Marché nord-américain du diagnostic du cancer du col de l'utérus, par type de produit (test d'imagerie, test de dépistage, examen visuel, biopsies cervicales et autres procédures), groupe d'âge (moins de 21 ans, 21-29 ans, 30-65 ans, 65 ans et plus), stades (stade I, stade II, stade III, stade IV), utilisateurs finaux (hôpitaux, laboratoires de diagnostic, cliniques spécialisées, centres de santé communautaires, organisation de recherche sur le cancer, centres de cancérologie et de radiothérapie), canal de distribution (appel d'offres direct, ventes au détail, ventes en ligne) - Tendances et prévisions de l'industrie jusqu'en 2030.
Analyse et perspectives du marché du diagnostic du cancer du col de l'utérus en Amérique du Nord
Le marché nord-américain du diagnostic du cancer du col de l'utérus connaît une croissance au cours de l'année de prévision en raison de l'augmentation du nombre d'acteurs du marché et de la disponibilité de divers produits et marques de diagnostic du cancer du col de l'utérus. Parallèlement à cela, les acteurs du marché sont engagés dans des diagnostics avancés du cancer du col de l'utérus. La prévalence croissante du cancer du col de l'utérus devrait encore stimuler la croissance du marché. Cependant, les règles et réglementations strictes pourraient freiner la croissance du marché au cours de la période de prévision. Les diverses collaborations gouvernementales et privées, l'augmentation des activités de R&D et les initiatives stratégiques des acteurs du marché offrent des opportunités au marché. Cependant, les faux résultats des tests de dépistage du cancer du col de l'utérus devraient constituer un défi majeur pour la croissance du marché.
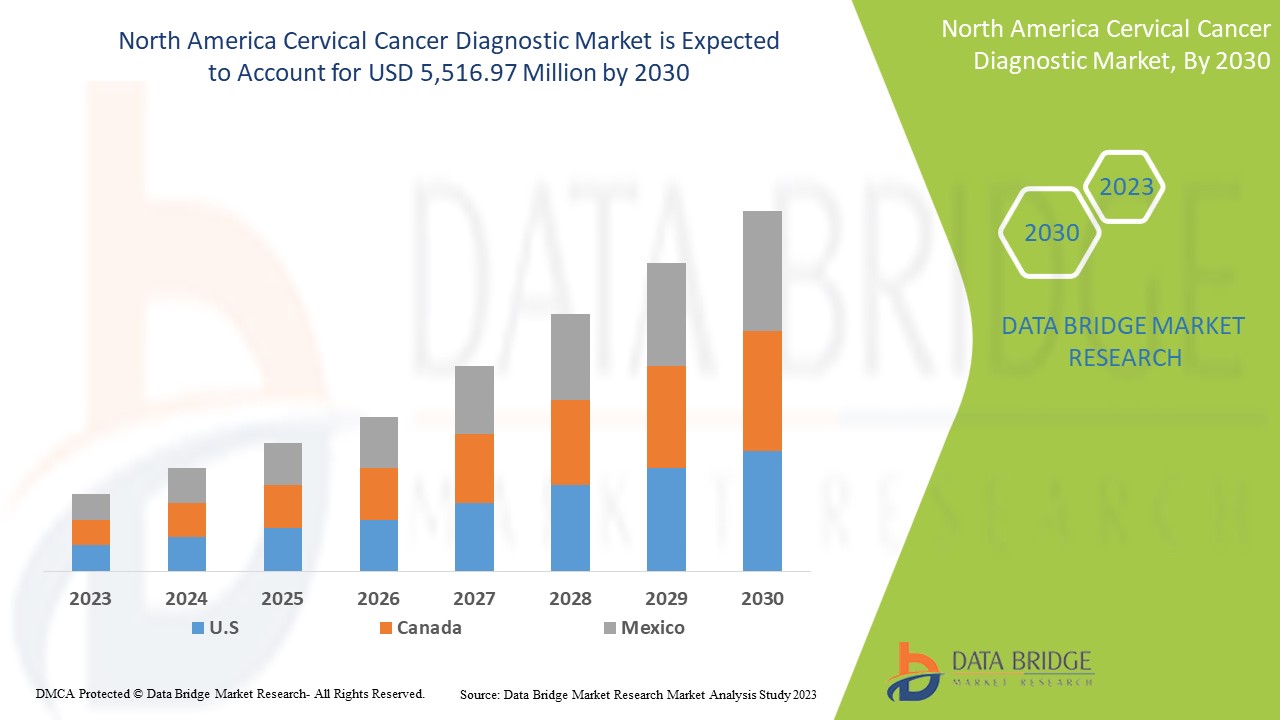
Le marché du diagnostic du cancer du col de l'utérus en Amérique du Nord devrait connaître une croissance du marché au cours de la période de prévision de 2023 à 2030. Data Bridge Market Research analyse que le marché croît avec un TCAC de 7,0 % au cours de la période de prévision de 2023 à 2030 et devrait atteindre 5 516,97 millions USD d'ici 2030.
