Global Pediatric Vaccine Market
Market Size in USD Billion
CAGR :
% 
 USD
51.94 Billion
USD
109.72 Billion
2025
2033
USD
51.94 Billion
USD
109.72 Billion
2025
2033
| 2026 - 2033 | |
| USD 51.94 Billion | |
| USD 109.72 Billion | |
|
|
|
|
Paediatric Vaccine Market Size
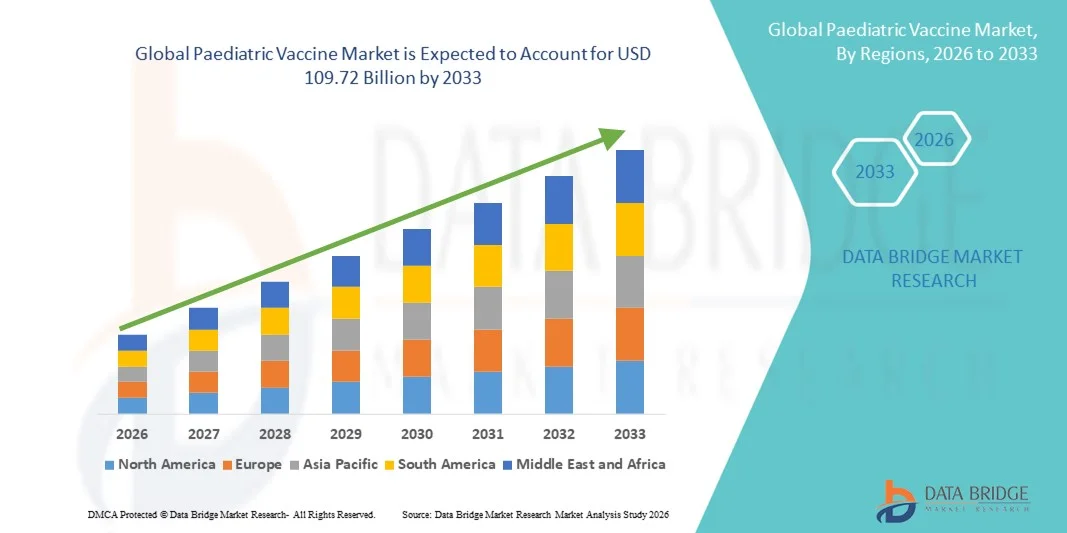
- The global paediatric vaccine market size was valued at USD 51.94 billion in 2025 and is expected to reach USD 109.72 billion by 2033, at a CAGR of 9.80% during the forecast period
- The market growth is largely fueled by expanding national immunization programs, rising government funding for childhood vaccination, and continuous advancements in vaccine development technologies, including combination and mRNA-based vaccines
- Furthermore, increasing awareness among parents regarding early disease prevention, strong support from global health organizations, and the introduction of new vaccines targeting emerging infectious diseases are establishing paediatric vaccines as a critical component of preventive healthcare. These converging factors are accelerating the demand for paediatric immunization solutions, thereby significantly boosting the industry's growth
Paediatric Vaccine Market Analysis
- Paediatric vaccines, developed to safeguard infants, children, and adolescents against a broad range of infectious and life-threatening diseases, play a central role in national immunization schedules and global public health initiatives due to their effectiveness in reducing childhood morbidity and mortality rates
- The escalating demand for paediatric vaccines is primarily fueled by expanding government-supported immunization programs, rising parental awareness regarding early disease prevention, and ongoing advancements in vaccine technologies, including improved formulations and enhanced delivery platforms
- North America dominated the paediatric vaccine market with the largest revenue share of 38.64% in 2025, characterized by high vaccination coverage, strong reimbursement systems, and the presence of leading vaccine manufacturers, with the U.S. maintaining steady demand through structured immunization schedules and continuous product innovation
- Asia-Pacific is expected to be the fastest growing region in the paediatric vaccine market during the forecast period due to large pediatric populations, expanding healthcare infrastructure, increasing public health investments, and supportive government initiatives promoting universal immunization
- Multivalent segment dominated the paediatric vaccine market with a market share of 44.3% in 2025, driven by their ability to provide protection against multiple diseases in a single administration, enhancing compliance, reducing healthcare visits, and improving overall immunization efficiency
Report Scope and Paediatric Vaccine Market Segmentation
|
Attributes |
Paediatric Vaccine Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Paediatric Vaccine Market Trends
Advancement of Combination and Next-Generation Vaccine Technologies
- A significant and accelerating trend in the global paediatric vaccine market is the increasing development of multivalent and next-generation vaccine platforms, including mRNA and recombinant technologies, aimed at improving immunogenicity, safety, and broader disease coverage. This evolution is significantly enhancing preventive healthcare outcomes among pediatric populations
- For instance, the expansion of combination vaccines that protect against multiple diseases such as DTP, Hepatitis B, and polio in a single formulation has streamlined immunization schedules and improved compliance rates. Similarly, ongoing research into mRNA-based vaccines for pediatric infectious diseases is opening new pathways for rapid vaccine development
- Technological integration in paediatric vaccines enables improved antigen design, enhanced adjuvant systems, and optimized delivery mechanisms that reduce adverse reactions while strengthening immune response. For instance, conjugate vaccine technologies have significantly improved protection against pneumococcal and meningococcal infections in children. Furthermore, innovative platforms support scalable and faster production in response to emerging infectious threats
