Europe Exocrine Pancreatic Insufficiency Epi Therapeutics And Diagnostics Market
حجم السوق بالمليار دولار أمريكي
CAGR :
% 
 USD
1.79 Billion
USD
3.01 Billion
2024
2032
USD
1.79 Billion
USD
3.01 Billion
2024
2032
| 2025 –2032 | |
| USD 1.79 Billion | |
| USD 3.01 Billion | |
|
|
|
|
تجزئة سوق علاجات وتشخيص قصور البنكرياس الإفرازي (EPI) في أوروبا، حسب التشخيص (فحوصات التصوير واختبارات وظائف البنكرياس)، والعلاج (إدارة التغذية وعلاج تعويض إنزيم البنكرياس (PERT)، ونوع الدواء (عام وذو علامة تجارية)، والمستخدم النهائي (المستشفيات، والعيادات المتخصصة، والرعاية المنزلية، ومراكز التشخيص، والمعاهد البحثية والأكاديمية، وغيرها)، وقنوات التوزيع (العطاء المباشر، وصيدليات التجزئة، والموزعون الخارجيون، وغيرها) - اتجاهات الصناعة وتوقعاتها حتى عام 2032
حجم سوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا
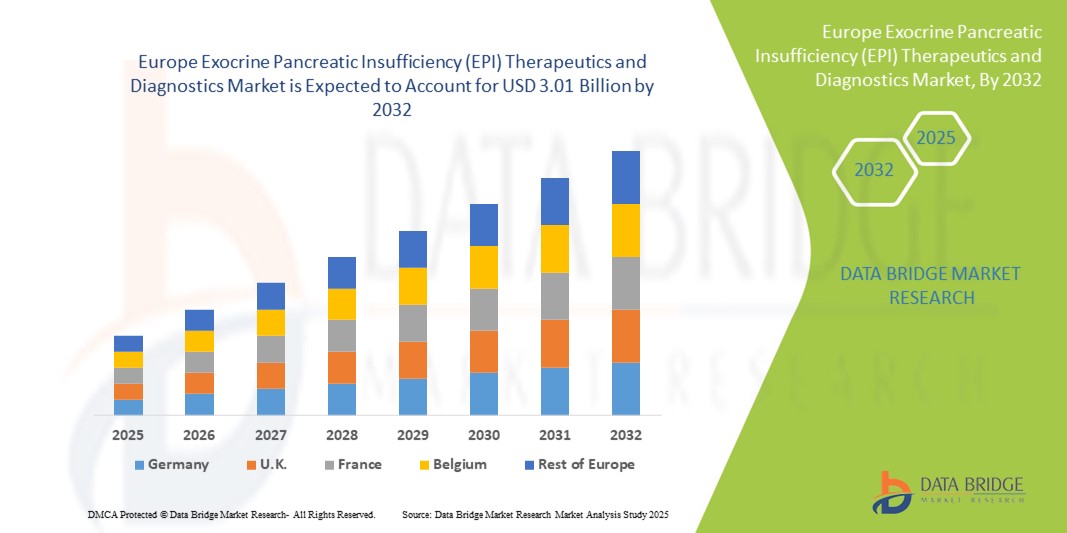
- تم تقييم حجم سوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا بنحو 1.79 مليار دولار أمريكي في عام 2024 ومن المتوقع أن يصل إلى 3.01 مليار دولار أمريكي بحلول عام 2032 ، بمعدل نمو سنوي مركب قدره 6.7٪ خلال الفترة المتوقعة.
- إن نمو السوق مدفوع إلى حد كبير بالانتشار المتزايد لاضطرابات البنكرياس والتليف الكيسي والتهاب البنكرياس المزمن ، إلى جانب تحسين الوصول إلى التصوير التشخيصي المتقدم وعلاجات استبدال الإنزيم في جميع أنحاء المنطقة.
- علاوة على ذلك، يُسهم تزايد الوعي بين مُقدّمي الرعاية الصحية والمرضى بشأن التشخيص المُبكر، إلى جانب الطلب المُتزايد على حلول علاجية فعّالة لتحسين جودة الحياة، في ترسيخ دور علاجات وتشخيصات EPI كقطاعٍ أساسي في رعاية أمراض الجهاز الهضمي. تُسرّع هذه العوامل المُجتمعة من اعتماد أدوات تشخيصية وعلاجات مُبتكرة، مما يُعزز نمو هذا القطاع بشكلٍ كبير.
