Market Analysis and Insights: Europe Induced Pluripotent Stem Cells (iPSCs) Market
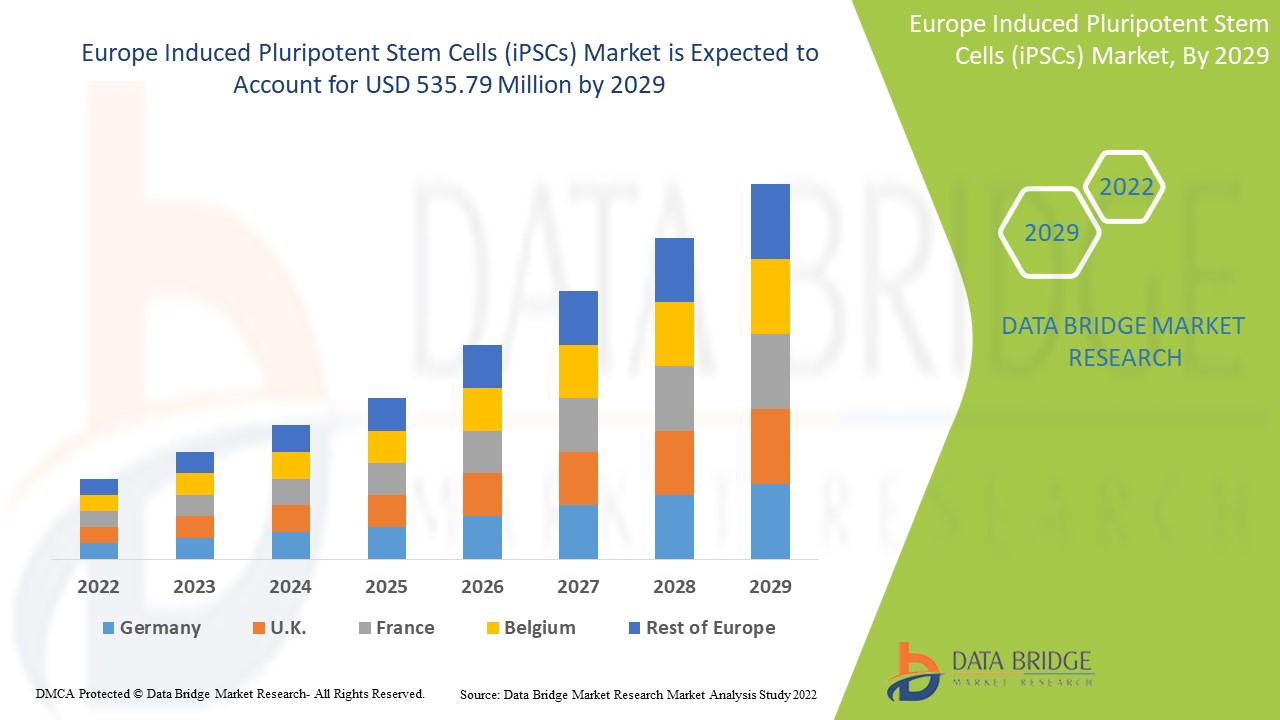
The Europe induced pluripotent stem cells (iPSCs) market is expected to gain market growth in the forecast period of 2022 to 2029. Data Bridge Market Research analyses that the market is growing with a CAGR of 8.9% in the forecast period of 2022 to 2029 and is expected to reach USD 535.79 million by 2029. Increasing research activities over stem cell therapies act as driver for the induced pluripotent stem cells (iPSCs) market growth.
The induced pluripotent stem cells are type of cells derived from the adult somatic tissues and re-programmed with set of genes and factors to gain pluripotent nature. Certain genes and factors are added to attain definite properties of embryonic stem cells. The induced pluripotent cells being nearly identical to cell donor they aid in disease modelling. Retroviruses are commonly used as vectors to reprogram induced pluripotent stem cells. Main applications of induced pluripotent stem cells are in disease modelling, drug discovery and development, toxicity studies and gene therapies. They are widely used in treatments for cardiovascular diseases, diabetes mellitus and various types of cancer. Human induced pluripotent stem cells shows the relevant properties of disease as it carries the specific genotype of the disease, thereby enables novel therapeutic options in patient specific way.
