de Salud Pública como la Ley Federal de Alimentos, Medicamentos y Cosméticos como estatutos habilitantes para la supervisión.
Por lo tanto, las regulaciones estrictas para los productos de terapia genética varían según los distintos países, lo que se espera que represente un desafío para el crecimiento del mercado.
Acontecimientos recientes
- En diciembre de 2022, Kite Pharma, Inc. y Daiichi Sankyo Co., Ltd. anunciaron que el Ministerio de Salud, Trabajo y Bienestar de Japón (MHLW) había aprobado Yescarta (axicabtagene ciloleucel), una terapia de células T con receptor de antígeno quimérico (CAR), para el tratamiento inicial de pacientes con linfoma de células B grandes recidivante/refractario (LBCL R/R): linfoma difuso de células B grandes, linfoma mediastínico primario de células B grandes, linfoma folicular transformado y linfoma de células B de alto grado. Solo los pacientes que no hayan recibido previamente una transfusión de células T CAR dirigidas contra el antígeno CD19 deben ser tratados con Yescarta.
- En diciembre, Ferring Pharmaceuticals anunció que la Administración de Alimentos y Medicamentos de los Estados Unidos (FDA) había aprobado Adstiladrin (nadofaragene firadenovec-vncg), una nueva terapia génica basada en un vector de adenovirus, para el tratamiento de pacientes adultos con cáncer de vejiga no músculo invasivo (NMIBC) de alto riesgo que no responde al bacilo de Calmette-Guérin (BCG) y que presenta carcinoma in situ (CIS) con o sin tumores papilares. Esto ha ayudado a la empresa a ampliar su cartera de productos.
Alcance del mercado de terapia génica en Asia y el Pacífico