تحليل سوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا
- يؤدي قصور البنكرياس الخارجي (EPI)، وهي حالة تتميز بعدم إفراز إنزيمات البنكرياس بشكل كافٍ، إلى زيادة الطلب على التشخيصات المتقدمة والحلول العلاجية في جميع أنحاء أوروبا، حيث تلعب علاجات استبدال الإنزيم وتقنيات التصوير دورًا محوريًا في تحسين إدارة المرض ونتائج المرضى.
- الطلب المتزايد على علاجات وتشخيصات EPI مدفوع في المقام الأول بالانتشار المتزايد لالتهاب البنكرياس المزمن وسرطان البنكرياس والتليف الكيسي، إلى جانب زيادة الوعي بصحة الجهاز الهضمي وفوائد الكشف المبكر.
- سيطرت ألمانيا على سوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا بأكبر حصة إيرادات بلغت 35٪ في عام 2024، وتتميز ببنية تحتية قوية للرعاية الصحية ومعدلات تشخيص عالية ووجود شركات أدوية رائدة تعمل على تطوير علاجات استبدال الإنزيم والابتكارات التشخيصية.
- من المتوقع أن تكون المملكة المتحدة أسرع دولة نموًا في سوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا خلال فترة التنبؤ بسبب التوسع في اعتماد الطب الدقيق ومبادرات الرعاية الصحية الحكومية ومجموعة المرضى المتزايدة التي تتطلب رعاية البنكرياس على المدى الطويل.
- هيمن قطاع العلاج باستبدال إنزيم البنكرياس (ERT) على سوق العلاجات والتشخيص لقصور البنكرياس الخارجي (EPI) في أوروبا بحصة سوقية بلغت 62.2% في عام 2024، مدفوعًا بفعاليته كمعيار للرعاية والتوافر المتزايد للتركيبات المحسنة المصممة لتعزيز التزام المريض ونوعية حياته.
نطاق التقرير وتقسيم سوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا
|
صفات |
رؤى السوق الرئيسية لقطاع علاج وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا |
|
القطاعات المغطاة |
|
|
الدول المغطاة |
أوروبا
|
|
اللاعبون الرئيسيون في السوق |
|
|
فرص السوق |
|
|
مجموعات معلومات البيانات ذات القيمة المضافة |
بالإضافة إلى الرؤى حول سيناريوهات السوق مثل القيمة السوقية ومعدل النمو والتجزئة والتغطية الجغرافية واللاعبين الرئيسيين، فإن تقارير السوق التي تم تنظيمها بواسطة Data Bridge Market Research تشمل أيضًا تحليلًا متعمقًا من الخبراء وتحليل التسعير وتحليل حصة العلامة التجارية واستطلاع رأي المستهلكين وتحليل التركيبة السكانية وتحليل سلسلة التوريد وتحليل سلسلة القيمة ونظرة عامة على المواد الخام / المواد الاستهلاكية ومعايير اختيار البائعين وتحليل PESTLE وتحليل Porter والإطار التنظيمي. |
اتجاهات سوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا
الطب الشخصي وتركيبات الإنزيمات المتقدمة
- إن الاتجاه المهم والمتسارع في سوق العلاجات والتشخيصات EPI في أوروبا هو تقدم الطب الشخصي وعلاجات استبدال الإنزيم من الجيل التالي (ERTs) المصممة لتحسين التزام المرضى ونتائج العلاج
- على سبيل المثال، تعمل الشركات على تطوير تركيبات إنزيمات البنكرياس المصممة خصيصًا مع تعزيز الاستقرار والتوافر البيولوجي لتلبية احتياجات المرضى الفردية بشكل أفضل، وتقليل الآثار الجانبية في الجهاز الهضمي وتحسين امتصاص العناصر الغذائية.
- تُمكّن الابتكارات في أساليب التشخيص، مثل اختبار الإيلاستاز-1 في البراز والكشف غير الجراحي عن المؤشرات الحيوية، من الكشف المبكر عن التهاب البنكرياس المزمن (EPI) لدى مرضى التهاب البنكرياس المزمن، أو التليف الكيسي، أو سرطان البنكرياس. علاوة على ذلك، تُحسّن تقنيات التصوير الحديثة دقة التشخيص ومراقبة العلاج.
- يدعم دمج أدوات الصحة الرقمية، بما في ذلك تطبيقات الهاتف المحمول وأنظمة المراقبة عن بعد، تعديلات الجرعات والعلاج المخصصة، مما يعزز إدارة الأمراض على المدى الطويل للمرضى في جميع أنحاء أوروبا
- هذا التوجه نحو حلول أكثر تركيزًا على المريض، ومخصصة، ومتطورة تكنولوجيًا، يُعيد تشكيل توقعات رعاية EPI. ونتيجةً لذلك، تستثمر الشركات في تركيبات إنزيمات مبتكرة، وتشخيصات مصاحبة، ومنصات رقمية لتعزيز تنافسيتها في السوق الأوروبية.