|
Rapport métrique |
Détails |
|
Période de prévision |
2023 à 2030 |
|
Année de base |
2022 |
|
Années historiques |
2021 (personnalisable pour 2020-2015) |
|
Unités quantitatives |
Chiffre d'affaires en millions USD |
|
Segments couverts |
Par type de produit (test d'imagerie, test de dépistage, examen visuel, biopsies cervicales et autres procédures), groupe d'âge (moins de 21 ans, 21-29 ans, 30-65 ans, 65 ans et plus), stades (stade I, stade II, stade III, stade IV), utilisateurs finaux (hôpitaux, laboratoires de diagnostic, cliniques spécialisées, centres de santé communautaires, organismes de recherche sur le cancer, centres de cancérologie et de radiothérapie), canal de distribution (appel d'offres direct, ventes au détail, ventes en ligne) |
|
Pays couverts |
États-Unis, Canada, Mexique |
|
Acteurs du marché couverts |
Siemens Healthcare GmbH, BD, F. Hoffmann-La Roche Ltd, Abbott, Hologic, Inc., Quest Diagnostics Incorporated, Bio-Rad Laboratories, Inc., QIAGEN, The Cooper Companies Inc., Seegene Inc., Sysmex Corporation, MobileODT, Zilico, Jiangsu Mole Bioscience Co. Ltd., Guided Therapeutics, Inc., GenomeMe Lab Inc., Arbor Vita Corporation et LCM GENECT Srl, entre autres |
Définition du marché :
Le cancer du col de l’utérus est un type de cancer qui se développe dans le col de l’utérus de l’appareil reproducteur féminin. Sa prévalence augmente à mesure que le nombre de patientes infectées par le VPH augmente et que l’accent est mis sur la détection et le traitement précoces, ce qui devrait accélérer le développement du diagnostic du cancer du col de l’utérus. L’augmentation des investissements publics dans la sensibilisation à la détection précoce du cancer et l’augmentation des dépenses de santé stimuleraient également la croissance des entreprises. La cause la plus courante du cancer du col de l’utérus est l’infection par le VPH (virus du papillome humain). D’autres choix de vie peuvent augmenter le risque, notamment le tabagisme, l’alcool, un régime pauvre en fruits et légumes, la prise de pilules contraceptives et les relations sexuelles à l’adolescence. Étant donné que le cancer du col de l’utérus est traitable une fois diagnostiqué à un stade précoce, les femmes à risque de développer la maladie doivent subir des tests de routine pour identifier la maladie à un stade précoce, ce qui permet au marché de se développer.
Dynamique du marché du diagnostic du cancer du col de l'utérus en Amérique du Nord
Cette section traite de la compréhension des moteurs, des opportunités, des contraintes et des défis du marché. Tout cela est discuté en détail ci-dessous :
Conducteurs
-
Sensibilisation accrue au diagnostic précoce du cancer du col de l'utérus
Il existe une très grande variété de facteurs de risque pour le cancer du col de l'utérus. Par conséquent, la sensibilisation à son diagnostic a augmenté ces dernières années. Différents tests de diagnostic sont disponibles, tels que le test PAP (Papanicolaou), le test du papillomavirus humain , la colposcopie , les biopsies cervicales et la cystoscopie, entre autres. Par conséquent, pour réduire les facteurs de risque, un diagnostic précoce est très important.
-
Augmentation de la prévalence et de l’incidence du cancer du col de l’utérus
Le cancer du col de l'utérus est un type de cancer qui se développe dans le col de l'utérus de l'appareil reproducteur féminin. Le développement irrégulier de cellules cancéreuses dans le tissu du col de l'utérus définit souvent le cancer du col de l'utérus. L'adénocarcinome ou le carcinome épidermoïde peuvent se développer à partir du cancer du col de l'utérus. La cause la plus courante du cancer du col de l'utérus est l'infection par le VPH (virus du papillome humain). Le cancer du col de l'utérus est classé en deux types : l'adénocarcinome et le carcinome épidermoïde. Le cancer du col de l'utérus est diagnostiqué à l'aide d'une variété de tests, d'outils et de procédures de laboratoire avancés qui évaluent les cellules et les souches anormales du virus du papillome humain (VPH).