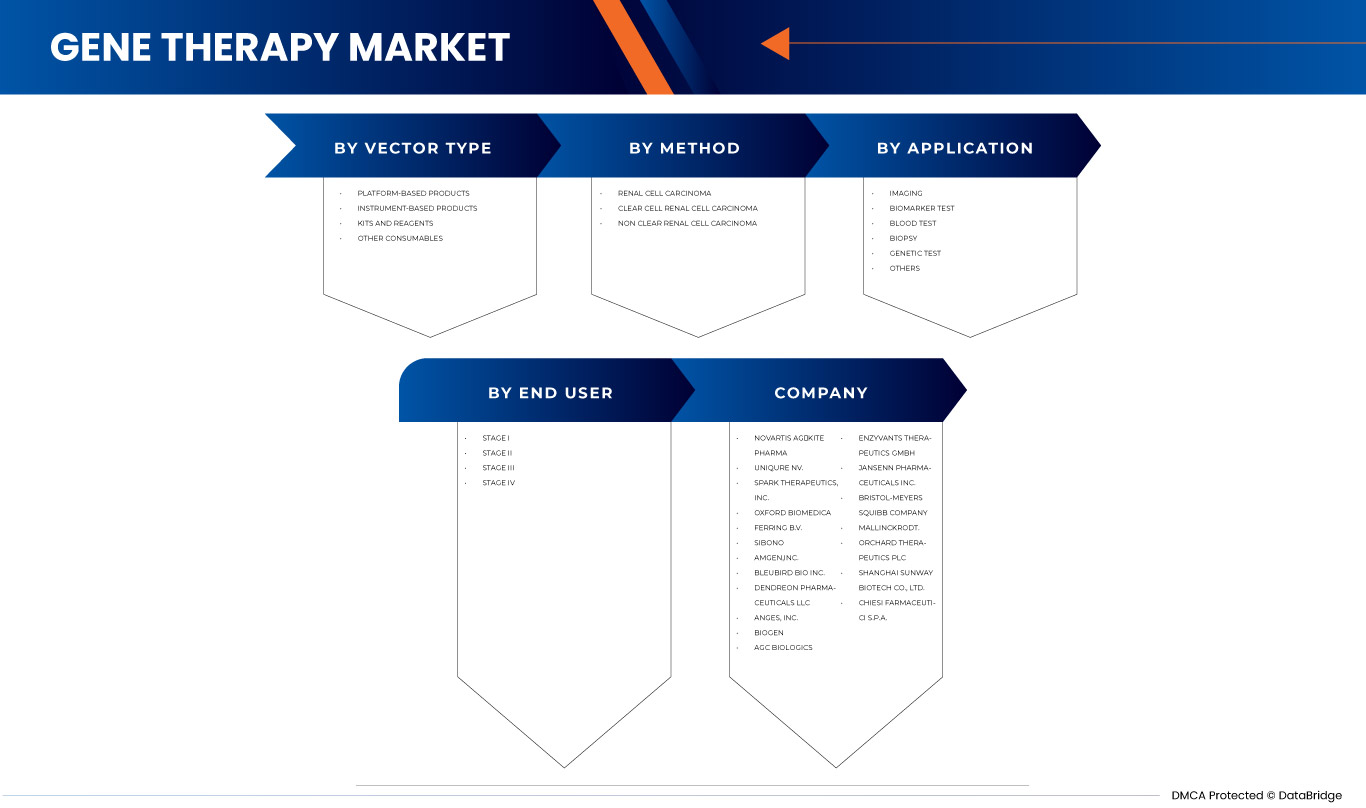
El mercado de terapia génica de Asia-Pacífico está segmentado en cuatro segmentos notables según el tipo de vector, el método, la aplicación y el usuario final. El crecimiento entre segmentos le ayuda a analizar nichos de crecimiento y estrategias para abordar el mercado y determinar sus áreas de aplicación principales y la diferencia en sus mercados objetivo.
POR TIPO DE VECTOR
- Vector viral
- Vector no viral
Según el tipo de vector, el mercado de terapia génica de Asia y el Pacífico está segmentado en vectores virales y no virales.
POR MÉTODO
- Ex vivo
- In vivo
Sobre la base del método, el mercado de terapia génica de Asia y el Pacífico está segmentado en ex vivo e in vivo.
POR APLICACIÓN
- Trastornos oncológicos
- Enfermedades cardiovasculares
- Enfermedad infecciosa
- Enfermedades raras
- Trastornos neurológicos
- Otras enfermedades
Sobre la base de la aplicación, el mercado de terapia génica de Asia-Pacífico está segmentado en trastornos oncológicos, enfermedades cardiovasculares, enfermedades infecciosas, enfermedades raras, trastornos neurológicos y otras enfermedades.
POR USUARIO FINAL
- Institutos del cáncer
- Hospitales
- Institutos de investigación
- Otros
Sobre la base del usuario final, el mercado de terapia genética de Asia-Pacífico está segmentado en institutos de cáncer, hospitales, institutos de investigación y otros.
Análisis y perspectivas regionales del mercado de terapia génica en Asia y el Pacífico
El mercado de terapia génica de Asia y el Pacífico está segmentado en cuatro segmentos notables según el tipo de vector, el método, la aplicación y el usuario final.
