North America Laboratory Information Management Systems Market
Market Size in USD Million
CAGR :
% 
 USD
714.08 Million
USD
1,465.16 Million
2025
2033
USD
714.08 Million
USD
1,465.16 Million
2025
2033
| 2026 –2033 | |
| USD 714.08 Million | |
| USD 1,465.16 Million | |
|
|
|
|
North America Laboratory Information Management Systems (LIMS) Market Size
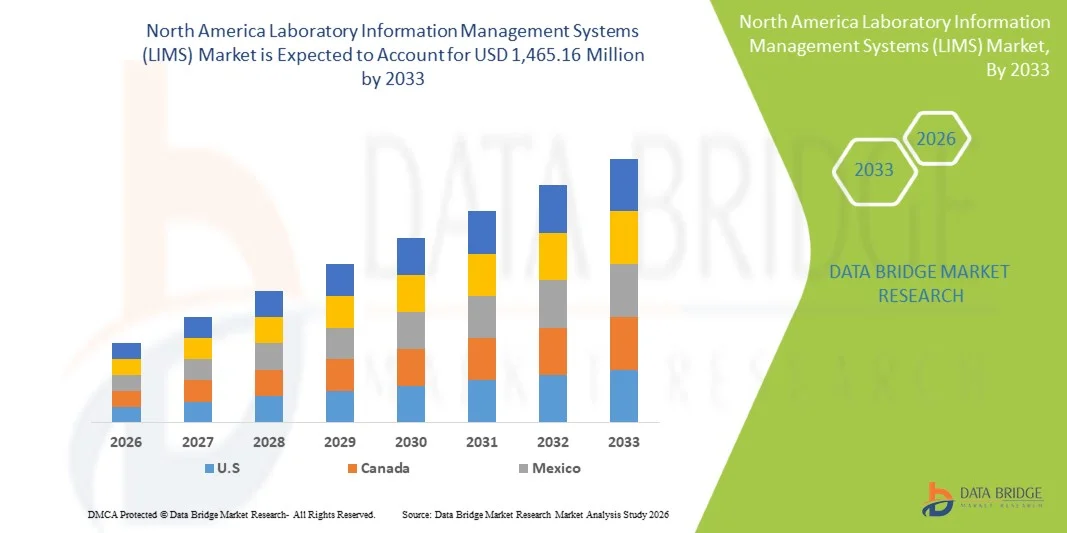
- The North America Laboratory Information Management Systems (LIMS) market size was valued at USD 714.08 million in 2025 and is expected to reach USD 1,465.16 million by 2033, at a CAGR of 9.40% during the forecast period
- The market growth is largely fueled by increasing implementation of LIMS across diverse laboratory environments to streamline sample tracking, enhance data accuracy, meet stringent regulatory compliance demands, and improve operational productivity; integration with advanced technologies such as cloud‑based solutions, AI‑driven analytics, and interoperable digital systems is further accelerating uptake across North America
- Furthermore, rising demand from highly regulated sectors such as life sciences, healthcare diagnostics, and pharmaceutical R&D for secure, efficient, and integrated laboratory information systems is establishing LIMS as a core component of modern laboratory infrastructure, significantly boosting the industry’s growth
North America Laboratory Information Management Systems (LIMS) Market Analysis
- Laboratory Information Management Systems (LIMS), providing digital management of laboratory samples, workflows, and data, are increasingly vital components of modern laboratories in clinical, pharmaceutical, biotech, and environmental testing settings due to their enhanced efficiency, data accuracy, and seamless integration with automated laboratory instruments
- The escalating demand for LIMS is primarily fueled by the widespread adoption of laboratory automation technologies, stringent regulatory compliance requirements, and a rising preference for streamlined, paperless workflows that improve productivity and traceability
- The United States dominated the North America LIMS market with the largest revenue share of 83.2% in 2025, characterized by early adoption of laboratory automation, high investment in R&D, and a strong presence of key industry players, with substantial growth in LIMS deployments, particularly in pharmaceutical and clinical laboratories, driven by innovations in cloud-based platforms, AI-driven analytics, and interoperable systems
- Canada is expected to be the fastest growing country in the North America LIMS market during the forecast period due to increasing adoption of advanced laboratory technologies, rising life sciences and healthcare investments, and expanding regulatory frameworks
- Cloud-based segment dominated the market with a market share of 46.9% in 2025, driven by its scalability, remote accessibility, and ability to integrate with other laboratory instruments and enterprise systems
Report Scope and North America Laboratory Information Management Systems (LIMS) Market Segmentation
|
Attributes |
North America Laboratory Information Management Systems (LIMS) Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
North America Laboratory Information Management Systems (LIMS) Market Trends
“Enhanced Efficiency Through AI and Cloud Integration”
- A significant and accelerating trend in the North America LIMS market is the integration of artificial intelligence (AI) and cloud-based platforms, enhancing laboratory efficiency, data analytics, and workflow automation
- For instance, cloud-enabled LIMS solutions from LabWare allow laboratories to access and manage sample data remotely while applying AI algorithms to optimize testing schedules and resource allocation
- AI integration in LIMS enables predictive analytics for experiment outcomes, anomaly detection, and intelligent workflow suggestions, improving decision-making and reducing human error