- The seamless incorporation of advanced vaccine technologies into national immunization programs facilitates broader disease prevention strategies and more efficient public health management. Through integrated vaccination schedules, healthcare providers can administer protection against multiple diseases within fewer clinical visits, creating a more structured and accessible immunization ecosystem
- This trend toward safer, more effective, and technologically advanced paediatric vaccines is fundamentally reshaping global immunization strategies. Consequently, companies such as Pfizer and GlaxoSmithKline are investing heavily in expanding their pediatric vaccine pipelines, focusing on multivalent formulations and innovative biologics
- The demand for advanced paediatric vaccines offering broader protection and improved safety profiles is growing rapidly across both developed and emerging economies, as governments and healthcare organizations increasingly prioritize comprehensive childhood immunization coverage
- Growing focus on equitable vaccine access through global alliances and tiered pricing strategies is supporting wider distribution of essential paediatric vaccines in low- and middle-income countries
Paediatric Vaccine Market Dynamics
Driver
Expanding Government Immunization Programs and Rising Awareness of Preventive Healthcare
- The increasing emphasis on childhood immunization through national vaccination schedules and global health initiatives, coupled with rising parental awareness regarding early disease prevention, is a significant driver for the heightened demand for paediatric vaccines
- For instance, in recent years, multiple countries have expanded their universal immunization programs to include pneumococcal, rotavirus, and HPV vaccines for children, strengthening vaccine procurement and distribution networks. Such strategies by public health authorities are expected to drive paediatric vaccine market growth during the forecast period
- As awareness of vaccine-preventable diseases grows and healthcare access improves, paediatric vaccines offer proven benefits such as long-term immunity, reduced hospitalization rates, and lower childhood mortality, reinforcing their essential role in preventive medicine
- Furthermore, increasing collaborations between governments, global organizations, and vaccine manufacturers are enhancing supply chain efficiency and ensuring wider accessibility of essential pediatric vaccines across low- and middle-income regions
- The structured implementation of immunization schedules, school-entry vaccination requirements, and digital tracking systems are key factors propelling vaccine uptake in both urban and rural populations. The continued expansion of healthcare infrastructure and funding support further contributes to sustained market growth
- Rising birth rates in emerging economies and the expansion of public healthcare coverage programs are further strengthening consistent demand for paediatric vaccines worldwide
- Strategic partnerships between biotechnology firms and public health agencies are accelerating research, manufacturing scalability, and timely introduction of new pediatric vaccine formulations
Restraint/Challenge
Vaccine Hesitancy and Complex Regulatory Approval Processes
- Concerns surrounding vaccine safety perceptions, misinformation, and occasional adverse event reports pose a significant challenge to broader market expansion. As paediatric vaccines undergo rigorous clinical testing and monitoring, public skepticism in certain regions can slow adoption rates
- For instance, isolated reports of side effects or misinformation circulated through digital platforms have made some parents hesitant to follow recommended immunization schedules, affecting vaccination coverage in specific geographies
- Addressing these concerns through transparent communication, strong pharmacovigilance systems, and evidence-based awareness campaigns is crucial for maintaining public trust. Companies such as Sanofi and Merck emphasize robust clinical data and post-marketing surveillance to reassure healthcare providers and caregivers. In addition, lengthy and complex regulatory approval pathways for new paediatric vaccines can delay market entry and increase development costs