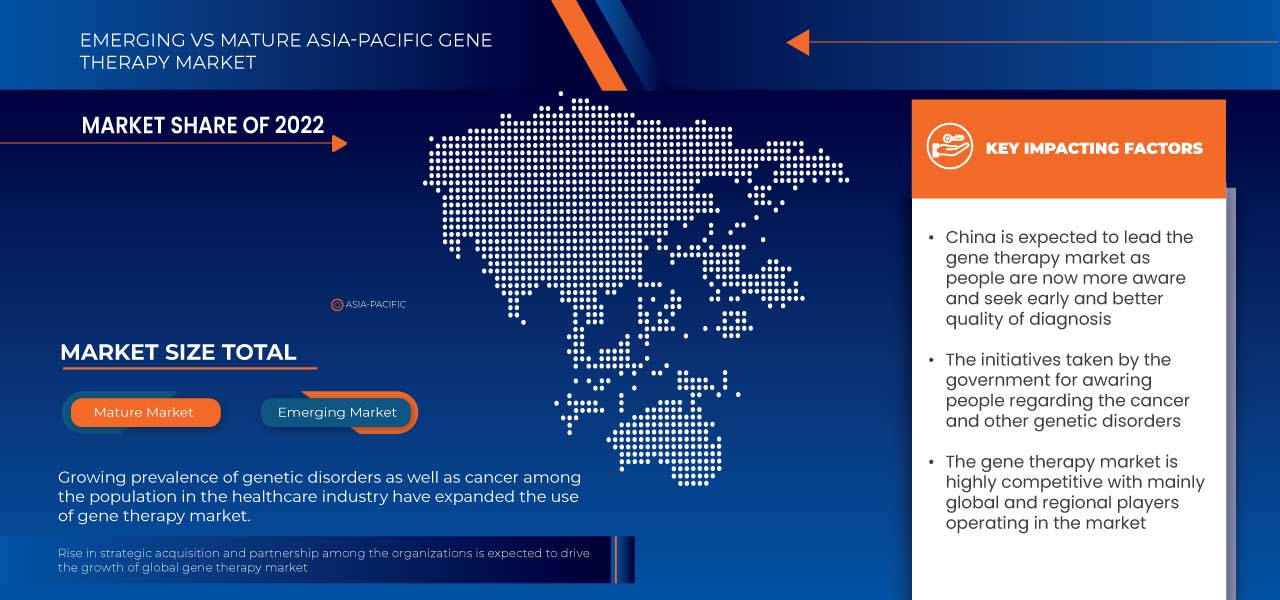
Los países cubiertos en este informe de mercado son China, Japón, India, Australia, Corea del Sur, Singapur, Malasia, Tailandia, Indonesia, Filipinas y el resto de Asia-Pacífico.
China domina debido a la presencia de actores clave en el mercado de consumo más grande y con un PIB elevado.
La sección de países del informe también proporciona factores de impacto individuales en el mercado y cambios en la regulación en el mercado a nivel nacional que afectan las tendencias actuales y futuras del mercado. Los puntos de datos como nuevas ventas, ventas de reemplazo, demografía del país, leyes regulatorias y aranceles de importación y exportación son algunos de los principales indicadores utilizados para pronosticar el escenario del mercado para países individuales. Además, se consideran la presencia y disponibilidad de marcas de Asia-Pacífico y los desafíos que enfrentan debido a la competencia grande o escasa de las marcas locales y nacionales, y el impacto de los canales de venta al proporcionar un análisis de pronóstico de los datos del país.
Análisis del panorama competitivo y de la cuota de mercado de la terapia génica en Asia y el Pacífico
El panorama competitivo del mercado de terapia génica de Asia-Pacífico proporciona detalles por competidor. Los detalles incluidos son una descripción general de la empresa, las finanzas de la empresa, los ingresos generados, el potencial de mercado, la inversión en I+D, las nuevas iniciativas de mercado, los sitios e instalaciones de producción, las fortalezas y debilidades de la empresa, el lanzamiento de productos, las aprobaciones de productos, la amplitud y el alcance de los productos, el dominio de las aplicaciones y la curva de vida de los tipos de productos. Los puntos de datos anteriores proporcionados solo están relacionados con el enfoque de la empresa en el mercado de terapia génica de Asia-Pacífico.
Algunos de los principales actores que operan en el mercado global de terapia génica son: Novartis AG, Kite Pharma (una subsidiaria de Gilead Sciences, Inc.), uniQure NV, Oxford Biomedica, Spark Therapeutics, Inc., SIBONO, bluebird bio, Inc., Shanghai Sunway Biotech Co. Ltd., Biogen, Dendreon Pharmaceuticals LLC., Amgen Inc., AnGes, Inc. y Enzyvant Therapeutics GmbHAudubon Bioscience, entre otros.