- يتزايد الطلب على العلاجات والتشخيصات التي توفر فعالية أفضل وراحة وتخصيصًا بسرعة، حيث يضع كل من المرضى ومقدمي الرعاية الصحية على رأس أولوياتهم بشكل متزايد تحسين نوعية الحياة وتحسين نتائج العلاج.
ديناميكيات سوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا
سائق
ارتفاع معدل انتشار اضطرابات البنكرياس وتزايد الوعي بها
- إن الانتشار المتزايد لاضطرابات البنكرياس مثل التهاب البنكرياس المزمن والتليف الكيسي وسرطان البنكرياس، إلى جانب الوعي المتزايد بصحة الجهاز الهضمي، يشكل محركًا مهمًا للطلب المتزايد على علاجات وتشخيصات EPI في أوروبا.
- على سبيل المثال، في فبراير 2024، قدمت مجموعات البحث الألمانية بروتوكولات تشخيصية متقدمة تعتمد على التصوير تهدف إلى تحسين الكشف المبكر عن EPI وبدء العلاج في الوقت المناسب، مما يسلط الضوء على التزام المنطقة بالابتكار في الرعاية الصحية.
- مع استمرار توسع مجموعة المرضى، أصبحت الحاجة إلى علاجات استبدال الإنزيمات الفعالة وأدوات التشخيص الموثوقة أكثر إلحاحًا، مما أدى إلى زيادة التبني في المستشفيات والعيادات المتخصصة ومختبرات التشخيص
- علاوة على ذلك، تعمل مبادرات الرعاية الصحية المدعومة من الحكومة، وهياكل السداد المحسنة، والتركيز القوي على التدخل المبكر للأمراض على تسريع الوصول إلى حلول تشخيصية وعلاجية مبتكرة في أوروبا.
- إن توافر خيارات العلاج المتقدمة، إلى جانب الطلب المتزايد على خدمات الرعاية الصحية الشخصية، يدفع عجلة تبني حلول رعاية EPI في كل من الاقتصادات الأوروبية الكبرى وأسواق الرعاية الصحية الناشئة. كما أن دمج الإرشادات السريرية المتخصصة يعزز نمو السوق بشكل أكبر.
ضبط النفس/التحدي
ارتفاع تكاليف العلاج والفجوات التشخيصية في جميع أنحاء أوروبا
- تشكل المخاوف المحيطة بالتكلفة العالية للعلاج ببدائل الإنزيم على المدى الطويل والتفاوت في الوصول إلى التشخيص عبر البلدان الأوروبية تحديًا كبيرًا أمام انتشار حلول رعاية EPI على نطاق أوسع في السوق
- على سبيل المثال، غالبًا ما يواجه المرضى في المناطق ذات الدخل المنخفض في شرق وجنوب أوروبا توفرًا محدودًا لاختبارات التشخيص المتقدمة وحواجز مالية أمام الاستخدام المستمر لـ ERT، مما يقلل من التبني مقارنة بأوروبا الغربية.
- إن معالجة هذه التحديات من خلال تطوير تركيبات إنزيمات بأسعار معقولة، وتوسيع نطاق تغطية التعويضات، وزيادة توافر اختبارات التشخيص الموحدة، أمرٌ بالغ الأهمية لضمان وصول المرضى إليها على قدم المساواة. كما تُركز الشركات على الشراكات مع مقدمي الرعاية الصحية لتحسين التوزيع.
- بالإضافة إلى ذلك، لا يزال تأخر التشخيص الناتج عن تداخل الأعراض مع اضطرابات الجهاز الهضمي الأخرى يعيق العلاج في الوقت المناسب، مما يؤثر على نتائج المرضى ويزيد من تكاليف الرعاية الصحية. وتُعدّ المبادرات التثقيفية للأطباء والمرضى بالغة الأهمية للتغلب على هذه العوائق.
- رغم التقدم المُحرز، لا تزال فجوة القدرة على تحمل التكاليف وعدم تكافؤ فرص الحصول على التشخيصات والعلاجات المتقدمة تُشكلان عائقين رئيسيين، لا سيما في أنظمة الرعاية الصحية التي تعاني من ضغوط الميزانية. وسيكون تحقيق التوازن بين الابتكار والقدرة على تحمل التكاليف أمرًا حيويًا لتحقيق نمو مستدام في سوق التشخيص الموسع الأوروبي.
نطاق سوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا
يتم تقسيم السوق على أساس التشخيص والعلاج ونوع الدواء والمستخدم النهائي وقناة التوزيع.