Increasing adoption of stem cell therapy, growing biotechnology sector with better investment and rising prevalence of chronic diseases acts as driver for the induced pluripotent stem cells (iPSCs) market. The other factors which are anticipated to propel the growth of the Europe induced pluripotent stem cells (iPSCs) market include the Wide range of clinical application of induced pluripotent stem cells and emerging technological advancements of iPSCs.
However, the factors such as the high cost associated with stem cell therapies, availability of alternatives for tumor treatment are hampering the growth of the Europe induced pluripotent stem cells (iPSCs) market. On the other hand, the rising number of pipeline products, increased interest of personalized medicine and surge in healthcare expenditure act as an opportunity for the growth of the Europe induced pluripotent stem cells (iPSCs) market. The stringent rules and regulations and Genomic instability of IPSC is the key market challenge faced in the Europe induced pluripotent stem cells (iPSCs) market.
The induced pluripotent stem cells (iPSCs) market report provides details of market share, new developments, and product pipeline analysis, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, product approvals, strategic decisions, product launches, geographic expansions, and technological innovations in the market. To understand the analysis and the induced pluripotent stem cells (iPSCs) market scenario contact Data Bridge Market Research for an Analyst Brief, our team will help you create a revenue impact solution to achieve your desired goal.
Induced Pluripotent Stem Cells (iPSCs) Market Scope and Market Size
The induced pluripotent stem cells (iPSCs) market is segmented on basis of cell source, type, product, applications, end users and distribution channel The growth among segments helps you analyze niche pockets of growth and strategies to approach the market and determine your core application areas and the difference in your target markets.
Europe induced pluripotent stem cells (iPSCs) market is categorized into six notable segments based on the cell source, type, product, applications, end users and distribution channel.
- On the basis of cell source, the Europe induced pluripotent stem cells (iPSCs) market is segmented into skin cells and blood cells. In 2022, skin cells segment is expected to dominate the market due to wide availability of skin cells sources and high awareness of the same.
- On the basis of type, the Europe induced pluripotent stem cells (iPSCs) market is segmented into human IPSCs and mouse IPSCs. In 2022, human IPSCs segment is expected to dominate the market due to expanding geriatric populace and increasing number of clinical trials in France.
- On the basis of product, the Europe induced pluripotent stem cells (iPSCs) market is segmented into instruments, consumables & kits and services. In 2022, consumables & kits segment is expected to dominate the market due to increasing R&D in the United Kingdom owing to presence of experts in developmental and reproduction biology.
- On the basis of application, the Europe induced pluripotent stem cells (iPSCs) market is segmented into academic research, regenerative medicine, cellular therapy, toxicology screening, drug discovery and development, disease modelling, stem cell banking, 3D bioprinting and others. In 2022, drug discovery and development segment is expected to dominate the market as outsourcing of iPSCs trials has eventually bolstered the process of drug approva.
- On the basis of end-users, the Europe induced pluripotent stem cells (iPSCs) market is segmented into biotechnology & pharmaceutical companies, research laboratories, diagnostic laboratories and others. In 2022, biotechnology & pharmaceutical companies segment is expected to dominate the market as region has recently become the hub for small and large-scale biopharmaceutical companies and has become dependent on the contract research organization sector and other clinical services for research and development operations.
- On the basis of distribution channel, the Europe induced pluripotent stem cells (iPSCs) market is segmented into direct tender and retail sales. In 2022, direct tender segment is expected to dominate the market due to the high number of delivery sources.
Induced Pluripotent Stem Cells (iPSCs) Market Country Level Analysis
The induced pluripotent stem cells (iPSCs) market is analyzed and market size information is provided by the country, cell source, type, product, applications, end users and distribution channel as referenced above.
The countries covered in the induced pluripotent stem cells (iPSCs) market report are the U.K., Germany, France, Spain, Italy, Netherlands, Switzerland, Russia, Turkey, Austria, Ireland, and Rest of Europe.
Human iPSCs segment in Germany of Europe region is expected to grow with the highest growth rate in the forecast period of 2022 to 2029 because of increasing usage of stem cell technology. Human iPSCs segment in France is dominating the European market owing to increasing cases of chronic diseases and high adoption of stem cell sources for better therapies. U.K. is leading the growth of the market and human iPSCs segment is dominating in this country due to increasing number of biotechnology centers and research activities.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points such as new sales, replacement sales, country demographics, regulatory acts and import-export tariffs are some of the major pointers used to forecast the market scenario for individual countries. Also, presence and availability of Europe brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of sales channels are considered while providing forecast analysis of the country data.