- While regulatory frameworks ensure high safety and efficacy standards, the time-intensive approval and compliance requirements can limit rapid innovation and accessibility, particularly for next-generation vaccine platforms in developing markets
- Overcoming these challenges through strengthened public education initiatives, streamlined regulatory harmonization, and sustained investment in research and manufacturing capacity will be vital for ensuring long-term growth and stability in the global paediatric vaccine market
- Cold chain storage requirements and logistical complexities in remote or underdeveloped regions can restrict vaccine distribution efficiency and increase operational costs
- Fluctuations in government funding allocations and procurement delays in public immunization programs may create supply-demand imbalances and impact manufacturer revenue predictability
Paediatric Vaccine Market Scope
The market is segmented on the basis of vaccine type, technology, and application
- By Vaccine Type
On the basis of vaccine type, the global paediatric vaccine market is segmented into monovalent and multivalent vaccines. The multivalent segment dominated the market with the largest revenue share of 44.3% in 2025, driven by its ability to provide protection against multiple diseases within a single formulation. Multivalent vaccines significantly reduce the number of injections required, improving compliance among children and parents while optimizing immunization schedules. Public health authorities strongly prefer these vaccines as they enhance coverage rates and simplify large-scale vaccination campaigns. Their cost-effectiveness in procurement, storage, and distribution further strengthens their adoption in national immunization programs. In addition, increasing development of advanced combination vaccines targeting multiple pathogens continues to reinforce the dominance of this segment.
The monovalent segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by rising demand for targeted immunization against specific emerging or high-risk diseases. Monovalent vaccines are often preferred in outbreak situations or where focused immunity against a single pathogen is required. Technological advancements have improved antigen purity and immune response specificity, supporting segment expansion. Growing research into novel single-disease pediatric vaccines, particularly for rare infectious conditions, is contributing to pipeline growth. Furthermore, regulatory flexibility and faster approval pathways for certain monovalent formulations are accelerating their market penetration.
- By Technology
On the basis of technology, the global paediatric vaccine market is segmented into live attenuated, inactivated, subunit, toxoid, conjugate, and other technologies. The conjugate segment held the largest market revenue share in 2025, driven by its strong immunogenic response and effectiveness in infants and young children. Conjugate vaccines are widely used against pneumococcal and meningococcal diseases due to their ability to enhance immune memory. Their integration into routine immunization schedules across developed and emerging economies further supports dominance. These vaccines also demonstrate improved safety profiles compared to some traditional platforms. Continuous innovation in carrier proteins and antigen combinations is expanding their applicability across multiple pediatric indications.
The subunit segment is expected to witness the fastest CAGR from 2026 to 2033, driven by advancements in recombinant DNA technology and improved safety characteristics. Subunit vaccines use specific antigens rather than whole pathogens, reducing adverse reactions and increasing parental acceptance. Growing investment in precision vaccine design and adjuvant systems is enhancing immune response efficiency. These vaccines are gaining traction for influenza and other infectious disease applications in children. In addition, their compatibility with modern manufacturing platforms supports scalable production and rapid commercialization.
