Global Pharmaceutical Excipients Market
Marktgröße in Milliarden USD
CAGR :
% 
 USD
8.85 Billion
USD
14.77 Billion
2024
2032
USD
8.85 Billion
USD
14.77 Billion
2024
2032
| 2025 –2032 | |
| USD 8.85 Billion | |
| USD 14.77 Billion | |
|
|
|
|
Globale Marktsegmentierung für pharmazeutische Hilfsstoffe nach Funktionalität (Binde- und Klebstoffe, Sprengmittel, Beschichtungsmaterial, Farbstoffe, Lösungsvermittler, Aromen, Süßstoffe, Verdünnungsmittel, Schmiermittel, Puffer, Emulgatoren, Konservierungsmittel, Antioxidantien, Sorbentien, Lösungsmittel, Erweichungsmittel, Gleitmittel, Chelatbildner, Entschäumer und andere), Darreichungsform (fest, halbfest und flüssig), Verabreichungsweg (orale Hilfsstoffe, topische Hilfsstoffe, parenterale Hilfsstoffe und andere Hilfsstoffe), Endverbraucher (Pharma- und Biopharmaunternehmen, Auftragsformulierer, Forschungsorganisationen und Akademiker und andere), Vertriebskanal (Direktausschreibung, Einzelhandel und andere) – Branchentrends und Prognose bis 2032
Marktgröße für pharmazeutische Hilfsstoffe
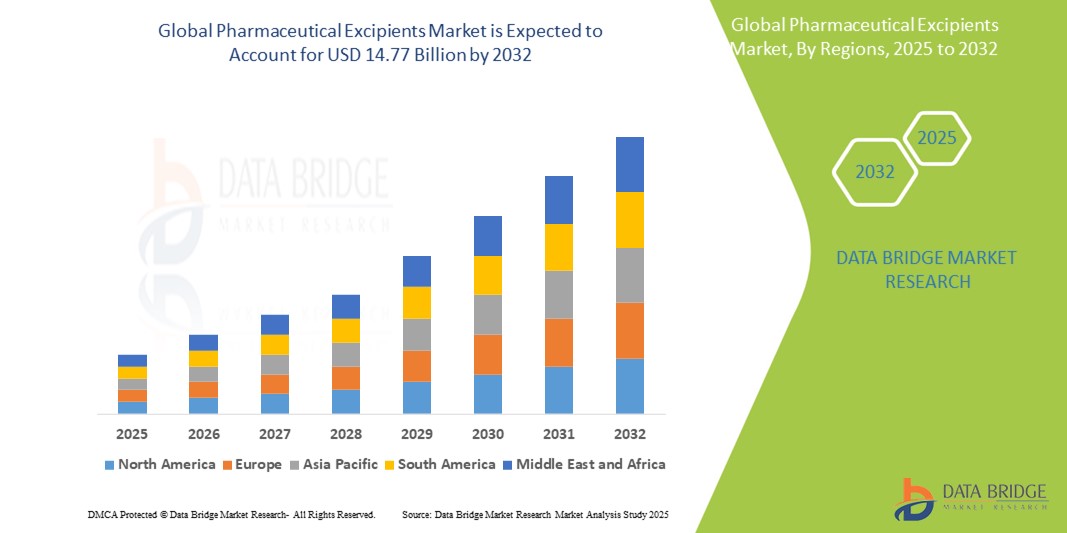
- Der globale Markt für pharmazeutische Hilfsstoffe wird im Jahr 2024 auf 8,85 Milliarden US-Dollar geschätzt und soll bis 2032 14,77 Milliarden US-Dollar erreichen , bei einer CAGR von 6,60 % im Prognosezeitraum.
- Dieses Wachstum wird durch Faktoren wie die Expansion der Pharmaindustrie, die Zunahme der Entwicklung von Generika und technologische Fortschritte bei Arzneimittelverabreichungssystemen vorangetrieben.
Marktanalyse für pharmazeutische Hilfsstoffe
- Pharmazeutische Hilfsstoffe sind essentielle, nicht-aktive Substanzen, die in Arzneimittelformulierungen verwendet werden, um Stabilität, Bioverfügbarkeit und Patientencompliance zu verbessern. Sie spielen eine entscheidende Rolle bei der Arzneimittelherstellung und dienen als Bindemittel, Füllstoffe, Beschichtungen und Stabilisatoren.