- حسب التشخيص
بناءً على التشخيص، يُقسّم سوق علاجات وتشخيصات قصور البنكرياس الخارجي (EPI) في أوروبا إلى فحوصات تصويرية وفحوصات وظائف البنكرياس. وقد هيمن قطاع فحوصات التصوير على السوق محققًا أكبر حصة من الإيرادات في عام 2024، بفضل انتشاره الواسع في المستشفيات ومراكز الرعاية الصحية المتخصصة، ودوره المحوري في تحديد التشوهات الهيكلية للبنكرياس التي غالبًا ما تكمن وراء قصور البنكرياس الخارجي. يعتمد الأطباء بشكل كبير على التصوير المقطعي المحوسب، والتصوير بالرنين المغناطيسي، والموجات فوق الصوتية بالمنظار للتشخيص التفريقي، وتخطيط العلاج، ومراقبة تطور المرض. إن قدرة التصوير على توفير تأكيد بصري لأمراض البنكرياس تجعله الأداة الأكثر ثقة بين الأطباء. علاوة على ذلك، يُواصل التطور في تقنيات التصوير عالي الدقة تحسين دقة الكشف، مما يعزز هيمنته في السوق.
من المتوقع أن يشهد قطاع اختبارات وظائف البنكرياس أسرع معدل نمو بين عامي 2025 و2032، مدفوعًا بالاعتماد المتزايد على الاختبارات التشخيصية غير الباضعة وعالية التخصص، مثل اختبار الإيلاستاز-1 البرازي. وتزداد تفضيلات هذه الاختبارات للكشف المبكر والدقيق عن نقص الإنزيم، لا سيما في العيادات الخارجية ومراكز التشخيص. ويساهم الطلب المتزايد على الطب الشخصي وتحسين فحص الفئات المعرضة للخطر في تسريع اعتماد الاختبارات الوظيفية. كما أن قدرتها على تقييم وظائف الغدد الصماء مباشرةً تجعلها أكثر موثوقية في تأكيد تشخيص نقص الإنزيمات الهاضمة (EPI) مقارنةً بالتصوير وحده. كما يُسهم التكامل المتزايد بين الاختبارات القائمة على المؤشرات الحيوية وحلول الاختبارات في نقطة الرعاية في النمو القوي لهذا القطاع.
- حسب العلاج
بناءً على العلاج، يُقسّم سوق علاجات وتشخيصات قصور البنكرياس الخارجي (EPI) في أوروبا إلى علاج تعويض إنزيم البنكرياس (PERT) وإدارة التغذية. وقد هيمن علاج تعويض إنزيم البنكرياس (PERT) على السوق محققًا أكبر حصة إيرادات بلغت 62.2% في عام 2024، كونه معيار الرعاية المعترف به عالميًا لإدارة قصور البنكرياس الخارجي، ويُوصف لغالبية المرضى المُشخّصين. فعاليته المُثبتة في تحسين امتصاص العناصر الغذائية وجودة الحياة تجعله الخيار العلاجي الأكثر استخدامًا. وتعزز الأدلة السريرية القوية، وتوافر تركيبات متعددة العلامات التجارية، وإدراجه في الإرشادات السريرية الأوروبية، مكانته الرائدة. بالإضافة إلى ذلك، أدت التركيبات المُحسّنة ذات الثبات والتوافر البيولوجي الأفضل إلى زيادة التزام المرضى بالعلاج، مما دعم الطلب المُستدام.
من المتوقع أن يشهد قطاع إدارة التغذية أسرع معدل نمو بين عامي 2025 و2032، مدفوعًا بزيادة التركيز على الرعاية الشاملة للمرضى وأهمية النظام الغذائي في إدارة مضاعفات سوء الامتصاص. ويتزايد اعتماد الدعم الغذائي، بما في ذلك الاستشارات الغذائية، ومكملات الفيتامينات القابلة للذوبان في الدهون، وخطط التغذية المتخصصة، إلى جانب العلاج الدوائي. ويعزز الوعي المتزايد بدور التغذية في نتائج المرضى على المدى الطويل من الاستفادة منها. علاوة على ذلك، فإن انتشار التغذية عن بُعد ومنصات الصحة الرقمية يجعل التوجيه الغذائي أكثر سهولة، مما يساهم في تسريع اعتماد إدارة التغذية في جميع أنحاء أوروبا.