Growing Strategic Activities by Major Market Players to Enhance the Awareness for Induced Pluripotent Stem Cells (iPSCs) Treatment, is Boosting the Market Growth of Induced Pluripotent Stem Cells (iPSCs) Market.
The induced pluripotent stem cells (iPSCs) market also provides you with detailed market analysis for every country growth in particular market. Additionally, it provides the detail information regarding the market players’ strategy and their geographical presence. The data is available for historic period 2011 to 2020.
Competitive Landscape and Induced Pluripotent Stem Cells (iPSCs) Market Share Analysis
Induced pluripotent stem cells (iPSCs) market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, production sites and facilities, company strengths and weaknesses, product launch, product trials pipelines, product approvals, patents, product width and breadth, application dominance, technology lifeline curve. The above data points provided are only related to the company’s focus related to induce pluripotent stem cells (iPSCs) market.
The major companies which are dealing in the induced pluripotent stem cells (iPSCs) are Thermo Fisher Scientific Inc., FUJIFILM Corporation, LumaCyte, Horizon Discovery Ltd., Takara Bio Inc., Lonza., Evotec SE., Axol Bioscience Ltd., R & D Systems, Inc., Charles River Laboratories International, Inc., Corning Incorporated, REPROCELL Inc., Merck KGaA and among others other domestic players. DBMR analysts understand competitive strengths and provide competitive analysis for each competitor separately.
Many contract and agreement are also initiated by the companies’ worldwide which are also accelerating the induced pluripotent stem cells (iPSCs) market.
For instance,
- In February 2021, Thermo Fisher Scientific Inc. announced it has won six awards in the annual CMO Leadership Awards. The awards, presented by Life Science Leader and Outsourced Pharma, recognize top contract manufacturing partners as evaluated by biopharma and biotech companies. It is estimated that this recognition anticipated to strengthen company’s footprints in Europe market and leading to upsurge Company’s growth in the coming years.
- In June 2020, the LumaCyte collaborated with Catalent who are the Europe provider of advanced delivery technologies, development, and manufacturing solutions for drugs, biologics, cell and gene therapies, and consumer health products. This collaboration aided in expanding the company’s stem cell technology product Radiance and its application.
Collaboration, product launch, business expansion, award and recognition, joint ventures and other strategies by the market player is enhancing the company footprints in the veterinary infusion pumps market which also provides the benefit for organization’s profit growth.
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Table of Content
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET
1.4 LIMITATIONS
1.5 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 CURRENCY AND PRICING
2.5 DBMR TRIPOD DATA VALIDATION MODEL
2.6 MULTIVARIATE MODELLING
2.7 CELL SOURCE LIFELINE CURVE
2.8 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.9 DBMR MARKET POSITION GRID