- By Application
On the basis of application, the global paediatric vaccine market is segmented into infectious disease, cancer, allergy, pneumococcal disease, influenza, measles, mumps, and rubella (MMR), and other applications. The infectious disease segment dominated the market in 2025, owing to the extensive global burden of vaccine-preventable diseases among children. National immunization programs primarily focus on preventing infectious conditions such as polio, hepatitis, rotavirus, and diphtheria. Strong governmental funding and global health initiatives ensure high procurement volumes for infectious disease vaccines. Continuous surveillance of emerging pathogens further sustains consistent demand. The broad scope of diseases covered under this category significantly contributes to its leading revenue share.
The cancer segment is projected to register the fastest growth during the forecast period, supported by increasing awareness and early adoption of pediatric preventive oncology vaccines such as HPV. Expanding recommendations for adolescent immunization against virus-induced cancers are strengthening uptake rates. Research advancements in therapeutic cancer vaccines for pediatric indications are further boosting segment potential. Growing collaboration between biotechnology firms and public health agencies is accelerating product development. In addition, supportive reimbursement frameworks and preventive healthcare policies are enhancing accessibility and long-term growth prospects in this segment.
Paediatric Vaccine Market Regional Analysis
- North America dominated the paediatric vaccine market with the largest revenue share of 38.64% in 2025, characterized by high vaccination coverage, strong reimbursement systems, and the presence of leading vaccine manufacturers
- Consumers and healthcare providers in the region prioritize early disease prevention, safety, and efficacy, leading to high adoption of recommended vaccines for infants, children, and adolescents, including multivalent and combination formulations
- This widespread adoption is further supported by advanced healthcare infrastructure, efficient cold chain logistics, and active awareness campaigns, establishing paediatric vaccines as a critical component of preventive healthcare for both urban and rural populations
U.S. Paediatric Vaccine Market Insight
The U.S. paediatric vaccine market captured the largest revenue share of 82% in 2025 within North America, fueled by well-established national immunization programs and strong healthcare infrastructure. Parents and healthcare providers increasingly prioritize preventive healthcare and early immunization against multiple childhood diseases. The growing emphasis on combination vaccines and next-generation technologies, along with extensive public awareness campaigns, further propels market growth. Moreover, robust government funding, insurance coverage, and active CDC recommendations are significantly contributing to the expansion of pediatric vaccination uptake across the country.
Europe Paediatric Vaccine Market Insight
The Europe paediatric vaccine market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by comprehensive immunization programs and stringent public health policies. Rising urbanization and increasing awareness about childhood disease prevention are fostering higher vaccine adoption rates. European parents and healthcare providers are drawn to vaccines that offer broad protection and improved safety profiles. The region is witnessing significant growth across public immunization campaigns, private healthcare facilities, and school-based vaccination initiatives, ensuring wide accessibility and coverage.
U.K. Paediatric Vaccine Market Insight
The U.K. paediatric vaccine market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by strong government-backed vaccination programs and increasing parental awareness regarding early disease prevention. Concerns over infectious disease outbreaks are encouraging high compliance with recommended immunization schedules. The U.K.’s advanced healthcare system, along with strong public health campaigns and digital immunization tracking, continues to stimulate market growth. In addition, availability of combination and multivalent vaccines enhances convenience and adherence among caregivers and healthcare providers.
Germany Paediatric Vaccine Market Insight
The Germany paediatric vaccine market is expected to expand at a considerable CAGR during the forecast period, fueled by rising awareness of vaccine-preventable diseases and strong emphasis on child health. Germany’s advanced healthcare infrastructure, coupled with regulatory support for innovative vaccines, promotes adoption across both urban and rural populations. Integration of vaccines into national immunization schedules ensures high coverage rates, while parental preference for safe, effective, and combination vaccines aligns with market growth. Furthermore, public health initiatives and school-based vaccination programs are reinforcing consistent demand.