- حسب نوع الدواء
بناءً على نوع الدواء، يُقسّم سوق علاجات وتشخيصات قصور البنكرياس الإفرازي (EPI) في أوروبا إلى أدوية عامة وأدوية ذات علامات تجارية. وقد حصدت الأدوية ذات العلامات التجارية أكبر حصة من الإيرادات في عام 2024، مدعومةً بالحضور القوي لشركات الأدوية الرائدة التي تُقدّم تركيبات مُعتمدة سريريًا وحاصلة على الموافقات التنظيمية. يُفضّل الأطباء العلاجات الإنزيمية ذات العلامات التجارية نظرًا لفعاليتها المُثبتة، وإرشادات الجرعات المُعتمدة، وجودتها العالية المُتوقعة مُقارنةً بالأدوية العامة. ويضمن إدراجها على نطاق واسع في قوائم الأدوية بالمستشفيات وبرامج السداد وصولًا واسعًا. علاوةً على ذلك، تُعزز استثمارات الشركات المُصنّعة في تثقيف الأطباء، وبرامج دعم المرضى، والبحث والتطوير المُستمر، مكانتها المُهيمنة في السوق.
من المتوقع أن يشهد قطاع الأدوية الجنيسة أسرع معدل نمو بين عامي 2025 و2032، ويعزى ذلك بشكل رئيسي إلى تدابير احتواء التكاليف والضغط المتزايد على ميزانيات الرعاية الصحية في جميع أنحاء أوروبا. تُقدم الأدوية الجنيسة بدائل أكثر فعالية من حيث التكلفة للعلاج طويل الأمد، مما يجعلها جذابة في المناطق ذات التمويل المحدود للرعاية الصحية. إن تزايد ثقة الأطباء في جودة الأدوية الجنيسة وتكافؤها الحيوي، إلى جانب أنظمة الشراء القائمة على المناقصات، يُسرّع من إقبال الناس عليها. إضافةً إلى ذلك، فإن انتهاء صلاحية براءات الاختراع على المنتجات ذات العلامات التجارية الرئيسية يفتح آفاقًا لمنافسة الأدوية الجنيسة. ومع تزايد أهمية القدرة على تحمل التكاليف، من المتوقع أن تستحوذ الأدوية الجنيسة على حصة كبيرة من سوق علاجات EPI في السنوات القادمة.
- حسب المستخدم النهائي
بناءً على المستخدم النهائي، يُقسّم سوق علاجات وتشخيص قصور البنكرياس الإفرازي (EPI) في أوروبا إلى مستشفيات، وعيادات متخصصة، ومراكز رعاية منزلية، ومراكز تشخيص، ومعاهد بحثية وأكاديمية، وغيرها. هيمن قطاع المستشفيات على السوق بحصة سوقية كبيرة في عام 2024، بفضل دوره المحوري في التشخيص، والرعاية الحادة، وبدء علاج قصور البنكرياس الإفرازي طويل الأمد. تمتلك المستشفيات البنية التحتية اللازمة لإجراء تصوير متقدم واختبارات وظائف مباشرة، مما يجعلها مراكز إحالة للحالات المعقدة. علاوة على ذلك، تُمكّن الفرق متعددة التخصصات في المستشفيات من تقديم رعاية شاملة، تشمل الدعم الغذائي والعلاج الدوائي. كما أن إدراجها في عقود مشتريات واسعة النطاق يضمن لها حصة سوقية مهيمنة.
من المتوقع أن يشهد قطاع الرعاية المنزلية أسرع نمو بين عامي 2025 و2032، وذلك بفضل التحول المتزايد نحو الرعاية الخارجية والإدارة طويلة الأمد لـ EPI. يفضل المرضى الرعاية المنزلية لراحتها، مدعومةً بتوافر PERT في تركيبات كبسولات يسهل تناولها ذاتيًا. كما أن التوسع في التطبيب عن بُعد والعلاجات التي تُقدم عبر الصيدليات يُعزز تبني هذه الخدمات. كما تُخفف الرعاية المنزلية من أعباء المستشفيات وتكاليفها، مما يجعلها جذابة للدافعين وأنظمة الرعاية الصحية. وتُعزز حملات التوعية المتزايدة وتثقيف المرضى التوجه نحو نماذج العلاج اللامركزية التي يُديرها المريض بنفسه.
- حسب قناة التوزيع
بناءً على قنوات التوزيع، يُقسّم سوق العلاجات والتشخيصات لقصور البنكرياس الإفرازي (EPI) في أوروبا إلى مناقصة مباشرة، وصيدليات التجزئة، وموزعين تابعين لجهات خارجية، وغيرها. هيمن قطاع المناقصة المباشرة على السوق في عام 2024 بفضل عمليات الشراء بالجملة التي تقوم بها المستشفيات ومؤسسات الرعاية الصحية العامة عبر أنظمة الشراء المركزية. تضمن العقود القائمة على المناقصة توريدًا آمنًا لمنتجات PERT عالية التكلفة وحلول التشخيص، مع تعزيز حجم المبيعات للموردين. غالبًا ما تُفضّل أنظمة الرعاية الصحية الوطنية المناقصات المباشرة لكفاءتها من حيث التكلفة وجودة المنتجات القياسية. تُوفّر هذه العقود تدفقات إيرادات متوقعة للمصنعين، مما يُعزز هيمنة هذا القطاع.