2.1 MARKET APPLICATION COVERAGE GRID
2.11 VENDOR SHARE ANALYSIS
2.12 SECONDARY SOURCES
2.13 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
5 EUROPE MEDICAL CARTS MARKET: REGULATIONS
5.1 REGULATION IN U.S.
5.2 REGULATION IN CANADA
5.3 REGULATION IN EUROPE
5.4 REGULATION IN INDIA
5.5 REGUALTION IN JAPAN
6 MARKET OVERVIEW
6.1 DRIVERS
6.1.1 WIDE RANGE OF CLINICAL APPLICATION OF INDUCED PLURIPOTENT STEM CELLS
6.1.2 EMERGING TECHNOLOGICAL ADVANTAGES OF IPSCS
6.1.3 RISING PREVALENCE OF SEVERAL CHRONIC DISEASES
6.1.4 INCREASING ADOPTION OF STEM CELL THERAPY
6.1.5 GROWING BIOTECHNOLOGY SECTOR WITH BETTER INVESTMENT
6.2 RESTRAINT
6.2.1 HIGH COST ASSOCIATED WITH STEM CELL THERAPIES AND LARGE-SCALE APPLICATIONS OF IPSCS
6.2.2 AVAILABILITY OF ALTERNATIVES FOR TUMOR TREATMENT
6.2.3 ADVERSE EFFECTS OF STEM CELL TRANSPLANTS
6.3 OPPORTUNITIES
6.3.1 INCREASING NUMBER OF PIPELINE PRODUCTS
6.3.2 INCREASING INTEREST OF PERSONALIZED MEDICINE
6.3.3 SURGE IN HEALTHCARE EXPENDITURE
6.3.4 STRATEGIC INITIATIVES BY KEY MARKET PLAYERS
6.4 CHALLENGES
6.4.1 GENOMIC INSTABILITY OF IPSCS IS THE KEY MARKET CHALLENGE
6.4.2 LACK OF SKILLED PROFESSIONALS
6.4.3 STRINGENT REGULATORY FRAMEWORK
7 IMPACT OF COVID-19 ON THE EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET
7.1 IMPACT ON PRICE
7.2 IMPACT ON DEMAND
7.3 IMPACT ON SUPPLY CHAIN
7.4 STRATEGIC DECISIONS BY MANUFACTURERS
7.5 CONCLUSION
8 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE
8.1 OVERVIEW
8.2 SKIN CELLS
8.2.1 FIBROBLAST
8.2.2 KERATINOCYTES
8.2.3 ADIPOSE DERIVED STEM CELLS
8.2.4 HEPATOCYTES
8.2.5 MELANOCYTES
8.2.6 NEURAL STEM CELLS
8.2.7 OTHERS
8.3 BLOOD CELLS
8.3.1 PERIPHERAL BLOOD
8.3.2 CORD BLOOD ENDOTHELIAL CELLS
8.3.3 CORD BLOOD STEM CELLS
8.3.4 OTHERS
9 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE
9.1 OVERVIEW
9.2 HUMAN IPSCS
9.3 MOUSE IPSCS
10 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT
10.1 OVERVIEW
10.2 CONSUMABLES & KITS
10.2.1 REPROGRAMMING KITS
10.2.2 MEDIA
10.2.3 TRANSFECTION KITS
10.2.4 CELL IDENTIFICATION KITS
10.2.5 ACCESSORIES
10.2.6 OTHERS
10.3 SERVICES
10.4 INSTRUMENTS
10.4.1 IMAGING SYSTEMS
10.4.2 ELECTROPORATION DEVICE
10.4.3 INCUBATORS
10.4.4 OTHERS
11 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION
11.1 OVERVIEW
11.2 DRUG DISCOVERY AND DEVELOPMENT
11.3 ACADEMIC RESEARCH
11.4 DISEASE MODELLING
11.5 CELLULAR THERAPY
11.6 REGENERATIVE MEDICINE
11.7 TOXICOLOGY SCREENING
11.8 STEM CELL BANKING
11.9 3D BIOPRINTING
11.1 OTHERS
12 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER
12.1 OVERVIEW
12.2 BIOTECHNOLOGY & PHARMACEUTICAL COMPANIES
12.3 RESEARCH LABORATORIES
12.4 DIAGNOSTIC LABORATORIES
12.5 OTHERS
13 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL
13.1 OVERVIEW
13.2 DIRECT TENDER
13.3 RETAIL SALES
14 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION
14.1 EUROPE
14.1.1 GERMANY
14.1.2 FRANCE
14.1.3 U.K.
14.1.4 ITALY
14.1.5 RUSSIA
14.1.6 SPAIN
14.1.7 TURKEY
14.1.8 NETHERLANDS
14.1.9 SWITZERLAND
14.1.10 BELGIUM
14.1.11 REST OF EUROPE
15 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: COMPANY LANDSCAPE
15.1 COMPANY SHARE ANALYSIS: EUROPE
16 SWOT ANALYSIS
17 COMPANY PROFILE
17.1 FUJIFILM CORPORATION
17.1.1 COMPANY SNAPSHOT
17.1.2 REVENUE ANALYSIS
17.1.3 COMPANY SHARE ANALYSIS
17.1.4 PRODUCT PORTFOLIO
17.1.5 RECENT DEVELOPMENT
17.1.5.1 ACQUISITION
17.2 THERMO FISHER SCIENTIFIC INC.
17.2.1 COMPANY SNAPSHOT
17.2.2 REVENUE ANALYSIS
17.2.3 COMPANY SHARE ANALYSIS
17.2.4 PRODUCT PORTFOLIO
17.2.5 RECENT DEVELOPMENTS
17.2.5.1 EVENT
17.2.5.2 ACQUISITION
17.3 LONZA.
17.3.1 COMPANY SNAPSHOT
17.3.2 REVENUE ANALYSIS
17.3.3 COMPANY SHARE ANALYSIS
17.3.4 PRODUCT PORTFOLIO
17.3.5 RECENT DEVELOPMENTS
17.3.5.1 EXPANSION
17.4 MERCK KGAA
17.4.1 COMPANY SNAPSHOT
17.4.2 REVENUE ANALYSIS
17.4.3 COMPANY SHARE ANALYSIS
17.4.4 PRODUCT PORTFOLIO
17.4.5 RECENT DEVELOPMENT
17.4.5.1 AGREEMENT
17.5 EVOTEC SE.
17.5.1 COMPANY SNAPSHOT
17.5.2 REVENUE ANALYSIS
17.5.3 COMPANY SHARE ANALYSIS
17.5.4 PRODUCT PORTFOLIO
17.5.5 RECENT DEVELOPMENTS
17.5.5.1 AGREEMENT
17.5.5.2 COLLABORATION
17.6 APPLIED STEMCELL.
17.6.1 COMPANY SNAPSHOT
17.6.2 PRODUCT PORTFOLIO
17.6.3 RECENT DEVELOPMENTS
17.6.3.1 PRODUCT LAUNCH
17.7 AXOL BIOSCIENCE LTD.
17.7.1 COMPANY SNAPSHOT
17.7.2 PRODUCT PORTFOLIO
17.7.3 RECENT DEVELOPMENTS
17.7.3.1 MERGER
17.7.3.2 PRODUCT LAUNCH
17.8 CELL APPLICATIONS, INC.
17.8.1 COMPANY SNAPSHOT
17.8.2 PRODUCT PORTFOLIO
17.8.3 RECENT DEVELOPMENT
17.8.3.1 PARTNERSHIP
17.9 CHARLES RIVER LABORATORIES INTERNATIONAL, INC.
17.9.1 COMPANY SNAPSHOT
17.9.2 REVENUE ANALYSIS
17.9.3 PRODUCT PORTFOLIO
17.9.4 RECENT DEVELOPMENT
17.9.4.1 ACQUISITION
17.1 CITIUS PHARMACEUTICALS, INC.
17.10.1 COMPANY SNAPSHOT
17.10.2 PRODUCT PORTFOLIO
17.10.3 RECENT DEVELOPMENT
17.10.3.1 AGREEMENT
17.11 CORNING INCORPORATED
17.11.1 COMPANY SNAPSHOT
17.11.2 REVENUE ANALYSIS
17.11.3 PRODUCT PORTFOLIO
17.11.4 RECENT DEVELOPMENT
17.11.4.1 AGREEMENT
17.12 FATE THERAPEUTICS
17.12.1 COMPANY SNAPSHOT
17.12.2 PRODUCT PORTFOLIO
17.12.3 RECENT DEVELOPMENT
17.12.3.1 CLINICAL TRIAL
17.13 GENECOPOEIA, INC.