- Die Nachfrage nach pharmazeutischen Hilfsstoffen wird maßgeblich durch die steigende Prävalenz chronischer Krankheiten, die zunehmende Produktion von Generika und Fortschritte bei der Arzneimittelverabreichung getrieben.
- Nordamerika dominiert den Markt für pharmazeutische Hilfsstoffe mit einem Anteil von 39,8 % am globalen Markt für pharmazeutische Hilfsstoffe. Dies ist auf die fortschrittlichen pharmazeutischen Produktionskapazitäten, die hohen Gesundheitsausgaben und die starke Präsenz wichtiger Marktteilnehmer zurückzuführen.
- Der asiatisch-pazifische Markt hält einen Anteil von 30,4 % am globalen Markt für pharmazeutische Hilfsstoffe, angetrieben durch die schnelle Expansion der Arzneimittelproduktion, steigende Gesundheitsausgaben und die wachsende Nachfrage nach kostengünstigen Arzneimittelformulierungen
- Es wird erwartet, dass Bindemittel und Klebstoffe den Markt für pharmazeutische Hilfsstoffe mit einem Anteil von 56,72 % im Jahr 2023 dominieren werden, was auf ihre entscheidende Rolle bei der Formulierung von Tabletten und Kapseln zurückzuführen ist. Diese Hilfsstoffe sorgen für strukturelle Integrität und einen einheitlichen Wirkstoffgehalt und unterstützen so konsistente therapeutische Ergebnisse.
Berichtsumfang und Marktsegmentierung für pharmazeutische Hilfsstoffe
|
Eigenschaften |
Wichtige Markteinblicke zu pharmazeutischen Hilfsstoffen |
|
Abgedeckte Segmente |
|
|
Abgedeckte Länder |
Nordamerika
Europa
Asien-Pazifik
Naher Osten und Afrika
Südamerika
|
|
Wichtige Marktteilnehmer |
|
|
Marktchancen |
|
|
Wertschöpfungsdaten-Infosets |
Zusätzlich zu den Einblicken in Marktszenarien wie Marktwert, Wachstumsrate, Segmentierung, geografische Abdeckung und wichtige Akteure enthalten die von Data Bridge Market Research kuratierten Marktberichte auch Import-Export-Analysen, eine Übersicht über die Produktionskapazität, eine Analyse des Produktionsverbrauchs, eine Preistrendanalyse, ein Szenario des Klimawandels, eine Lieferkettenanalyse, eine Wertschöpfungskettenanalyse, eine Übersicht über Rohstoffe/Verbrauchsmaterialien, Kriterien für die Lieferantenauswahl, eine PESTLE-Analyse, eine Porter-Analyse und regulatorische Rahmenbedingungen. |
Markttrends für pharmazeutische Hilfsstoffe
„Innovationen bei pharmazeutischen Hilfsstoffen für eine fortschrittliche Arzneimittelverabreichung“
- Ein wichtiger Trend auf dem Markt für pharmazeutische Hilfsstoffe ist die zunehmende Konzentration auf multifunktionale Hilfsstoffe, die fortschrittliche Arzneimittelverabreichungssysteme unterstützen, darunter kontrollierte Freisetzung, gezielte Verabreichung und oral zerfallende Formulierungen.
- Diese innovativen Hilfsstoffe verbessern die Arzneimittelstabilität, Bioverfügbarkeit und Patientencompliance und spielen eine entscheidende Rolle bei der Formulierung komplexer Arzneimittel, Biologika und personalisierter Medikamente.
- Beispielsweise verbessern Hilfsstoffe wie Cyclodextrine die Löslichkeit und Stabilität schlecht wasserlöslicher Arzneimittel, während Hilfsstoffe auf Lipidbasis die Freisetzung hoch lipophiler Verbindungen unterstützen und so eine effiziente Absorption und therapeutische Wirkung ermöglichen.
- Diese Fortschritte verändern die Arzneimittelentwicklung und ermöglichen es Pharmaunternehmen, Formulierungsprobleme zu überwinden, therapeutische Ergebnisse zu verbessern und die Nachfrage nach neuartigen, leistungsstarken Hilfsstoffen anzukurbeln.