- The seamless integration of LIMS with laboratory instruments and enterprise systems facilitates centralized management of samples, inventory, and regulatory compliance across multiple laboratory sites
- This trend toward more intelligent, automated, and interconnected laboratory management systems is reshaping expectations for operational efficiency and data accuracy
- The demand for AI-enabled, cloud-based LIMS solutions is growing rapidly across pharmaceutical, clinical, and research laboratories as organizations prioritize productivity and streamlined operations
- Integration with Internet of Things (IoT) laboratory devices is increasing, allowing LIMS to automatically collect instrument data, reduce manual entry errors, and improve throughput
North America Laboratory Information Management Systems (LIMS) Market Dynamics
Driver
“Growing Need Due to Rising Regulatory Compliance and Laboratory Automation”
- The increasing focus on stringent regulatory compliance, along with the adoption of automated laboratory workflows, is a significant driver for the growing demand for LIMS
- For instance, in March 2025, LabWare launched a new AI-powered cloud LIMS designed to help pharmaceutical labs maintain compliance with FDA and EMA standards while improving operational efficiency
- As laboratories face higher requirements for traceability, reporting, and quality assurance, LIMS offers centralized data management, audit trails, and automated reporting, providing a compelling upgrade over manual systems
- Furthermore, the growing adoption of laboratory automation and connected instruments is making LIMS an integral component of modern laboratories, facilitating seamless workflow integration and instrument interoperability
- The ability to manage samples, data, and reports remotely through cloud platforms, combined with AI-driven process optimization, is driving LIMS adoption across pharmaceutical, clinical, and environmental testing laboratories
- For instance, Agilent’s LIMS solutions integrate with laboratory robots to automate sample handling, improving throughput and accuracy
- Increasing collaboration between laboratories and research institutions is pushing demand for LIMS systems that support multi-site data sharing, standardized protocols, and collaborative reporting
Restraint/Challenge
“Data Security Concerns and High Implementation Costs”
- Concerns surrounding data security and compliance with privacy regulations pose a significant challenge to broader adoption of LIMS in North America
- For instance, high-profile cyberattacks targeting laboratory data systems have made some organizations hesitant to migrate sensitive sample and research data to cloud-based LIMS
- Addressing these concerns through secure cloud infrastructure, advanced encryption, and compliance with HIPAA and FDA 21 CFR Part 11 standards is crucial for building trust among laboratory managers
- In addition, the relatively high cost of implementing advanced LIMS, particularly with AI and cloud integration, can be a barrier for smaller laboratories or budget-constrained organizations
- While subscription-based cloud LIMS models are gradually reducing upfront costs, the perceived premium for sophisticated features such as real-time analytics, instrument integration, and predictive workflows can still hinder widespread adoption
- Overcoming these challenges through robust security protocols, training, and cost-effective deployment options will be vital for sustained growth in the North America LIMS market
- Integration complexity with legacy laboratory systems can slow down adoption, requiring specialized IT support and change management strategies
- Limited awareness and understanding of advanced LIMS capabilities among smaller labs can restrict market penetration, emphasizing the need for targeted education and demonstration initiatives
North America Laboratory Information Management Systems (LIMS) Market Scope
The market is segmented on the basis of component, product type, delivery, industry type, and distribution channel.