17.13.1 COMPANY SNAPSHOT
17.13.2 PRODUCT PORTFOLIO
17.13.3 RECENT DEVELOPMENT
17.14 HOPSTEM BIOTECHNOLOGY LLC.
17.14.1 COMPANY SNAPSHOT
17.14.2 PRODUCT PORTFOLIO
17.14.3 RECENT DEVELOPMENT
17.14.3.1 PARTNERSHIP
17.15 HORIZON DISCOVERY LTD.
17.15.1 COMPANY SNAPSHOT
17.15.2 PRODUCT PORTFOLIO
17.15.3 RECENT DEVELOPMENT
17.16 LUMACYTE
17.16.1 COMPANY SNAPSHOT
17.16.2 PRODUCT PORTFOLIO
17.16.3 RECENT DEVELOPMENT
17.16.3.1 COLLABORATION
17.17 R & D SYSTEMS, INC.
17.17.1 COMPANY SNAPSHOT
17.17.2 REVENUE ANALYSIS
17.17.3 PRODUCT PORTFOLIO
17.17.4 RECENT DEVELOPMENT
17.18 REPROCELL INC.
17.18.1 COMPANY SNAPSHOT
17.18.2 PRODUCT PORTFOLIO
17.18.3 RECENT DEVELOPMENTS
17.18.3.1 COLLABORATION
17.18.3.2 FACILITY EXPANSION
17.18.3.3 SERVICE LAUNCH
17.19 TAKARA BIO INC.
17.19.1 COMPANY SNAPSHOT
17.19.2 REVENUE ANALYSIS
17.19.3 PRODUCT PORTFOLIO
17.19.4 RECENT DEVELOPMENTS
17.19.4.1 NEW FACILITY LAUNCH
17.2 UNIVERSAL CELLS INC. (AN ASTELLAS COMPANY)
17.20.1 COMPANY SNAPSHOT
17.20.2 REVENUE ANALYSIS
17.20.3 PRODUCT PORTFOLIO
17.20.4 RECENT DEVELOPMENT
17.20.4.1 ACQUISITION
18 QUESTIONNAIRE
19 RELATED REPORTS
List of Table
TABLE 1 NEW CANCER CASES, AGES 85+, IN THE U.S.
TABLE 2 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 3 EUROPE SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 4 EUROPE SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 5 EUROPE BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 6 EUROPE BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 7 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 8 EUROPE HUMAN IPSCS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 9 EUROPE MOUSE IPSCS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 10 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 11 EUROPE CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 12 EUROPE CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 13 EUROPE SERVICES IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 14 EUROPE INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 15 EUROPE INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 16 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 17 EUROPE DRUG DISCOVERY AND DEVELOPMENT IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 18 EUROPE ACADEMIC RESEARCH IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 19 EUROPE DISEASE MODELLING IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 20 EUROPE CELLULAR THERAPY IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 21 EUROPE REGENERATIVE MEDICINE IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 22 EUROPE TOXICOLOGY SCREENING IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 23 EUROPE STEM CELL BANKING IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 24 EUROPE 3D BIOPRINTING IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 25 EUROPE OTHERS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 26 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 27 EUROPE BIOTECHNOLOGY & PHARMACEUTICAL COMPANIES IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 28 EUROPE RESEARCH LABORATORIES IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 29 EUROPE DIAGNOSTIC LABORATORIES IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 30 EUROPE OTHERS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 31 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 32 EUROPE DIRECT TENDER IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 33 EUROPE RETAIL SALES IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 34 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY COUNTRY, 2020-2029 (USD MILLION)
TABLE 35 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 36 EUROPE SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 37 EUROPE BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 38 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 39 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 40 EUROPE INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 41 EUROPE CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 42 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 43 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 44 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 45 GERMANY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 46 GERMANY SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 47 GERMANY BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 48 GERMANY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 49 GERMANY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 50 GERMANY INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 51 GERMANY CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 52 GERMANY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 53 GERMANY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 54 GERMANY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 55 FRANCE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 56 FRANCE SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 57 FRANCE BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 