Marktdynamik für pharmazeutische Hilfsstoffe
Treiber
„Steigende Nachfrage nach innovativen Drug-Delivery-Systemen“
- Der zunehmende Fokus auf patientenzentrierte Therapien und personalisierte Medizin treibt die Nachfrage nach fortschrittlichen pharmazeutischen Hilfsstoffen, die innovative Arzneimittelverabreichungssysteme unterstützen, wie z. B. kontrollierte Freisetzung, gezielte Verabreichung und im Mund zerfallende Tabletten, erheblich an.
- Da Pharmaunternehmen bestrebt sind, die Therapieergebnisse und die Patienten-Compliance zu verbessern, steigt der Bedarf an Hilfsstoffen, die die Bioverfügbarkeit, Stabilität und Löslichkeit von pharmazeutischen Wirkstoffen (APIs) verbessern können.
- Darüber hinaus treibt der Trend zu Biologika und komplexen Arzneimittelformulierungen, die oft spezielle Hilfsstoffe erfordern, das Marktwachstum weiter voran.
Zum Beispiel,
- Laut einem Bericht der Pharmaceutical Research and Manufacturers of America (PhRMA) aus dem Jahr 2024 hat sich die Zahl der in der Entwicklung befindlichen Biologika im letzten Jahrzehnt mehr als verdoppelt. Dies führt zu einer erheblichen Nachfrage nach neuartigen Hilfsstoffen, die die besonderen Herausforderungen biologischer Formulierungen bewältigen können.
- Da die Pharmaindustrie weiterhin Innovationen hervorbringt, wird die Nachfrage nach fortschrittlichen Hilfsstoffen, die diese hochmodernen Therapien unterstützen können, voraussichtlich deutlich steigen.
Gelegenheit
„Expansion in Schwellenmärkte mit schnellem pharmazeutischem Wachstum“
- Schwellenländer, insbesondere im asiatisch-pazifischen Raum und in Lateinamerika, bieten Herstellern pharmazeutischer Hilfsstoffe erhebliche Chancen. Sie werden durch schnelles Wirtschaftswachstum, steigende Gesundheitsausgaben und den Ausbau der pharmazeutischen Produktionskapazitäten angetrieben.
- In diesen Regionen steigt die Nachfrage nach kostengünstigen Generika, rezeptfreien Medikamenten und Biologika stark an. Dadurch entsteht ein attraktiver Markt für Hilfsstoffe, die den unterschiedlichen Formulierungsanforderungen gerecht werden können.
- Darüber hinaus dürften eine günstige Regierungspolitik, ein steigendes Gesundheitsbewusstsein und wachsende Investitionen in die lokale Arzneimittelproduktion die Nachfrage nach Hilfsstoffen weiter ankurbeln.
Zum Beispiel,
- Laut einem Bericht der Indian Pharmaceutical Alliance vom Januar 2025 wird Indien voraussichtlich bis 2030 der drittgrößte Pharmamarkt der Welt sein, angetrieben von einer starken Inlandsnachfrage und wachsenden Exportmöglichkeiten. Dieses Wachstum wird die Nachfrage nach pharmazeutischen Hilfsstoffen für die Formulierung und Herstellung von Arzneimitteln deutlich erhöhen.
- Daher können Hersteller von Hilfsstoffen, die strategisch in diese schnell wachsenden Märkte expandieren, von steigenden Umsätzen und Marktanteilen profitieren.
Einschränkung/Herausforderung
„Strenge regulatorische Anforderungen und hohe Compliance-Kosten“
- Der Markt für pharmazeutische Hilfsstoffe steht vor erheblichen Herausforderungen im Zusammenhang mit strengen gesetzlichen Anforderungen an die Sicherheit, Qualität und Leistung der in Arzneimittelformulierungen verwendeten Hilfsstoffe.
- Diese Vorschriften, die sich je nach Region erheblich unterscheiden, erfordern oft umfangreiche Tests, Dokumentationen und Validierungen, was die Gesamtkosten und den Zeitaufwand für die Produktentwicklung und -vermarktung erhöht.