SKU-
Obtenga acceso en línea al informe sobre la primera nube de inteligencia de mercado del mundo
- Panel de análisis de datos interactivo
- Panel de análisis de empresas para oportunidades con alto potencial de crecimiento
- Acceso de analista de investigación para personalización y consultas
- Análisis de la competencia con panel interactivo
- Últimas noticias, actualizaciones y análisis de tendencias
- Aproveche el poder del análisis de referencia para un seguimiento integral de la competencia
Tabla de contenido
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF THE ASIA PACIFIC GENE THERAPY MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATIONS
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 DBMR TRIPOD DATA VALIDATION MODEL
2.5 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.6 MULTIVARIATE MODELLING
2.7 MARKET END USER COVERAGE GRID
2.8 PRODUCT LIFELINE CURVE
2.9 DBMR MARKET POSITION GRID
2.1 VENDOR SHARE ANALYSIS
2.11 SECONDARY SOURCES
2.12 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PESTEL ANALYSIS
4.2 PORTER FIVE ANALYSIS
5 UPDATE ON GERMLINE GENE THERAPY
5.1 GERMLINE GENE THERAPY
6 ASIA PACIFIC GENE THERAPY MARKET, MO
6.1 DRIVERS
6.1.1 NOVEL APPROACHES TO GENE THERAPY
6.1.2 INCREASING PREVALENCE OF GENETIC DISORDERS
6.1.3 THE GROWING INVESTMENT BY BIOTECHNOLOGY & PHARMACEUTICAL COMPANIES
6.1.4 GROWING DEMAND FOR PERSONALIZED MEDICINE
6.2 RESTRAINTS
6.2.1 HIGH COST OF GENE THERAPY
6.2.2 ETHICAL AND SAFETY CONCERNS
6.2.3 COMPLEXITY OF GENE THERAPY
6.3 OPPORTUNITIES
6.3.1 RISE IN STRATEGIC ACQUISITION AND PARTNERSHIP AMONG ORGANIZATIONS
6.3.2 RISING APPROVAL FOR GENE THERAPY PRODUCTS
6.4 CHALLENGES
6.4.1 STRINGENT REGULATIONS FOR GENE THERAPY PRODUCTS
6.4.2 LONG-TERM SAFETY AND EFFICACY
7 ASIA PACIFIC GENE THERAPY MARKET, BY VECTOR TYPE
7.1 OVERVIEW
7.2 VIRAL VECTOR
7.2.1 ADENOVIRUS
7.2.2 RETROVIRUS
7.2.3 LENTIVIRUS
7.2.4 ADENO-ASSOCIATED VIRUS
7.2.5 VACCINIA VIRUS
7.2.6 HERPES SIMPLEX VIRUS
7.2.7 OTHERS
7.3 NON-VIRAL VECTOR
7.3.1 LIPOFECTION
7.3.2 INJECTION OF NAKED DNA
8 ASIA PACIFIC GENE THERAPY MARKET, BY METHOD
8.1 OVERVIEW
8.2 EX-VIVO
8.3 IN-VIVO
9 ASIA PACIFIC GENE THERAPY MARKET, BY APPLICATION
9.1 OVERVIEW
9.2 ONCOLOGICAL DISORDERS
9.3 CARDIOVASCULAR DISEASES
9.4 INFECTIOUS DISEASES
9.5 RARE DISEASES
9.6 NUEROLOGICAL DISORDERS
9.7 OTHER DISEASES
10 ASIA PACIFIC GENE THERAPY MARKET, BY END USER
10.1 OVERVIEW
10.2 CANCER INSTITUTES
10.3 HOSPITALS
10.4 RESEARCH INSTITUTES
10.5 OTHERS
11 ASIA PACIFIC GENE THERAPY MARKET, BY REGION
11.1 ASIA-PACIFIC
11.1.1 CHINA
11.1.2 JAPAN
11.1.3 SOUTH KOREA
11.1.4 INDIA
11.1.5 AUSTRALIA
11.1.6 SINGAPORE
11.1.7 THAILAND
11.1.8 MALAYSIA
11.1.9 INDONESIA
11.1.10 PHILIPPINES
11.1.11 REST OF ASIA-PACIFIC
12 ASIA PACIFIC GENE THERAPY MARKET, COMPANY LANDSCAPE
12.1 COMPANY SHARE ANALYSIS: ASIA PACIFIC
13 COMPANY PROFILES
13.1 BIOGEN
13.1.1 COMPANY SNAPSHOT
13.1.2 REVENUE ANALYSIS
13.1.3 COMPANY SHARE ANALYSIS
13.1.4 SWOT ANALYSIS
13.1.5 PRODUCT PORTFOLIO
13.1.6 RECENT DEVELOPMENT
13.2 KITE PHARMA
13.2.1 COMPANY SNAPSHOT
13.2.2 REVENUE ANALYSIS
13.2.3 COMPANY SHARE ANALYSIS
13.2.4 SWOT ANALYSIS
13.2.5 PRODUCT PORTFOLIO
13.2.6 RECENT DEVELOPMENT
13.3 NOVARTIS AG
13.3.1 COMPANY SNAPSHOT
13.3.2 REVENUE ANALYSIS
13.3.3 COMPANY SHARE ANALYSIS
13.3.4 SWOT ANALYSIS
13.3.5 PRODUCT PORTFOLIO
13.3.6 RECENT DEVELOPMENTS
13.4 BRISTOL-MYERS SQUIBB COMPANY.
13.4.1 COMPANY SNAPSHOT
13.4.2 REVENUE ANALYSIS