58 FRANCE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 59 FRANCE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 60 FRANCE INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 61 FRANCE CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 62 FRANCE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 63 FRANCE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 64 FRANCE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 65 U.K. INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 66 U.K. SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 67 U.K. BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 68 U.K. INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 69 U.K. INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 70 U.K. INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 71 U.K. CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 72 U.K. INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 73 U.K. INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 74 U.K. INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 75 ITALY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 76 ITALY SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 77 ITALY BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 78 ITALY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 79 ITALY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 80 ITALY INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 81 ITALY CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 82 ITALY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 83 ITALY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 84 ITALY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 85 RUSSIA INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 86 RUSSIA SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 87 RUSSIA BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 88 RUSSIA INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 89 RUSSIA INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 90 RUSSIA INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 91 RUSSIA CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 92 RUSSIA INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 93 RUSSIA INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 94 RUSSIA INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 95 SPAIN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 96 SPAIN SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 97 SPAIN BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 98 SPAIN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 99 SPAIN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 100 SPAIN INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 101 SPAIN CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 102 SPAIN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 103 SPAIN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 104 SPAIN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 105 TURKEY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 106 TURKEY SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 107 TURKEY BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 108 TURKEY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 109 TURKEY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 110 TURKEY INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 111 TURKEY CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 112 TURKEY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 113 TURKEY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 114 TURKEY INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 115 NETHERLANDS INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 116 NETHERLANDS SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 117 NETHERLANDS BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 118 NETHERLANDS INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 119 NETHERLANDS INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 120 NETHERLANDS INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 121 NETHERLANDS CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 122 NETHERLANDS INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 123 NETHERLANDS INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 124 NETHERLANDS INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 125 SWITZERLAND INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 126 SWITZERLAND SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 127 SWITZERLAND BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 128 SWITZERLAND INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 129 SWITZERLAND INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 130 SWITZERLAND INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 131 SWITZERLAND CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 132 SWITZERLAND INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 133 SWITZERLAND INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 134 SWITZERLAND INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 135 BELGIUM INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 136 BELGIUM SKIN CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 137 BELGIUM BLOOD CELLS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