- Darüber hinaus erhöht die Notwendigkeit der Einhaltung der Guten Herstellungspraxis (GMP), der Arzneibuchstandards und anderer Qualitätszertifizierungen die finanzielle Belastung der Hersteller von Hilfsstoffen.
Zum Beispiel,
- Laut einem Bericht des International Pharmaceutical Excipients Council (IPEC) aus dem Jahr 2024 sind die Kosten für die Einhaltung gesetzlicher Vorschriften für Hersteller von Hilfsstoffen in den letzten fünf Jahren um über 30 % gestiegen, da die Aufsichtsbehörden weltweit ihre Aufsicht verschärfen, um die Patientensicherheit und die Wirksamkeit der Produkte zu gewährleisten.
- Dieser regulatorische Druck kann den Markteintritt kleinerer Akteure einschränken und die Gesamtkosten der Hilfsstoffproduktion erhöhen, was sich auf die Rentabilität und das Marktwachstum auswirkt.
Marktumfang für pharmazeutische Hilfsstoffe
Der Markt ist nach Funktionalität, Darreichungsform, Verabreichungsweg, Endverbraucher und Vertriebskanal segmentiert.
|
Segmentierung |
Untersegmentierung |
|
Nach Funktionalität |
|
|
Nach Darreichungsform |
|
|
Nach Verabreichungsweg |
|
|
Nach Endbenutzer |
|
|
Nach Vertriebskanal |
|
Im Jahr 2025 werden die Bindemittel und Klebstoffe voraussichtlich den Markt dominieren und den größten Anteil im Funktionalitätssegment haben.
Das Segment Bindemittel und Klebstoffe wird voraussichtlich den Markt für pharmazeutische Hilfsstoffe mit einem Anteil von 56,72 % dominieren, was auf ihre entscheidende Rolle in Tabletten- und Kapselformulierungen zurückzuführen ist. Diese Hilfsstoffe sorgen für strukturelle Integrität und einen einheitlichen Wirkstoffgehalt, was zu konsistenten Therapieergebnissen führt. Ihre breite Anwendung wird durch die hohe Nachfrage nach festen oralen Darreichungsformen weiter vorangetrieben, die aufgrund ihrer praktischen Anwendung, Stabilität und Patientencompliance bevorzugt werden und ihre Marktdominanz untermauern.
Es wird erwartet, dass die oralen Hilfsstoffe im Prognosezeitraum den größten Anteil am Markt für Verabreichungswege ausmachen werden
Im Jahr 2025 dominierte das Segment der oralen Hilfsstoffe den Markt für pharmazeutische Hilfsstoffe mit dem größten Marktanteil von 55,49 %, was auf die weit verbreitete Verwendung und die Präferenz der Patienten für die orale Verabreichung zurückzuführen ist. Diese Formulierungen werden aufgrund ihrer einfachen Verabreichung, Kosteneffizienz und hohen Patienten-Compliance bevorzugt. Hilfsstoffe dieser Kategorie sind unerlässlich, um die Bioverfügbarkeit, Stabilität und den Geschmack oraler Medikamente zu verbessern und so ihre Marktdominanz zu stärken.
Regionale Analyse des Marktes für pharmazeutische Hilfsstoffe
„Nordamerika hält den größten Anteil am Markt für pharmazeutische Hilfsstoffe“
- Nordamerika dominiert den Markt für pharmazeutische Hilfsstoffe mit einem Anteil von 39,8 % am globalen Markt für pharmazeutische Hilfsstoffe. Dies ist auf die fortschrittlichen pharmazeutischen Produktionskapazitäten, die hohen Gesundheitsausgaben und die starke Präsenz wichtiger Marktteilnehmer zurückzuführen.
- Die USA halten einen signifikanten Anteil von 39 % aufgrund ihrer gut etablierten Pharmaindustrie, umfangreicher Forschungs- und Entwicklungsaktivitäten und einer wachsenden Nachfrage nach innovativen Arzneimittelverabreichungssystemen, einschließlich kontrollierter Freisetzung und gezielter Formulierungen.
- Darüber hinaus stärken die Präsenz großer Pharmaunternehmen und strenge regulatorische Standards für Arzneimittelsicherheit und -wirksamkeit den Markt in der Region weiter.