- By Component
On the basis of component, the North America LIMS market is segmented into software and services. The Software segment dominated the market with the largest revenue share in 2025, driven by the widespread adoption of LIMS platforms for sample tracking, data management, and regulatory compliance. Laboratories across life sciences, diagnostics, and pharmaceutical industries increasingly rely on software solutions to automate workflows, reduce manual errors, and ensure traceability. The availability of modular and scalable software that can integrate with instruments and third-party systems further strengthens adoption. Advanced software features such as AI-driven analytics, predictive maintenance, and cloud integration enhance operational efficiency and decision-making. Compliance with FDA, ISO, and HIPAA standards also encourages software deployment in clinical and pharmaceutical laboratories. Overall, software solutions are considered the backbone of LIMS deployment, providing core functionality and workflow automation essential for modern laboratory operations.
The Services segment is expected to witness the fastest growth rate during 2026–2033, fueled by increasing demand for implementation, customization, training, and maintenance services. Many laboratories, particularly small and mid-sized ones, require expert support for integrating LIMS with legacy systems, validating software according to regulatory standards, and training staff for optimized usage. Professional services also include remote monitoring, software upgrades, and cloud migration assistance, ensuring continuous operational efficiency. As laboratories adopt advanced AI-enabled and cloud-based solutions, the need for specialized consulting and technical support is expected to grow significantly. In addition, the complexity of multi-site deployments and compliance-driven validation drives the demand for external LIMS services.
- By Product Type
On the basis of product type, the market is segmented into broad-based and industry-specific LIMS. The Broad-Based LIMS segment dominated the market in 2025, accounting for the largest share, due to its versatility across multiple laboratory types such as clinical, pharmaceutical, and environmental testing. These solutions provide flexible configurations that cater to various workflow requirements, supporting multiple instruments and regulatory standards. Broad-based LIMS offer modular architectures that allow laboratories to add features as needed, reducing initial investment barriers. Integration capabilities with enterprise systems, cloud platforms, and AI analytics also drive adoption in large, multi-site laboratories. In addition, broad-based LIMS are often preferred by organizations seeking standardized operations and centralized data management across different departments. Their adaptability and comprehensive features make them a reliable choice for laboratories with diverse testing needs.
The Industry-Specific LIMS segment is expected to witness the fastest growth from 2026 to 2033, driven by tailored solutions for industries such as biopharmaceuticals, food & beverage, clinical research, and diagnostics. Industry-specific LIMS provide pre-configured workflows, compliance modules, and reporting tools that address the unique requirements of each sector. For instance, a clinical research organization benefits from features such as audit trails, protocol management, and regulatory reporting, while a food & beverage lab relies on allergen testing modules and traceability features. Customization and industry-focused functionality enhance operational efficiency and reduce validation timelines, accelerating adoption. The increasing specialization of laboratories and growing regulatory demands are key factors driving this segment’s rapid growth.
- By Delivery
On the basis of delivery, the North America LIMS market is segmented into On-Premise, Cloud-Based, and Remotely-Hosted solutions. The Cloud-Based segment dominated the market with a market share of 46.9% in 2025, driven by scalability, remote accessibility, cost-efficiency, and integration with AI analytics and IoT-enabled laboratory instruments. Cloud LIMS allows multiple sites to manage samples and workflows in real time, reducing IT infrastructure costs and improving collaboration. Small-to-medium laboratories particularly benefit from subscription-based cloud models that provide flexibility and lower upfront investments. Regulatory-compliant cloud platforms with encryption and secure access further enhance adoption across pharmaceutical and clinical laboratories.