من المتوقع أن ينمو قطاع صيدليات التجزئة بأسرع معدل نمو سنوي مركب بين عامي 2025 و2032، مدفوعًا بتزايد أنماط وصف الأدوية للمرضى الخارجيين وتفضيل المرضى لقنوات العلاج المتاحة. تلعب صيدليات التجزئة دورًا حاسمًا في توزيع علاجات PERT والمكملات الغذائية، وتقديم الاستشارات ودعم الالتزام بالعلاج. كما أن نمو الصيدليات الإلكترونية وخدمات التوصيل المنزلي يُعزز هذا التوسع. بالإضافة إلى ذلك، يُعزز تزايد عدد الموزعين الخارجيين وتجار التجزئة المتخصصين مرونة سلسلة التوريد، مما يُحسّن توافرها في المناطق التي تعاني نقصًا في الخدمات. ومع انتقال المزيد من المرضى إلى رعاية المرضى الخارجيين المزمنة، فإن قنوات التجزئة والجهات الخارجية مهيأة للنمو السريع.
تحليل إقليمي لسوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا
- سيطرت ألمانيا على سوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا بأكبر حصة إيرادات بلغت 35٪ في عام 2024، وتتميز ببنية تحتية قوية للرعاية الصحية ومعدلات تشخيص عالية ووجود شركات أدوية رائدة تعمل على تطوير علاجات استبدال الإنزيم والابتكارات التشخيصية.
- يستفيد المرضى في ألمانيا من التغطية التأمينية الشاملة والوعي الطبي القوي والوصول إلى التركيبات ذات العلامات التجارية والعامة، مما يجعل العلاجات متاحة على نطاق واسع
- ويتم دعم هذا التبني الواسع النطاق من خلال التركيز الذي توليه الدولة على البحث والابتكار السريري، إلى جانب التعاون بين المعاهد الأكاديمية والمستشفيات وشركات تصنيع الأدوية.
نظرة ثاقبة على سوق العلاج والتشخيص في ألمانيا (EPI)
شكّل سوق علاجات وتشخيصات قصور البنكرياس الإفرازي (EPI) في ألمانيا أكبر حصة من الإيرادات في أوروبا عام 2024، بفضل التوافر الواسع لـ PERT، والوعي التشخيصي العالي، والتغطية التأمينية الشاملة. يُركّز مقدمو الرعاية الصحية الألمان على دقة الاختبارات من خلال تحاليل الإيلاستاز البرازية والتصوير، مما يضمن التدخل في الوقت المناسب. يُعزز التعاون الوثيق بين المستشفيات والجامعات وشركات الأدوية الابتكار السريري، ويدعم التوسع في استخدام العلاجات ذات العلامات التجارية والأدوية الجنيسة. ومع انخراط المرضى بشكل كبير في إدارة الأمراض على المدى الطويل، تحافظ ألمانيا على مكانتها كمركز رائد في علاجات وتشخيصات قصور البنكرياس الإفرازي في أوروبا.
نظرة عامة على سوق العلاج والتشخيص في المملكة المتحدة (EPI)
من المتوقع أن ينمو سوق علاجات وتشخيصات قصور البنكرياس الإفرازي (EPI) في المملكة المتحدة بمعدل نمو سنوي مركب ملحوظ خلال الفترة المتوقعة، مدفوعًا بارتفاع معدل انتشار اضطرابات الجهاز الهضمي والتركيز المتزايد على الرعاية المتخصصة. تدعم هيئة الخدمات الصحية الوطنية (NHS) توفير إمكانية الوصول على نطاق واسع إلى اختبارات PERT والتشخيص، بينما يُسهم ازدياد وعي المرضى في التشخيص المبكر. يُسهم الابتكار التكنولوجي، بما في ذلك اعتماد الاختبارات القائمة على المؤشرات الحيوية، في زيادة دقة العلاج. علاوة على ذلك، يُسهم النظام البيئي القوي للأبحاث السريرية في المملكة المتحدة ووجود شركات الأدوية العالمية في تسريع الابتكار، مما يُعزز دورها كسوق سريعة النمو في أوروبا.