- Der zunehmende Fokus auf Biologika, personalisierte Medizin und Spezialmedikamente trägt ebenfalls zur hohen Nachfrage nach hochwertigen Hilfsstoffen bei und stärkt die Marktführerschaft Nordamerikas.
„Im asiatisch-pazifischen Raum wird auf dem Markt für pharmazeutische Hilfsstoffe voraussichtlich die höchste durchschnittliche jährliche Wachstumsrate verzeichnet “
- Im asiatisch-pazifischen Raum wird mit einem Marktanteil von 30,4 % die höchste Wachstumsrate im Markt für pharmazeutische Hilfsstoffe erwartet. Dies ist auf die schnelle Expansion der Arzneimittelproduktion, steigende Gesundheitsausgaben und die wachsende Nachfrage nach kostengünstigen Arzneimittelformulierungen zurückzuführen.
- Länder wie China, Indien und Japan entwickeln sich zu Schlüsselmärkten, unterstützt durch eine günstige Regierungspolitik, eine große Patientenpopulation und eine zunehmende Verbreitung chronischer Krankheiten, die eine langfristige Behandlung erfordern
- Japan ist bekannt für seine hochmoderne pharmazeutische Forschung und fortschrittliche Technologien zur Arzneimittelherstellung und bleibt ein wichtiger Markt für hochwertige Hilfsstoffe, die innovative Arzneimittelformulierungen unterstützen.
- Indien wird voraussichtlich mit einer jährlichen Wachstumsrate von 5,1 % den höchsten Marktanteilszuwachs verzeichnen. Dies ist auf das schnelle Wachstum der Generikaindustrie, den Ausbau der Gesundheitsinfrastruktur und steigende Investitionen in die pharmazeutische Forschung und Entwicklung zurückzuführen und macht das Land zu einem wichtigen Akteur auf dem globalen Markt für Hilfsstoffe.
Marktanteil pharmazeutischer Hilfsstoffe
Die Wettbewerbslandschaft des Marktes liefert detaillierte Informationen zu den einzelnen Wettbewerbern. Zu den Details gehören Unternehmensübersicht, Unternehmensfinanzen, Umsatz, Marktpotenzial, Investitionen in Forschung und Entwicklung, neue Marktinitiativen, globale Präsenz, Produktionsstandorte und -anlagen, Produktionskapazitäten, Stärken und Schwächen des Unternehmens, Produkteinführung, Produktbreite und -umfang sowie Anwendungsdominanz. Die oben genannten Datenpunkte beziehen sich ausschließlich auf die Marktausrichtung der Unternehmen.
Die wichtigsten Marktführer auf dem Markt sind:
- Kerry Group plc (Irland)
- DFE Pharma (Deutschland)
- Cargill, Incorporated (USA)
- Pfanstiehl Inc. (USA)
- Colorcon Inc. (USA)
- MEGGLE Group GmbH (Deutschland)
- Omya International AG (Schweiz)
- Peter Greven GmbH & Co. KG (Deutschland)
- Ashland (USA)
- Evonik Industries AG (Deutschland)
- Dow (USA)
- Croda International Plc (Großbritannien)
- Roquette Frères (Frankreich)
- Die Lubrizol Corporation (USA)
- BASF (Deutschland)
- Avantor, Inc. (USA)
- BENEO GmbH (Deutschland)
- Chemiehandel (Indien)
Neueste Entwicklungen auf dem globalen Markt für pharmazeutische Hilfsstoffe
- Im November 2024 stellte Clariant auf der Fachmesse CPHI India 2024 acht neue leistungsstarke pharmazeutische Hilfsstoffe vor. Diese Hilfsstoffe unterstützen die Entwicklung sicherer und wirksamer Medikamente und eignen sich für Anwendungen wie empfindliche APIs, parenterale Formulierungen und Formulierungen, die farblose Lösungen erfordern.
- Im Januar 2025 brachte Roquette drei neue Hilfsstofftypen auf den Markt, die speziell für feuchtigkeitsempfindliche pharmazeutische Wirkstoffe (APIs) entwickelt wurden und die Stabilität und Wirksamkeit in anspruchsvollen Formulierungen verbessern.
- Im Januar 2025 erweiterte Evonik sein Portfolio an lipidbasierten pharmazeutischen Hilfsstoffen durch strategische Akquisitionen und unterstützte damit die wachsende Nachfrage nach neuartigen Wirkstoffträgersystemen und hochwertigen Hilfsstoffen, die die Bioverfügbarkeit und Stabilität verbessern.