The Remotely-Hosted segment is expected to witness the fastest growth during 2026–2033, fueled by rising adoption among small and medium laboratories seeking cost-effective, secure, and remotely managed LIMS solutions. Remotely-hosted platforms reduce IT management burdens, allow real-time access to data, and provide automated software updates. Integration with AI analytics and cloud capabilities enhances operational efficiency, while enabling multi-site management. Growing demand for digital transformation and secure remote data access is driving the rapid growth of remotely-hosted LIMS
- By Industry Type
On the basis of industry type, the market is segmented into life sciences industry, chemical/energy, food & beverage & agriculture industries, diagnostics/medical device, clinical research organization/contract manufacturing organizations (CRO/CMO), and others. The Life Sciences Industry dominated the market in 2025, driven by high R&D activities, stringent compliance requirements, and the need for efficient data management in pharmaceutical and biotech laboratories. LIMS adoption in life sciences helps streamline drug development workflows, manage clinical trial samples, maintain regulatory compliance, and integrate laboratory instruments with enterprise systems. Large-scale life sciences organizations prefer robust LIMS solutions with AI analytics, cloud capabilities, and multi-site management features. The critical nature of clinical data integrity and the cost of errors make life sciences laboratories the primary adopters of advanced LIMS solutions. Broad functionality and regulatory alignment reinforce its market dominance.
The Diagnostics/Medical Device segment is expected to witness the fastest growth from 2026 to 2033, fueled by rising demand for accurate testing, faster turnaround times, and regulatory compliance. Diagnostic laboratories increasingly rely on LIMS to manage patient samples, automate workflows, and generate reliable reports while adhering to CLIA, ISO, and HIPAA standards. Integration with automated instruments and cloud-based reporting enhances operational efficiency. In addition, growing adoption of personalized medicine and genomic testing drives LIMS deployment in diagnostics labs. The focus on digital transformation in healthcare and the need for scalable, compliant solutions contribute to the rapid growth of this segment.
- By Distribution Channel
On the basis of distribution channel, the North America LIMS market is segmented into direct tenders and retail sales. The Direct Tenders segment dominated the market in 2025, driven by large laboratory chains, hospitals, and pharmaceutical companies procuring LIMS directly from vendors to meet specific regulatory and customization requirements. Direct procurement ensures better vendor support, SLA agreements, and tailored deployment strategies. Enterprises often prefer direct tenders to ensure compliance with data security standards, integrate with existing laboratory instruments, and receive training and maintenance services. Large-scale implementations with complex workflows favor direct procurement over indirect channels. Strong vendor relationships and project-based customization reinforce the dominance of this segment.
The Retail Sales segment is expected to witness the fastest growth during 2026–2033, fueled by increasing adoption of LIMS by small and mid-sized laboratories seeking out-of-the-box software solutions. Retail sales provide easier access to packaged LIMS products with standard configurations, lower upfront costs, and subscription-based models. The growing trend of cloud-based offerings and user-friendly interfaces makes retail sales attractive to laboratories with limited IT support. Online marketplaces and vendor websites are expanding access to LIMS products, accelerating adoption among smaller labs and CROs. Cost-effectiveness, simplicity, and scalability are driving the rapid growth of the retail sales channel.
North America Laboratory Information Management Systems (LIMS) Market Regional Analysis
- The United States dominated the North America LIMS market with the largest revenue share of 83.2% in 2025, characterized by early adoption of laboratory automation, high investment in R&D, and a strong presence of key industry players
- Laboratories in the region highly value the efficiency, data accuracy, and centralized sample management offered by LIMS, along with seamless integration with laboratory instruments, cloud platforms, and AI-driven analytics for optimized workflows
- This widespread adoption is further supported by strong healthcare and life sciences infrastructure, high IT spending, and a focus on digital transformation, establishing LIMS as a critical solution for operational efficiency, regulatory compliance, and multi-site laboratory management across the United States
U.S. Laboratory Information Management Systems (LIMS) Market Insight
The U.S. LIMS market captured the largest revenue share of 83.2% in 2025 within North America, fueled by widespread adoption of laboratory automation and high investment in life sciences, clinical research, and pharmaceutical R&D. Laboratories prioritize operational efficiency, data accuracy, and regulatory compliance through advanced LIMS platforms. The growing preference for cloud-based and AI-enabled LIMS solutions, combined with seamless integration with laboratory instruments and enterprise systems, further drives market growth. In addition, multi-site laboratory operations and strict FDA, HIPAA, and ISO standards are significantly contributing to LIMS adoption.