Opportunités
-
Augmentation des dépenses de santé
Les dépenses de santé ont augmenté dans le monde entier à mesure que le revenu disponible des citoyens de divers pays augmentait. De plus, pour répondre aux besoins de la population, les organismes gouvernementaux et les organisations de santé prennent des initiatives pour accélérer les dépenses de santé. L'augmentation des dépenses de santé aide simultanément les établissements de santé à améliorer leurs installations de traitement pour le diagnostic du cancer du col de l'utérus, car cette maladie est très répandue ces dernières années.
En outre, les initiatives stratégiques prises par les principaux acteurs du marché assureront l’intégrité structurelle et les opportunités futures du marché du diagnostic du cancer du col de l’utérus en Amérique du Nord au cours de la période de prévision.
Contraintes/Défis
Cependant, les médicaments de diagnostic du cancer du col de l’utérus peuvent avoir des effets secondaires indésirables ; il est difficile d’équilibrer les risques avec les avantages du traitement en raison des effets secondaires croissants des médicaments contre le cancer qui entravent la demande du marché.
De plus, l’approbation croissante des vaccins contre le VPH par les autorités réglementaires devrait freiner la croissance du marché nord-américain du diagnostic du cancer du col de l’utérus. Ces développements se produisent rapidement dans le monde entier pour réduire les cas de cancer du col de l’utérus, ce qui peut constituer un facteur de restriction pour le marché.
L'utilisation de divers médicaments de traitement à travers le monde augmente rapidement et, avec la prévalence croissante du cancer du col de l'utérus, il est nécessaire de procéder à un diagnostic et à un traitement rapides. Dans le même temps, les fabricants de diagnostics du cancer du col de l'utérus sur le marché doivent suivre certaines réglementations pour obtenir l'approbation des autorités supérieures pour lancer le produit sur le marché. Ces directives strictes doivent être respectées ; c'est l'une des tâches les plus difficiles de toutes les étapes. L'approbation préalable à la mise sur le marché de divers médicaments varie d'un pays à l'autre.
Développement récent
- En août 2020, Siemens Healthcare GmbH a conclu un accord d'acquisition avec Varian Medical Systems, Inc. ; avec cette acquisition, Siemens Healthcare a contribué à développer des solutions avancées pour traiter le cancer et à renforcer sa position dans le secteur de la santé
Portée du marché du diagnostic du cancer du col de l'utérus en Amérique du Nord
Le marché nord-américain du diagnostic du cancer du col de l'utérus est segmenté en type de produit, groupe d'âge, stades, utilisateurs finaux et canal de distribution. La croissance parmi ces segments vous aidera à analyser les segments de faible croissance dans les industries et à fournir aux utilisateurs un aperçu précieux du marché et des informations sur le marché pour prendre des décisions stratégiques afin d'identifier les principales applications du marché.
Type de produit
- Test d'imagerie
- Test de dépistage
- Examen visuel
- Biopsies cervicales
- Autres procédures
Sur la base du type de produit, le marché nord-américain du diagnostic du cancer du col de l’utérus est segmenté en test d’imagerie, test de dépistage, examen visuel, biopsies cervicales et autres procédures.
Groupe d'âge
- Moins de 21 ans
- 21-29
- 30-65
- 65 ans et plus
Sur la base de la tranche d'âge, le marché du diagnostic du cancer du col de l'utérus en Amérique du Nord est segmenté en moins de 21 ans, 21-29 ans, 30-65 ans et 65 ans et plus.
Étapes
- Étape I
- Stade II
- Stade III
- Stade IV
Sur la base du stade, le marché nord-américain du diagnostic du cancer du col de l’utérus est segmenté en stade I, stade II, stade III et stade IV.
Utilisateurs finaux
- Centres de cancérologie et de radiothérapie
- Hôpitaux
- Cliniques spécialisées
- Organisation de recherche sur le cancer
- Laboratoires de diagnostic
- Centres de santé communautaires
Sur la base des utilisateurs finaux, le marché nord-américain du diagnostic du cancer du col de l’utérus est segmenté en hôpitaux, laboratoires de diagnostic, cliniques spécialisées, centres de santé communautaires, organisations de recherche sur le cancer et centres de cancérologie et de radiothérapie.