Asia-Pacific Paediatric Vaccine Market Insight
The Asia-Pacific paediatric vaccine market is poised to grow at the fastest CAGR of 23% during the forecast period of 2026 to 2033, driven by large pediatric populations, improving healthcare access, and increasing awareness of childhood disease prevention. Rising government investments in immunization programs and public health campaigns are accelerating adoption. Technological advancements in vaccine manufacturing and distribution, along with growing collaborations between domestic and multinational vaccine producers, are expanding availability. In addition, the introduction of affordable vaccines and multivalent formulations is improving access and compliance across the region.
Japan Paediatric Vaccine Market Insight
The Japan paediatric vaccine market is gaining momentum due to the country’s advanced healthcare infrastructure, high parental awareness, and emphasis on preventive pediatric care. Vaccination coverage is supported by strong government recommendations and school-based programs. The adoption of combination and next-generation vaccines is driving increased efficiency and convenience for caregivers. Furthermore, Japan’s focus on child health and safety, along with its well-developed cold chain logistics, is fueling demand in both public and private healthcare settings.
India Paediatric Vaccine Market Insight
The India paediatric vaccine market accounted for the largest market revenue share in Asia-Pacific in 2025, attributed to a large child population, increasing healthcare awareness, and expanding immunization programs. India is one of the fastest-growing markets for vaccines due to strong government initiatives, such as the Universal Immunization Program, and rising private healthcare penetration. Affordable vaccines, widespread vaccination campaigns, and the presence of domestic manufacturers are key factors propelling market growth. Furthermore, improving cold chain infrastructure and digital health tracking are enhancing access and coverage across urban and rural regions.
Paediatric Vaccine Market Share
The Paediatric Vaccine industry is primarily led by well-established companies, including:
- Pfizer Inc. (U.S.)
- Sanofi (France)
- GSK plc (U.K.)
- Merck & Co., Inc. (U.S.)
- AstraZeneca (U.K.)
- Novavax, Inc. (U.S.)
- Bharat Biotech (India)
- Serum Institute of India Pvt. Ltd. (India)
- Panacea Biotec Ltd. (India)
- CSL Limited (Australia)
- Sinovac Biotech Ltd. (China)
- Indian Immunologicals Ltd. (India)
- Bio Med Pvt. Ltd. (India)
- Daiichi Sankyo Co., Ltd. (Japan)
- Johnson & Johnson Services, Inc. (U.S.)
- Biological E Limited (India)
- Takeda Pharmaceutical Company Limited (Japan)
- Moderna, Inc. (U.S.)
- Bavarian Nordic A/S (Denmark)
- SK Bioscience Co., Ltd. (South Korea)
What are the Recent Developments in Global Paediatric Vaccine Market?
- In November 2025, the World Health Organization (WHO) declared the end of the poliovirus outbreak in Indonesia after nearly three years of intensified vaccination campaigns, highlighting the success of paediatric immunisation efforts and the role of increased vaccine administration in eradicating vaccine‑preventable diseases
- In June 2025, the U.S. Food and Drug Administration (FDA) approved Merck’s preventive antibody shot Enflonsia (clesrovimab) to protect infants up to one year of age against respiratory syncytial virus (RSV), marking a significant expansion of paediatric prevention tools for a major infant respiratory pathogen
- In May 2025, the U.S. FDA approved Sanofi’s meningococcal vaccine MenQuadfi for use in infants as young as six weeks old, providing earlier protection against four common strains of meningococcal bacteria and expanding paediatric immunisation options
- In August 2024, UNICEF delivered 60,000 doses of the PENTA‑Hib combined vaccine to children in Ukraine, protecting them against five serious diseases diphtheria, tetanus, pertussis, hepatitis B, and Haemophilus influenzae type b demonstrating the use of multivalent paediatric vaccines to improve immunisation coverage in conflict‑affected settings
- In April 2024, Valneva announced that it had vaccinated the first participant in the pediatric Phase 2 clinical trial of its single‑shot chikungunya vaccine, advancing efforts to address an unmet vaccine need among children for this mosquito‑borne disease
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.