13.4.3 COMPANY SHARE ANALYSIS
13.4.4 SWOT ANALYSIS
13.4.5 PRODUCT PORTFOLIO
13.4.6 RECENT DEVELOPMENT
13.5 OXFORD BIOMEDICA
13.5.1 COMPANY SNAPSHOT
13.5.2 REVENUE ANALYSIS
13.5.3 COMPANY SHARE ANALYSIS
13.5.4 SWOT ANALYSIS
13.5.5 PRODUCT PORTFOLIO
13.5.6 RECENT DEVELOPMENTS
13.6 AGC BIOLOGICS
13.6.1 COMPANY SNAPSHOT
13.6.2 PRODUCT PORTFOLIO
13.6.3 RECENT DEVELOPMENT
13.7 ANGES, INC
13.7.1 COMPANY SNAPSHOT
13.7.2 REVENUE ANALYSIS
13.7.3 PRODUCT PORTFOLIO
13.7.4 RECENT DEVELOPMENT
13.8 AMGEN INC.
13.8.1 COMPANY SNAPSHOT
13.8.2 REVENUE ANALYSIS
13.8.3 PRODUCT PORTFOLIO
13.8.4 RECENT DEVELOPMENT
13.9 BLUEBIRD BIO, INC.
13.9.1 COMPANY SNAPSHOT
13.9.2 PRODUCT PORTFOLIO
13.9.3 RECENT DEVELOPMENT
13.1 CHIESI FARMACEUTICI S.P.A
13.10.1 COMPANY SNAPSHOT
13.10.2 REVENUE ANALYSIS
13.10.3 PRODUCT PORTFOLIO
13.10.4 RECENT DEVELOPMENT
13.11 DENDREON PHARMACEUTICALS LLC
13.11.1 COMPANY SNAPSHOT
13.11.2 PRODUCT PORTFOLIO
13.11.3 RECENT DEVELOPMENT
13.12 ENZYVANT THERAPEUTICS GMBH
13.12.1 COMPANY SNAPSHOT
13.12.2 RODUCT PORTFOLIO
13.12.3 RECENT DEVELOPMENT
13.13 FERRING B.V.
13.13.1 COMPANY SNAPSHOT
13.13.2 PRODUCT PORTFOLIO
13.13.3 RECENT DEVELOPMENT
13.14 JANSSEN PHARMACEUTICALS, INC.
13.14.1 COMPANY SNAPSHOT
13.14.2 PRODUCT PORTFOLIO
13.14.3 RECENT DEVELOPMENT
13.15 MALLINCKRODT.
13.15.1 COMPANY SNAPSHOT
13.15.2 REVENUE ANALYSIS
13.15.3 PRODUCT PORTFOLIO
13.15.4 RECENT DEVELOPMENT
13.16 ORCHARD THERAPEUTICS PLC.
13.16.1 COMPANY SNAPSHOT
13.16.2 REVENUE ANALYSIS
13.16.3 PRODUCT PORTFOLIO
13.16.4 RECENT DEVELOPMENT
13.17 SHANGHAI SUNWAY BIOTECH CO., LTD.
13.17.1 COMPANY SNAPSHOT
13.17.2 PRODUCT PORTFOLIO
13.17.3 RECENT DEVELOPMENT
13.18 SIBONO
13.18.1 COMPANY SNAPSHOT
13.18.2 PRODUCT PORTFOLIO
13.18.3 RECENT DEVELOPMENT
13.19 SPARK THERAPEUTICS, INC.
13.19.1 COMPANY SNAPSHOT
13.19.2 PRODUCT PORTFOLIO
13.19.3 RECENT DEVELOPMENT
13.2 UNIQURE NV.
13.20.1 COMPANY SNAPSHOT
13.20.2 REVENUE ANALYSIS
13.20.3 PRODUCT PORTFOLIO
13.20.4 RECENT DEVELOPMENT
14 QUESTIONNAIRE
15 RELATED REPORTS
Lista de Tablas
TABLE 1 ASIA PACIFIC GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 2 ASIA PACIFIC VIRAL VECTOR IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 3 ASIA PACIFIC VIRAL VECTOR IN GENE THERAPY MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 4 ASIA PACIFIC NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 5 ASIA PACIFIC NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 6 ASIA PACIFIC GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 7 ASIA PACIFIC EX-VIVO IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 8 ASIA PACIFIC IN –VIVO IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 9 ASIA PACIFIC GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 10 ASIA PACIFIC ONCOLOGICAL DISORDERS IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 11 ASIA PACIFIC CARDIOVASCULAR DISEASES IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 12 ASIA PACIFIC INFECTIOUS DISEASES IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 13 ASIA PACIFIC RARE DISEASES IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 14 ASIA PACIFIC NEUROLOGICAL DISORDERS IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 15 ASIA PACIFIC OTHER DISEASES IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 16 ASIA PACIFIC GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 17 ASIA PACIFIC CANCER INSTITUTES IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 18 ASIA PACIFIC HOSPITALS IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 19 ASIA PACIFIC RESEARCH INSTITUTES IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 20 ASIA PACIFIC OTHERS IN GENE THERAPY MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 21 ASIA-PACIFIC GENE THERAPY MARKET, BY COUNTRY, 2021-2030 (USD MILLION)