Canal de distribution
- Appel d'offres direct
- Ventes au détail
- Ventes en ligne

Sur la base du canal de distribution, le marché du diagnostic du cancer du col de l’utérus en Amérique du Nord est segmenté en appels d’offres directs, ventes au détail et ventes en ligne.
Analyse/perspectives régionales du marché du diagnostic du cancer du col de l'utérus
Le marché du diagnostic du cancer du col de l’utérus en Amérique du Nord est analysé et des informations et tendances sur la taille du marché sont fournies par pays, type de produit, tranche d’âge, étapes, utilisateurs finaux et canal de distribution comme référencé ci-dessus.
Certains pays couverts sur le marché nord-américain du diagnostic du cancer du col de l’utérus sont les États-Unis, le Canada et le Mexique.
Les États-Unis devraient dominer le marché nord-américain du diagnostic du cancer du col de l'utérus en termes de part de marché et de chiffre d'affaires et continueront de renforcer leur domination au cours de la période de prévision. Cela est dû à la sensibilisation croissante au diagnostic précoce du cancer du col de l'utérus.
La section par pays du rapport fournit également des facteurs individuels ayant un impact sur le marché et des changements dans la réglementation du marché qui ont un impact sur les tendances actuelles et futures du marché. Des points de données, tels que les ventes de produits neufs et de remplacement, la démographie des pays, l'épidémiologie des maladies et les tarifs d'importation et d'exportation, sont quelques-uns des principaux indicateurs utilisés pour prévoir le scénario de marché pour les différents pays. En outre, la présence et la disponibilité des marques nord-américaines et les défis auxquels elles sont confrontées en raison de la forte concurrence des marques locales et nationales et l'impact des canaux de vente sont pris en compte tout en fournissant une analyse prévisionnelle des données nationales.
Analyse du paysage concurrentiel et des parts de marché du diagnostic du cancer du col de l'utérus en Amérique du Nord
Le paysage concurrentiel du marché du diagnostic du cancer du col de l'utérus en Amérique du Nord fournit des détails sur les concurrents. Les détails comprennent un aperçu de l'entreprise, les finances de l'entreprise, les revenus générés, le potentiel du marché, les investissements dans la recherche et le développement, les nouvelles initiatives du marché, la présence en Amérique du Nord, les sites et installations de production, les capacités de production, les forces et les faiblesses de l'entreprise, le lancement du produit, la largeur et l'étendue du produit et la domination des applications. Les points de données ci-dessus ne concernent que les entreprises se concentrant sur le marché du diagnostic du cancer du col de l'utérus en Amérique du Nord.
Certains des principaux acteurs opérant sur le marché du diagnostic du cancer du col de l'utérus en Amérique du Nord sont Siemens Healthcare GmbH, BD, F. Hoffmann-La Roche Ltd, Abbott, Hologic, Inc., Quest Diagnostics Incorporated, Bio-Rad Laboratories, Inc., QIAGEN, The Cooper Companies Inc., Seegene Inc., Sysmex Corporation, MobileODT, Zilico, Jiangsu Mole Bioscience Co. Ltd., Guided Therapeutics, Inc., GenomeMe Lab Inc., Arbor Vita Corporation et LCM GENECT Srl, entre autres.