Canada Laboratory Information Management Systems (LIMS) Market Insight
The Canada LIMS market is anticipated to grow at a substantial CAGR during the forecast period, driven by the increasing adoption of automated laboratory workflows and strong regulatory compliance requirements. Clinical, pharmaceutical, and environmental laboratories are investing in LIMS for centralized sample management, enhanced data accuracy, and streamlined reporting. Cloud-based and AI-enabled LIMS platforms are gaining popularity, providing remote access, workflow optimization, and integration with laboratory instruments. Government initiatives promoting research digitalization and quality standards further support the adoption of advanced LIMS solutions across both public and private laboratories.
Mexico Laboratory Information Management Systems (LIMS) Market Insight
The Mexico LIMS market is expected to expand steadily, fueled by growing pharmaceutical, biotech, and clinical research activities in the country. Laboratories are increasingly implementing LIMS to improve sample tracking, data integrity, and compliance with local and international regulatory standards. Rising awareness of laboratory automation and the adoption of cloud-based LIMS solutions are supporting market growth. In addition, cost-effective and scalable LIMS platforms are enabling small and medium laboratories to modernize workflows, reduce manual errors, and enhance operational efficiency. Increasing collaborations with U.S. and international laboratories also contribute to LIMS deployment in Mexico.
North America Laboratory Information Management Systems (LIMS) Market Share
The North America Laboratory Information Management Systems (LIMS) industry is primarily led by well-established companies, including:
- Thermo Fisher Scientific Inc. (U.S.)
- PerkinElmer (U.S.)
- Autoscribe Informatics (U.K.)
- Shimadzu Corporation (Japan)
- Novatek International (U.S.)
- Benchling (U.S.)
- Agaram Technologies Pvt Ltd (India)
- Agilent Technologies, Inc. (U.S.)
- LabWare (U.S.)
- STARLIMS Corporation (U.S.)
- IBM Corporation (U.S.)
- Roper Technologies, Inc (U.S.)
- Illumina, Inc. (U.S.)
- McKesson Corporation (U.S.)
- Infors AG (Switzerland)
- Siemens (Germany)
- Zoho Corporation Pvt. Ltd. (India)
- LabVantage Solutions Inc. (U.S.)
- CloudLIMS.com (U.S.)
What are the Recent Developments in North America Laboratory Information Management Systems (LIMS) Market?
- In October 2025, InstantGMP, Inc. launched a fully integrated Laboratory Information Management System (LIMS) software built directly into its quality management and manufacturing platform, enabling seamless data flow across QC, inventory, and production processes for pharmaceutical and biotech labs, enhancing efficiency and reducing manual data entry
- In April 2025, LabWare announced the official launch of LabWare ASSURE, a specialized SaaS LIMS platform for food and beverage labs that integrates digital control over quality, safety, and compliance workflows enhancing traceability, automation, and operational efficiency
- In March 2025, LabWare expanded its LIMS offerings by launching an expanded SaaS portfolio including LabWare ASSURE, LabWare QAQC, and LabWare GROW providing scalable SaaS‑based LIMS options designed to improve adoption, efficiency, and compliance for laboratories of all sizes across industries
- In October 2024, Xybion acquired Autoscribe Informatics, subsequently launching Xybion LIMS 10.0, a modern SaaS‑centric LIMS platform designed to enhance lab efficiency and scalability with a user‑friendly digital interface for a wide range of laboratory applications
- In August 2024, Veeva Systems announced that Veeva Vault LIMS gained significant momentum among life sciences companies by unifying quality control data and processes within a single cloud‑based platform, enabling shared data across QC workflows and improving batch release decisions
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.