- Im Januar 2025 kündigte Akums Drugs and Pharmaceuticals die Entwicklung einer neuen Anlage zur Herstellung gefriergetrockneter und steriler Dosierungen mit Schwerpunkt auf Injektionspräparaten und Biologika an, um der steigenden Nachfrage nach sterilen und biologischen Arzneimitteln gerecht zu werden.
- Im September 2024 brachte Glenmark Pharmaceuticals eine Biosimilar-Variante des Antidiabetikums Liraglutid auf den Markt. Damit leistete das Unternehmen Pionierarbeit in der indischen Pharmabranche und erweiterte sein Biologika-Portfolio.
SKU-
Erhalten Sie Online-Zugriff auf den Bericht zur weltweit ersten Market Intelligence Cloud
- Interaktives Datenanalyse-Dashboard
- Unternehmensanalyse-Dashboard für Chancen mit hohem Wachstumspotenzial
- Zugriff für Research-Analysten für Anpassungen und Abfragen
- Konkurrenzanalyse mit interaktivem Dashboard
- Aktuelle Nachrichten, Updates und Trendanalyse
- Nutzen Sie die Leistungsfähigkeit der Benchmark-Analyse für eine umfassende Konkurrenzverfolgung
Forschungsmethodik
Die Datenerfassung und Basisjahresanalyse werden mithilfe von Datenerfassungsmodulen mit großen Stichprobengrößen durchgeführt. Die Phase umfasst das Erhalten von Marktinformationen oder verwandten Daten aus verschiedenen Quellen und Strategien. Sie umfasst die Prüfung und Planung aller aus der Vergangenheit im Voraus erfassten Daten. Sie umfasst auch die Prüfung von Informationsinkonsistenzen, die in verschiedenen Informationsquellen auftreten. Die Marktdaten werden mithilfe von marktstatistischen und kohärenten Modellen analysiert und geschätzt. Darüber hinaus sind Marktanteilsanalyse und Schlüsseltrendanalyse die wichtigsten Erfolgsfaktoren im Marktbericht. Um mehr zu erfahren, fordern Sie bitte einen Analystenanruf an oder geben Sie Ihre Anfrage ein.
Die wichtigste Forschungsmethodik, die vom DBMR-Forschungsteam verwendet wird, ist die Datentriangulation, die Data Mining, die Analyse der Auswirkungen von Datenvariablen auf den Markt und die primäre (Branchenexperten-)Validierung umfasst. Zu den Datenmodellen gehören ein Lieferantenpositionierungsraster, eine Marktzeitlinienanalyse, ein Marktüberblick und -leitfaden, ein Firmenpositionierungsraster, eine Patentanalyse, eine Preisanalyse, eine Firmenmarktanteilsanalyse, Messstandards, eine globale versus eine regionale und Lieferantenanteilsanalyse. Um mehr über die Forschungsmethodik zu erfahren, senden Sie eine Anfrage an unsere Branchenexperten.
Anpassung möglich
Data Bridge Market Research ist ein führendes Unternehmen in der fortgeschrittenen formativen Forschung. Wir sind stolz darauf, unseren bestehenden und neuen Kunden Daten und Analysen zu bieten, die zu ihren Zielen passen. Der Bericht kann angepasst werden, um Preistrendanalysen von Zielmarken, Marktverständnis für zusätzliche Länder (fordern Sie die Länderliste an), Daten zu klinischen Studienergebnissen, Literaturübersicht, Analysen des Marktes für aufgearbeitete Produkte und Produktbasis einzuschließen. Marktanalysen von Zielkonkurrenten können von technologiebasierten Analysen bis hin zu Marktportfoliostrategien analysiert werden. Wir können so viele Wettbewerber hinzufügen, wie Sie Daten in dem von Ihnen gewünschten Format und Datenstil benötigen. Unser Analystenteam kann Ihnen auch Daten in groben Excel-Rohdateien und Pivot-Tabellen (Fact Book) bereitstellen oder Sie bei der Erstellung von Präsentationen aus den im Bericht verfügbaren Datensätzen unterstützen.