SKU-
Accédez en ligne au rapport sur le premier cloud mondial de veille économique
- Tableau de bord d'analyse de données interactif
- Tableau de bord d'analyse d'entreprise pour les opportunités à fort potentiel de croissance
- Accès d'analyste de recherche pour la personnalisation et les requêtes
- Analyse de la concurrence avec tableau de bord interactif
- Dernières actualités, mises à jour et analyse des tendances
- Exploitez la puissance de l'analyse comparative pour un suivi complet de la concurrence
Table des matières
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET
1.4 LIMITATIONS
1.5 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 CURRENCY AND PRICING
2.5 DBMR TRIPOD DATA VALIDATION MODEL
2.6 MULTIVARIATE MODELLING
2.7 PRODUCT TYPE LIFELINE CURVE
2.8 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.9 DBMR MARKET POSITION GRID
2.1 MARKET END USER COVERAGE GRID
2.11 VENDOR SHARE ANALYSIS
2.12 SECONDARY SOURCES
2.13 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PESTEL ANALYSIS
4.2 PORTER'S FIVE FORCES MODEL
4.3 INDUSTRY INSIGHTS:
4.3.1 CERVICAL CANCER DIAGNOSIS
4.3.2 STRATEGIC INITIATIVES BY MARKET PLAYERS
5 REGULATORY FRAMEWORK
6 MARKET OVERVIEW
6.1 DRIVERS
6.1.1 INCREASING PREVALENCE AND INCIDENCE OF CERVICAL CANCER
6.1.2 RISING AWARENESS OF EARLY DIAGNOSIS OF CERVICAL CANCER
6.1.3 HIGH PREVALENCE OF HPV-INFECTED PATIENTS AND RISING INCIDENCE OF TEENAGE SEXUAL ENCOUNTERS
6.2 RESTRAINTS
6.2.1 DEVELOPMENT IN THE FIELD OF HPV VACCINE
6.2.2 SIDE EFFECTS OF TREATMENT DRUGS
6.3 OPPORTUNITIES
6.3.1 RISING HEALTHCARE EXPENDITURE
6.3.2 STRATEGIC INITIATIVES BY MARKET PLAYERS
6.4 CHALLENGES
6.4.1 STRINGENT RULES AND REGULATIONS
6.4.2 FALSE RESULTS IN SCREENING TESTS AND UNAVAILABILITY OF IMPROVED HEALTHCARE INFRASTRUCTURE
7 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE
7.1 OVERVIEW
7.2 SCREENING TEST
7.2.1 HPV TEST
7.2.1.1 ASSAYS
7.2.1.2 KITS/REAGENTS
7.2.2 PAP TEST
7.2.2.1 ASSAYS
7.2.2.2 KITS/REAGENTS
7.2.3 BLOOD TEST
7.3 IMAGING TEST
7.3.1 PET CT-SCAN
7.3.2 MAGNETIC RESONANCE IMAGING (MRI)
7.3.3 ULTRASOUND
7.3.4 X-RAY
7.3.5 OTHERS
7.4 CERVICAL BIOPSIES
7.4.1 LIQUID BIOPSY
7.4.2 ENDOCERVICAL CURETTAGE
7.4.3 COLPOSCOPIC BIOPSY
7.5 VISUAL EXAMINATION
7.5.1 CYSTOSCOPY
7.5.2 SIGMOIDOSCOPY
7.6 OTHER PROCEDURES
8 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY AGE GROUP
8.1 OVERVIEW
8.2 30-65
8.3 65 AND ABOVE
8.4 21-29
8.5 BELOW 21
9 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY STAGES
9.1 OVERVIEW
9.2 STAGE I
9.3 STAGE II
9.4 STAGE III
9.5 STAGE IV
10 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY END USER
10.1 OVERVIEW
10.2 CANCER AND RADIATION THERAPY CENTERS
10.3 HOSPITALS
10.4 SPECIALTY CLINICS
10.5 CANCER RESEARCH ORGANIZATION
10.6 DIAGNOSTIC LABORATORIES
10.7 COMMUNITY HEALTH CENTERS
11 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL
11.1 OVERVIEW
11.2 DIRECT TENDER
11.3 RETAIL SALES
11.4 ONLINE SALES
12 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION
12.1 NORTH AMERICA
12.1.1 U.S.
12.1.2 CANADA
12.1.3 MEXICO
13 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: COMPANY LANDSCAPE
13.1 COMPANY SHARE ANALYSIS: NORTH AMERICA
14 SWOT ANALYSIS
15 COMPANY PROFILE
15.1 SIEMENS HEALTHCARE GMBH
15.1.1 COMPANY SNAPSHOT
15.1.2 REVENUE ANALYSIS
15.1.3 COMPANY SHARE ANALYSIS
15.1.4 PRODUCT PORTFOLIO
15.1.5 RECENT DEVELOPMENT
15.2 BD
15.2.1 COMPANY SNAPSHOT
15.2.2 REVENUE ANALYSIS
15.2.3 COMPANY SHARE ANALYSIS
15.2.4 PRODUCT PORTFOLIO
15.2.5 RECENT DEVELOPMENT
15.3 F. HOFFMANN- LA ROCHE LTD
15.3.1 COMPANY SNAPSHOT
15.3.2 REVENUE ANALYSIS
15.3.3 COMPANY SHARE ANALYSIS
15.3.4 PRODUCT PORTFOLIO
15.3.5 RECENT DEVELOPMENT
15.4 ABBOTT
15.4.1 COMPANY SNAPSHOT
15.4.2 REVENUE ANALYSIS
15.4.3 COMPANY SHARE ANALYSIS
15.4.4 PRODUCT PORTFOLIO
15.4.5 RECENT DEVELOPMENTS
15.5 HOLOGIC, INC.
15.5.1 COMPANY SNAPSHOT
15.5.2 REVENUE ANALYSIS
15.5.3 COMPANY SHARE ANALYSIS
15.5.4 PRODUCT PORTFOLIO
15.5.5 RECENT DEVELOPMENTS
15.6 ARBOR VITA CORPORATION
15.6.1 COMPANY SNAPSHOT
15.6.2 PRODUCT PORTFOLIO
15.6.3 RECENT DEVELOPMENTS
15.7 BIO-RAD LABORATORIES, INC.
15.7.1 COMPANY SNAPSHOT
15.7.2 REVENUE ANALYSIS
15.7.3 PRODUCT PORTFOLIO
15.7.4 RECENT DEVELOPMENT
15.8 GENOMEME LAB INC.
15.8.1 COMPANY SNAPSHOT
15.8.2 PRODUCT PORTFOLIO
15.8.3 RECENT DEVELOPMENTS
15.9 GUIDED THERAPEUTICS, INC
15.9.1 COMPANY SNAPSHOT
15.9.2 REVENUE ANALYSIS
15.9.3 PRODUCT PORTFOLIO
15.9.4 RECENT DEVELOPMENT
15.1 JIANGSU MOLE BIOSCIENCE CO., LTD.
15.10.1 COMPANY SNAPSHOT
15.10.2 PRODUCT PORTFOLIO
15.10.3 RECENT DEVELOPMENTS