TABLE 138 BELGIUM INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 139 BELGIUM INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 140 BELGIUM INSTRUMENTS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 141 BELGIUM CONSUMABLES & KITS IN INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY PRODUCT, 2020-2029 (USD MILLION)
TABLE 142 BELGIUM INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 143 BELGIUM INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 144 BELGIUM INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 145 REST OF EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, BY CELL SOURCE, 2020-2029 (USD MILLION)
List of Figure
FIGURE 1 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: SEGMENTATION
FIGURE 2 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: DATA TRIANGULATION
FIGURE 3 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: DROC ANALYSIS
FIGURE 4 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: EUROPE VS REGIONAL MARKET ANALYSIS
FIGURE 5 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: DBMR MARKET POSITION GRID
FIGURE 8 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: MARKET APPLICATION COVERAGE GRID
FIGURE 9 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: VENDOR SHARE ANALYSIS
FIGURE 10 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: SEGMENTATION
FIGURE 11 THE WIDE RANGE OF CLINICAL APPLICATION OF INDUCED PLURIPOTENT STEM CELLS (IPSC) ARE EXPECTED TO DRIVE THE EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET IN THE FORECAST PERIOD OF 2022 TO 2029
FIGURE 12 SKIN CELLS SEGMENT IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET IN 2022 & 2029
FIGURE 13 NORTH AMERICA IS EXPECTED TO DOMINATE THE EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET, AND ASIA-PACIFIC IS EXPECTED TO GROW WITH THE HIGHEST CAGR IN THE FORECAST PERIOD OF 2022 TO 2029
FIGURE 14 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET
FIGURE 15 PREVALENCE OF CHRONIC DISEASES
FIGURE 16 NUMBER OF PEOPLE WITH DIABETES (MILLION) AMONG AGES 20–79 YEARS
FIGURE 17 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY CELL SOURCE, 2021
FIGURE 18 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY CELL SOURCE, 2020-2029 (USD MILLION)
FIGURE 19 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY CELL SOURCE, CAGR (2022-2029)
FIGURE 20 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY CELL SOURCE, LIFELINE CURVE
FIGURE 21 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY TYPE, 2021
FIGURE 22 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY TYPE, 2020-2029 (USD MILLION)
FIGURE 23 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY TYPE, CAGR (2022-2029)
FIGURE 24 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY TYPE, LIFELINE CURVE
FIGURE 25 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY PRODUCT, 2021
FIGURE 26 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY PRODUCT, 2020-2029 (USD MILLION)
FIGURE 27 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY PRODUCT, CAGR (2022-2029)
FIGURE 28 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY PRODUCT, LIFELINE CURVE
FIGURE 29 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY APPLICATION, 2021
FIGURE 30 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY APPLICATION, 2020-2029 (USD MILLION)
FIGURE 31 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY APPLICATION, CAGR (2022-2029)
FIGURE 32 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY APPLICATION, LIFELINE CURVE
FIGURE 33 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY END USER, 2021
FIGURE 34 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY END USER, 2020-2029 (USD MILLION)
FIGURE 35 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY END USER, CAGR (2022-2029)
FIGURE 36 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY END USER, LIFELINE CURVE
FIGURE 37 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY DISTRIBUTION CHANNEL, 2021
FIGURE 38 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
FIGURE 39 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY DISTRIBUTION CHANNEL, CAGR (2022-2029)
FIGURE 40 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 41 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: SNAPSHOT (2021)
FIGURE 42 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY COUNTRY (2021)
FIGURE 43 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY COUNTRY (2022 & 2029)
FIGURE 44 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY COUNTRY (2021 & 2029)
FIGURE 45 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: BY CELL SOURCE (2022-2029)
FIGURE 46 EUROPE INDUCED PLURIPOTENT STEM CELLS (IPSCS) MARKET: COMPANY SHARE 2021 (%)

Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.