TABLE 22 ASIA-PACIFIC GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 23 ASIA-PACIFIC VIRAL VECTOR IN GENE THERAPY MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 24 ASIA-PACIFIC NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 25 ASIA-PACIFIC GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 26 ASIA-PACIFIC GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 27 ASIA-PACIFIC GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 28 CHINA GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 29 CHINA VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 30 CHINA NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 31 CHINA GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 32 CHINA GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 33 CHINA GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 34 JAPAN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 35 JAPAN VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 36 JAPAN NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 37 JAPAN GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 38 JAPAN GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 39 JAPAN GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 40 SOUTH KOREA GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 41 SOUTH KOREA VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 42 SOUTH KOREA NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 43 SOUTH KOREA GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 44 SOUTH KOREA GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 45 SOUTH KOREA GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 46 INDIA GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 47 INDIA VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 48 INDIA NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 49 INDIA GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 50 INDIA GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 51 INDIA GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 52 AUSTRALIA GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 53 AUSTRALIA VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 54 AUSTRALIA NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 55 AUSTRALIA GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 56 AUSTRALIA GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 57 AUSTRALIA GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 58 SINGAPORE GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 59 SINGAPORE VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 60 SINGAPORE NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 61 SINGAPORE GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 62 SINGAPORE GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 63 SINGAPORE GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 64 THAILAND GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 65 THAILAND VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 66 THAILAND NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 67 THAILAND GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 68 