15.11 LCM GENECT SRL
15.11.1 COMPANY SNAPSHOT
15.11.2 PRODUCT PORTFOLIO
15.11.3 RECENT DEVELOPMENT
15.12 MOBILEODT
15.12.1 COMPANY SNAPSHOT
15.12.2 PRODUCT PORTFOLIO
15.12.3 RECENT DEVELOPMENT
15.13 QIAGEN
15.13.1 COMPANY SNAPSHOT
15.13.2 REVENUE ANALYSIS
15.13.3 PRODUCT PORTFOLIO
15.13.4 RECENT DEVELOPMENTS
15.14 QUEST DIAGNOSTICS INCORPORATED
15.14.1 COMPANY SNAPSHOT
15.14.2 REVENUE ANALYSIS
15.14.3 PRODUCT PORTFOLIO
15.14.4 RECENT DEVELOPMENTS
15.15 SEEGENE INC.
15.15.1 COMPANY SNAPSHOT
15.15.2 PRODUCT PORTFOLIO
15.15.3 RECENT DEVELOPMENTS
15.16 SYSMEX CORPORATION
15.16.1 COMPANY SNAPSHOT
15.16.2 PRODUCT PORTFOLIO
15.16.3 RECENT DEVELOPMENT
15.17 THE COOPER COMPANIES INC
15.17.1 COMPANY SNAPSHOT
15.17.2 REVENUE ANALYSIS
15.17.3 PRODUCT PORTFOLIO
15.17.4 RECENT DEVELOPMENT
15.18 ZILICO
15.18.1 COMPANY SNAPSHOT
15.18.2 PRODUCT PORTFOLIO
15.18.3 RECENT DEVELOPMENT
16 QUESTIONNAIRE
17 RELATED REPORTS
Liste des tableaux
TABLE 1 CERVICAL CANCER CAN BE DIAGNOSED BY VARIOUS TESTS AND BIOPSIES, AS LISTED BELOW:
TABLE 2 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 3 NORTH AMERICA SCREENING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 4 NORTH AMERICA SCREENING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 5 NORTH AMERICA HPV TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 6 NORTH AMERICA PAP TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 7 NORTH AMERICA IMAGING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 8 NORTH AMERICA IMAGING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 9 NORTH AMERICA CERVICAL BIOPSIES IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 10 NORTH AMERICA CERVICAL BIOPSIES IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 11 NORTH AMERICA VISUAL EXAMINATION IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 12 NORTH AMERICA VISUAL EXAMINATION IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 13 NORTH AMERICA OTHER PROCEDURES IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 14 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY AGE GROUP, 2021-2030 (USD MILLION)
TABLE 15 NORTH AMERICA 30-65 IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 16 NORTH AMERICA 65 AND ABOVE IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 17 NORTH AMERICA 21-29 IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 18 NORTH AMERICA BELOW 21 IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 19 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY STAGES, 2021-2030 (USD MILLION)
TABLE 20 NORTH AMERICA STAGE I IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 21 NORTH AMERICA STAGE II IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 22 NORTH AMERICA STAGE III IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 23 NORTH AMERICA STAGE IV IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 24 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 25 NORTH AMERICA CANCER AND RADIATION THERAPY CENTERS IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 26 NORTH AMERICA HOSPITALS IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 27 NORTH AMERICA SPECIALTY CLINICS IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 28 NORTH AMERICA CANCER RESEARCH ORGANIZATION IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 29 NORTH AMERICA DIAGNOSTIC LABORATORIES IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 30 NORTH AMERICA COMMUNITY HEALTH CENTERS IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 31 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 32 NORTH AMERICA DIRECT TENDER IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 33 NORTH AMERICA RETAIL SALES IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 34 NORTH AMERICA ONLINE SALES IN CERVICAL CANCER DIAGNOSTIC MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 35 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY COUNTRY, 2021-2030 (USD MILLION)
TABLE 36 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 37 NORTH AMERICA SCREENING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 38 NORTH AMERICA HPV TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 39 NORTH AMERICA PAP TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 40 NORTH AMERICA IMAGING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 41 NORTH AMERICA CERVICAL BIOPSIES IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 42 NORTH AMERICA VISUAL EXAMINATION IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 43 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY AGE GROUP, 2021-2030 (USD MILLION)