نظرة عامة على سوق العلاج والتشخيص في فرنسا (EPI)
من المتوقع أن يشهد سوق علاجات وتشخيصات قصور البنكرياس الخارجي (EPI) في فرنسا نموًا مطردًا، مدعومًا بالاستثمارات المتزايدة في أبحاث أمراض الجهاز الهضمي والمبادرات الوطنية الهادفة إلى تحسين الوصول إلى إدارة الأمراض المزمنة. يساهم ارتفاع معدلات التليف الكيسي ومضاعفات التهاب البنكرياس في زيادة الطلب على علاج PERT. تلعب المستشفيات والعيادات التخصصية دورًا حاسمًا في التشخيص وتقديم العلاج على نطاق واسع، بينما تُسهّل سياسات السداد الحكومية اعتماد العلاج. يُعزز تركيز فرنسا على الرعاية المتكاملة للمرضى والتجارب السريرية في علاجات استبدال الإنزيم مسار نموها في سوق EPI الأوروبي.
نظرة عامة على سوق علاجات وتشخيصات EPI في إيطاليا
يكتسب سوق علاجات وتشخيص قصور البنكرياس الإفرازي (EPI) في إيطاليا زخمًا متزايدًا، مدعومًا بحملات التوعية المتزايدة، وتحسين القدرات التشخيصية، وتزايد عبء اضطرابات الجهاز الهضمي. يركز مقدمو الرعاية الصحية الإيطاليون على مناهج تركز على المريض، ويدمجون برنامج PERT في برامج إدارة التغذية لتحسين جودة الحياة. ويتزايد اعتماد تقنيات التشخيص المتقدمة، مثل فحص وظائف البنكرياس، في العيادات التخصصية. بالإضافة إلى ذلك، يعزز التعاون مع شركات الأدوية العالمية ومراكز البحث الأكاديمي قدرة إيطاليا على تقديم حلول مبتكرة، مما يجعلها سوقًا واعدة للنمو في أوروبا.
حصة السوق الأوروبية للعلاجات والتشخيصات لقصور البنكرياس الخارجي (EPI)
إن صناعة علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا يقودها في المقام الأول شركات راسخة، بما في ذلك:
- شركة AbbVie Inc. (الولايات المتحدة)
- نستله للعلوم الصحية (سويسرا)
- شركة أدالفو المحدودة (مالطا)
- شركة دايجستف كير (الولايات المتحدة)
- شركة فياتريس (الولايات المتحدة)
- شركة ألكريستا ثيرابيوتكس (الولايات المتحدة)
- شركة أناجرام ثيرابيوتكس (المملكة المتحدة)
- كودكسيس، المحدودة (الولايات المتحدة)
- نوردمارك أرزنيميتيل / مجموعة نوردمارك (ألمانيا)
- شركة بيوهيت أويج (فنلندا)
- شركة شيبو بيوتيك إيه جي (ألمانيا)
- دياسورين سبا (إيطاليا)
- Gentian Diagnostics ASA (النرويج)
- EUSA Pharma (المملكة المتحدة)
- شركة ألفا سيجما (إيطاليا)
- شركة كويست دياجنوستيكس المحدودة (الولايات المتحدة)
- شركة إف. هوفمان-لا روش المحدودة (سويسرا)
- شركة سيمنز هيلثينيرز إيه جي (ألمانيا)
- شركة تاكيدا للأدوية المحدودة (اليابان)
- بيوميرو (فرنسا)
ما هي التطورات الأخيرة في سوق علاجات وتشخيص قصور البنكرياس الخارجي (EPI) في أوروبا؟
- في يونيو 2025، أعلنت شركة أدالفو عن استعداداتها لإطلاق رئيسي للبنكرياتين في أوروبا، وهو العلاج الإنزيمي الأكثر شيوعًا لـ EPI، استنادًا إلى الدراسات السريرية التي تؤكد تحسن امتصاص الدهون والبروتينات والكربوهيدرات لدى المرضى الذين يعانون من EPI بسبب التهاب البنكرياس المزمن والتليف الكيسي واستئصال البنكرياس.
- في يناير 2025، دخلت شركة Anagram Therapeutics في شراكة مع Epicured لتطوير وتنفيذ برنامج تغذية مُصمم خصيصًا لدراسة جرعات سريرية تتضمن علاجًا فمويًا جديدًا ببدائل الإنزيم لمرضى التليف الكيسي وقصور البنكرياس الخارجي (EPI). استُخدم حل Epicured الغذائي في 20 موقعًا وطنيًا لدعم اتساق البروتوكول ونتائج المرضى.