THAILAND GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 69 THAILAND GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 70 MALAYSIA GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 71 MALAYSIA VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 72 MALAYSIA NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 73 MALAYSIA GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 74 MALAYSIA GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 75 MALAYSIA GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 76 INDONESIA GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 77 INDONESIA VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 78 INDONESIA NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 79 INDONESIA GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 80 INDONESIA GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 81 INDONESIA GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 82 PHILIPPINES GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 83 PHILIPPINES VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 84 PHILIPPINES NON-VIRAL VECTOR IN GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
TABLE 85 PHILIPPINES GENE THERAPY MARKET, BY METHOD, 2021-2030 (USD MILLION)
TABLE 86 PHILIPPINES GENE THERAPY MARKET, BY APPLICATION, 2021-2030 (USD MILLION)
TABLE 87 PHILIPPINES GENE THERAPY MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 88 REST OF ASIA-PACIFIC GENE THERAPY MARKET, BY VECTOR TYPE, 2021-2030 (USD MILLION)
Lista de figuras
FIGURE 1 ASIA PACIFIC GENE THERAPY MARKET: SEGMENTATION
FIGURE 2 ASIA PACIFIC GENE THERAPY MARKET: DATA TRIANGULATION
FIGURE 3 ASIA PACIFIC GENE THERAPY MARKET: DROC ANALYSIS
FIGURE 4 ASIA PACIFIC GENE THERAPY MARKET: ASIA PACIFIC VS REGIONAL MARKET ANALYSIS
FIGURE 5 ASIA PACIFIC GENE THERAPY MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 ASIA PACIFIC GENE THERAPY MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 ASIA PACIFIC GENE THERAPY MARKET: MARKET END USER COVERAGE GRID
FIGURE 8 ASIA PACIFIC GENE THERAPY MARKET: DBMR MARKET POSITION GRID
FIGURE 9 ASIA PACIFIC GENE THERAPY MARKET: VENDOR SHARE ANALYSIS
FIGURE 10 ASIA PACIFIC GENE THERAPY MARKET: SEGMENTATION
FIGURE 11 THE INCREASING PREVALENCE OF GENETIC DISORDERS AND GROWING DEMAND FOR PERSONALIZED MEDICINE ARE EXPECTED TO DRIVE THE GROWTH OF THE ASIA PACIFIC GENE THERAPY MARKET FROM 2023 TO 2030
FIGURE 12 THE VIRAL VECTOR SEGMENT IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE ASIA PACIFIC GENE THERAPY MARKET IN 2023 & 2030
FIGURE 13 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF THE ASIA PACIFIC GENE THERAPY MARKET
FIGURE 14 ASIA PACIFIC GENE THERAPY MARKET: BY VECTOR TYPE, 2022
FIGURE 15 ASIA PACIFIC GENE THERAPY MARKET: BY VECTOR TYPE, 2023-2030 (USD MILLION)
FIGURE 16 ASIA PACIFIC GENE THERAPY MARKET: BY VECTOR TYPE, CAGR (2023-2030)
FIGURE 17 ASIA PACIFIC GENE THERAPY MARKET: BY VECTOR TYPE, LIFELINE CURVE
FIGURE 18 ASIA PACIFIC GENE THERAPY MARKET: BY METHOD, 2022
FIGURE 19 ASIA PACIFIC GENE THERAPY MARKET: BY METHOD, 2023-2030 (USD MILLION)
FIGURE 20 ASIA PACIFIC GENE THERAPY MARKET: BY METHOD, CAGR (2023-2030)
FIGURE 21 ASIA PACIFIC GENE THERAPY MARKET: BY METHOD, LIFELINE CURVE
FIGURE 22 ASIA PACIFIC GENE THERAPY MARKET: BY APPLICATION, 2022
FIGURE 23 ASIA PACIFIC GENE THERAPY MARKET: BY APPLICATION, 2023-2030 (USD MILLION)
FIGURE 24 ASIA PACIFIC GENE THERAPY MARKET: BY APPLICATION, CAGR (2023-2030)