TABLE 44 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY STAGES, 2021-2030 (USD MILLION)
TABLE 45 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 46 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 47 U.S. CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 48 U.S. SCREENING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 49 U.S. HPV TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 50 U.S. PAP TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 51 U.S. IMAGING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 52 U.S. CERVICAL BIOPSIES IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 53 U.S. VISUAL EXAMINATION IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 54 U.S. CERVICAL CANCER DIAGNOSTIC MARKET, BY AGE GROUP, 2021-2030 (USD MILLION)
TABLE 55 U.S. CERVICAL CANCER DIAGNOSTIC MARKET, BY STAGES, 2021-2030 (USD MILLION)
TABLE 56 U.S. CERVICAL CANCER DIAGNOSTIC MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 57 U.S. CERVICAL CANCER DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 58 CANADA CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 59 CANADA SCREENING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 60 CANADA HPV TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 61 CANADA PAP TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 62 CANADA IMAGING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 63 CANADA CERVICAL BIOPSIES IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 64 CANADA VISUAL EXAMINATION IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 65 CANADA CERVICAL CANCER DIAGNOSTIC MARKET, BY AGE GROUP, 2021-2030 (USD MILLION)
TABLE 66 CANADA CERVICAL CANCER DIAGNOSTIC MARKET, BY STAGES, 2021-2030 (USD MILLION)
TABLE 67 CANADA CERVICAL CANCER DIAGNOSTIC MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 68 CANADA CERVICAL CANCER DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 69 MEXICO CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 70 MEXICO SCREENING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 71 MEXICO HPV TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 72 MEXICO PAP TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 73 MEXICO IMAGING TEST IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 74 MEXICO CERVICAL BIOPSIES IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 75 MEXICO VISUAL EXAMINATION IN CERVICAL CANCER DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2021-2030 (USD MILLION)
TABLE 76 MEXICO CERVICAL CANCER DIAGNOSTIC MARKET, BY AGE GROUP, 2021-2030 (USD MILLION)
TABLE 77 MEXICO CERVICAL CANCER DIAGNOSTIC MARKET, BY STAGES, 2021-2030 (USD MILLION)
TABLE 78 MEXICO CERVICAL CANCER DIAGNOSTIC MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 79 MEXICO CERVICAL CANCER DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
Liste des figures
FIGURE 1 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: SEGMENTATION
FIGURE 2 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: DATA TRIANGULATION
FIGURE 3 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: DROC ANALYSIS
FIGURE 4 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: NORTH AMERICA VS REGIONAL MARKET ANALYSIS
FIGURE 5 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: DBMR MARKET POSITION GRID
FIGURE 8 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: MARKET END USER COVERAGE GRID
FIGURE 9 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: VENDOR SHARE ANALYSIS
FIGURE 10 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: SEGMENTATION
FIGURE 11 THE RISING PREVALENCE AND INCIDENCE OF CERVICAL CANCER ARE EXPECTED TO DRIVE THE NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET IN THE FORECAST PERIOD
FIGURE 12 SCREENING TEST SEGMENT IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET IN 2023 AND 2030
FIGURE 13 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF THE NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET
FIGURE 14 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET : BY PRODUCT TYPE, 2022
FIGURE 15 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET : BY PRODUCT TYPE, 2023-2030 (USD MILLION)
FIGURE 16 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET : BY PRODUCT TYPE, CAGR (2023-2030)
FIGURE 17 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET : BY PRODUCT TYPE, LIFELINE CURVE
FIGURE 18 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET : BY AGE GROUP 2022
FIGURE 19 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET : BY AGE GROUP, 2023-2030 (USD MILLION)