- في أكتوبر 2024، كشفت دراسة سجل أوروبية مستقبلية أن الجرعة المثلى من PERT تختلف بشكل كبير اعتمادًا على السبب الكامن وراء مرضى EPI المصابين بسرطان البنكرياس أو بعد استئصال البنكرياس غالبًا ما يحتاجون إلى جرعات إنزيمية أعلى بكثير، وغالبًا ما تكون هناك حاجة إلى مثبط مضخة البروتون (PPI) لتحقيق الفعالية العلاجية.
- في يونيو 2024، تم اعتماد المبادئ التوجيهية الأوروبية لتشخيص وعلاج قصور إفراز البنكرياس رسميًا من قبل الجمعيات الرئيسية بما في ذلك UEG وEPC وEDS وESPEN وESPGHAN وESDO وESPCG، والتي أيدت نهجًا تشخيصيًا شاملاً (الأعراض + التقييم الغذائي + اختبار إفراز البنكرياس) وأكدت على PERT بالإضافة إلى الدعم الغذائي باعتباره حجر الزاوية العلاجي.
- في أبريل 2024، تم الإبلاغ عن أداة فحص سريرية جديدة لـ EPI مع التركيز على الكشف المبكر عن التهاب البنكرياس المزمن، مما يسلط الضوء على الافتقار إلى الاختبارات البسيطة والدقيقة والحاجة إلى استراتيجيات تشخيصية محسنة.
SKU-
احصل على إمكانية الوصول عبر الإنترنت إلى التقرير الخاص بأول سحابة استخبارات سوقية في العالم
- لوحة معلومات تحليل البيانات التفاعلية
- لوحة معلومات تحليل الشركة للفرص ذات إمكانات النمو العالية
- إمكانية وصول محلل الأبحاث للتخصيص والاستعلامات
- تحليل المنافسين باستخدام لوحة معلومات تفاعلية
- آخر الأخبار والتحديثات وتحليل الاتجاهات
- استغل قوة تحليل المعايير لتتبع المنافسين بشكل شامل
منهجية البحث
يتم جمع البيانات وتحليل سنة الأساس باستخدام وحدات جمع البيانات ذات أحجام العينات الكبيرة. تتضمن المرحلة الحصول على معلومات السوق أو البيانات ذات الصلة من خلال مصادر واستراتيجيات مختلفة. تتضمن فحص وتخطيط جميع البيانات المكتسبة من الماضي مسبقًا. كما تتضمن فحص التناقضات في المعلومات التي شوهدت عبر مصادر المعلومات المختلفة. يتم تحليل بيانات السوق وتقديرها باستخدام نماذج إحصائية ومتماسكة للسوق. كما أن تحليل حصة السوق وتحليل الاتجاهات الرئيسية هي عوامل النجاح الرئيسية في تقرير السوق. لمعرفة المزيد، يرجى طلب مكالمة محلل أو إرسال استفسارك.
منهجية البحث الرئيسية التي يستخدمها فريق بحث DBMR هي التثليث البيانات والتي تتضمن استخراج البيانات وتحليل تأثير متغيرات البيانات على السوق والتحقق الأولي (من قبل خبراء الصناعة). تتضمن نماذج البيانات شبكة تحديد موقف البائعين، وتحليل خط زمني للسوق، ونظرة عامة على السوق ودليل، وشبكة تحديد موقف الشركة، وتحليل براءات الاختراع، وتحليل التسعير، وتحليل حصة الشركة في السوق، ومعايير القياس، وتحليل حصة البائعين على المستوى العالمي مقابل الإقليمي. لمعرفة المزيد عن منهجية البحث، أرسل استفسارًا للتحدث إلى خبراء الصناعة لدينا.
التخصيص متاح
تعد Data Bridge Market Research رائدة في مجال البحوث التكوينية المتقدمة. ونحن نفخر بخدمة عملائنا الحاليين والجدد بالبيانات والتحليلات التي تتطابق مع هدفهم. ويمكن تخصيص التقرير ليشمل تحليل اتجاه الأسعار للعلامات التجارية المستهدفة وفهم السوق في بلدان إضافية (اطلب قائمة البلدان)، وبيانات نتائج التجارب السريرية، ومراجعة الأدبيات، وتحليل السوق المجدد وقاعدة المنتج. ويمكن تحليل تحليل السوق للمنافسين المستهدفين من التحليل القائم على التكنولوجيا إلى استراتيجيات محفظة السوق. ويمكننا إضافة عدد كبير من المنافسين الذين تحتاج إلى بيانات عنهم بالتنسيق وأسلوب البيانات الذي تبحث عنه. ويمكن لفريق المحللين لدينا أيضًا تزويدك بالبيانات في ملفات Excel الخام أو جداول البيانات المحورية (كتاب الحقائق) أو مساعدتك في إنشاء عروض تقديمية من مجموعات البيانات المتوفرة في التقرير.