FIGURE 25 ASIA PACIFIC GENE THERAPY MARKET: BY APPLICATION, LIFELINE CURVE
FIGURE 26 ASIA PACIFIC GENE THERAPY MARKET: BY END USER, 2022
FIGURE 27 ASIA PACIFIC GENE THERAPY MARKET: BY END USER, 2023-2030 (USD MILLION)
FIGURE 28 ASIA PACIFIC GENE THERAPY MARKET: BY END USER, CAGR (2023-2030)
FIGURE 29 ASIA PACIFIC GENE THERAPY MARKET: BY END USER, LIFELINE CURVE
FIGURE 30 ASIA-PACIFIC GENE THERAPY MARKET: SNAPSHOT (2022)
FIGURE 31 ASIA-PACIFIC GENE THERAPY MARKET: BY COUNTRY (2022)
FIGURE 32 ASIA-PACIFIC GENE THERAPY MARKET: BY COUNTRY (2023 & 2030)
FIGURE 33 ASIA-PACIFIC GENE THERAPY MARKET: BY COUNTRY (2022 & 2030)
FIGURE 34 ASIA-PACIFIC GENE THERAPY MARKET: VECTOR TYPE (2023-2030)
FIGURE 35 ASIA PACIFIC GENE THERAPY MARKET: COMPANY SHARE 2022 (%)

Metodología de investigación
La recopilación de datos y el análisis del año base se realizan utilizando módulos de recopilación de datos con muestras de gran tamaño. La etapa incluye la obtención de información de mercado o datos relacionados a través de varias fuentes y estrategias. Incluye el examen y la planificación de todos los datos adquiridos del pasado con antelación. Asimismo, abarca el examen de las inconsistencias de información observadas en diferentes fuentes de información. Los datos de mercado se analizan y estiman utilizando modelos estadísticos y coherentes de mercado. Además, el análisis de la participación de mercado y el análisis de tendencias clave son los principales factores de éxito en el informe de mercado. Para obtener más información, solicite una llamada de un analista o envíe su consulta.
La metodología de investigación clave utilizada por el equipo de investigación de DBMR es la triangulación de datos, que implica la extracción de datos, el análisis del impacto de las variables de datos en el mercado y la validación primaria (experto en la industria). Los modelos de datos incluyen cuadrícula de posicionamiento de proveedores, análisis de línea de tiempo de mercado, descripción general y guía del mercado, cuadrícula de posicionamiento de la empresa, análisis de patentes, análisis de precios, análisis de participación de mercado de la empresa, estándares de medición, análisis global versus regional y de participación de proveedores. Para obtener más información sobre la metodología de investigación, envíe una consulta para hablar con nuestros expertos de la industria.
Personalización disponible
Data Bridge Market Research es líder en investigación formativa avanzada. Nos enorgullecemos de brindar servicios a nuestros clientes existentes y nuevos con datos y análisis que coinciden y se adaptan a sus objetivos. El informe se puede personalizar para incluir análisis de tendencias de precios de marcas objetivo, comprensión del mercado de países adicionales (solicite la lista de países), datos de resultados de ensayos clínicos, revisión de literatura, análisis de mercado renovado y base de productos. El análisis de mercado de competidores objetivo se puede analizar desde análisis basados en tecnología hasta estrategias de cartera de mercado. Podemos agregar tantos competidores sobre los que necesite datos en el formato y estilo de datos que esté buscando. Nuestro equipo de analistas también puede proporcionarle datos en archivos de Excel sin procesar, tablas dinámicas (libro de datos) o puede ayudarlo a crear presentaciones a partir de los conjuntos de datos disponibles en el informe.