FIGURE 20 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET : BY AGE GROUP, CAGR (2023-2030)
FIGURE 21 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET : BY AGE GROUP, LIFELINE CURVE
FIGURE 22 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY STAGES, 2022
FIGURE 23 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY STAGES, 2023-2030 (USD MILLION)
FIGURE 24 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY STAGES, CAGR (2023-2030)
FIGURE 25 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET : BY STAGES, LIFELINE CURVE
FIGURE 26 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY END USER, 2022
FIGURE 27 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY END USER, 2023-2030 (USD MILLION)
FIGURE 28 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY END USER, CAGR (2023-2030)
FIGURE 29 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET : BY END USER, LIFELINE CURVE
FIGURE 30 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY DISTRIBUTION CHANNEL, 2022
FIGURE 31 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY DISTRIBUTION CHANNEL, 2023-2030 (USD MILLION)
FIGURE 32 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY DISTRIBUTION CHANNEL, CAGR (2023-2030)
FIGURE 33 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 34 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: SNAPSHOT (2022)
FIGURE 35 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY COUNTRY (2022)
FIGURE 36 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY COUNTRY (2023 & 2030)
FIGURE 37 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: BY COUNTRY (2022 & 2030)
FIGURE 38 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: PRODUCT TYPE (2023-2030)
FIGURE 39 NORTH AMERICA CERVICAL CANCER DIAGNOSTIC MARKET: COMPANY SHARE 2022 (%)

Méthodologie de recherche
La collecte de données et l'analyse de l'année de base sont effectuées à l'aide de modules de collecte de données avec des échantillons de grande taille. L'étape consiste à obtenir des informations sur le marché ou des données connexes via diverses sources et stratégies. Elle comprend l'examen et la planification à l'avance de toutes les données acquises dans le passé. Elle englobe également l'examen des incohérences d'informations observées dans différentes sources d'informations. Les données de marché sont analysées et estimées à l'aide de modèles statistiques et cohérents de marché. De plus, l'analyse des parts de marché et l'analyse des tendances clés sont les principaux facteurs de succès du rapport de marché. Pour en savoir plus, veuillez demander un appel d'analyste ou déposer votre demande.
La méthodologie de recherche clé utilisée par l'équipe de recherche DBMR est la triangulation des données qui implique l'exploration de données, l'analyse de l'impact des variables de données sur le marché et la validation primaire (expert du secteur). Les modèles de données incluent la grille de positionnement des fournisseurs, l'analyse de la chronologie du marché, l'aperçu et le guide du marché, la grille de positionnement des entreprises, l'analyse des brevets, l'analyse des prix, l'analyse des parts de marché des entreprises, les normes de mesure, l'analyse globale par rapport à l'analyse régionale et des parts des fournisseurs. Pour en savoir plus sur la méthodologie de recherche, envoyez une demande pour parler à nos experts du secteur.
Personnalisation disponible
Data Bridge Market Research est un leader de la recherche formative avancée. Nous sommes fiers de fournir à nos clients existants et nouveaux des données et des analyses qui correspondent à leurs objectifs. Le rapport peut être personnalisé pour inclure une analyse des tendances des prix des marques cibles, une compréhension du marché pour d'autres pays (demandez la liste des pays), des données sur les résultats des essais cliniques, une revue de la littérature, une analyse du marché des produits remis à neuf et de la base de produits. L'analyse du marché des concurrents cibles peut être analysée à partir d'une analyse basée sur la technologie jusqu'à des stratégies de portefeuille de marché. Nous pouvons ajouter autant de concurrents que vous le souhaitez, dans le format et le style de données que vous recherchez. Notre équipe d'analystes peut également vous fournir des données sous forme de fichiers Excel bruts, de tableaux croisés dynamiques (Fact book) ou peut vous aider à créer des présentations à partir des ensembles de données disponibles dans le rapport.













