Global Medical Device Regulatory Affairs Outsourcing Market
Marktgröße in Milliarden USD
CAGR :
% 
 USD
7.36 Billion
USD
19.30 Billion
2024
2032
USD
7.36 Billion
USD
19.30 Billion
2024
2032
| 2025 –2032 | |
| USD 7.36 Billion | |
| USD 19.30 Billion | |
|
|
|
|
Globale Marktsegmentierung für das Outsourcing von regulatorischen Angelegenheiten im Bereich Medizinprodukte nach Dienstleistungen (Dienstleistungen im Bereich regulatorische Angelegenheiten, Qualitätsberatung, Verfassen und Veröffentlichen von regulatorischen Angelegenheiten, Einreichung von Unterlagen für regulatorische Angelegenheiten, Anträge für klinische Studien, Produktregistrierungen, Rechtsvertretung und medizinisches Schreiben), Produkt (Fertigprodukte, Elektronik und Rohstoffe), Gerätetyp (Klasse I, Klasse II und Klasse III), Kategorie (Biologika, Arzneimittel und Medizinprodukte), Anwendung (Kardiologie, diagnostische Bildgebung, Orthopädie, IVD, Augenheilkunde, allgemeine und plastische Chirurgie, Arzneimittelverabreichung, Zahnmedizin, Endoskopie, Diabetesversorgung und andere), Endbenutzer (kleine Medizinproduktehersteller, mittlere Medizinproduktehersteller, Pharma- und Biotechnologieunternehmen, Medizinproduktehersteller und große Medizinproduktehersteller) – Branchentrends und Prognose bis 2032
Outsourcing der regulatorischen Angelegenheiten für Medizinprodukte Marktgröße
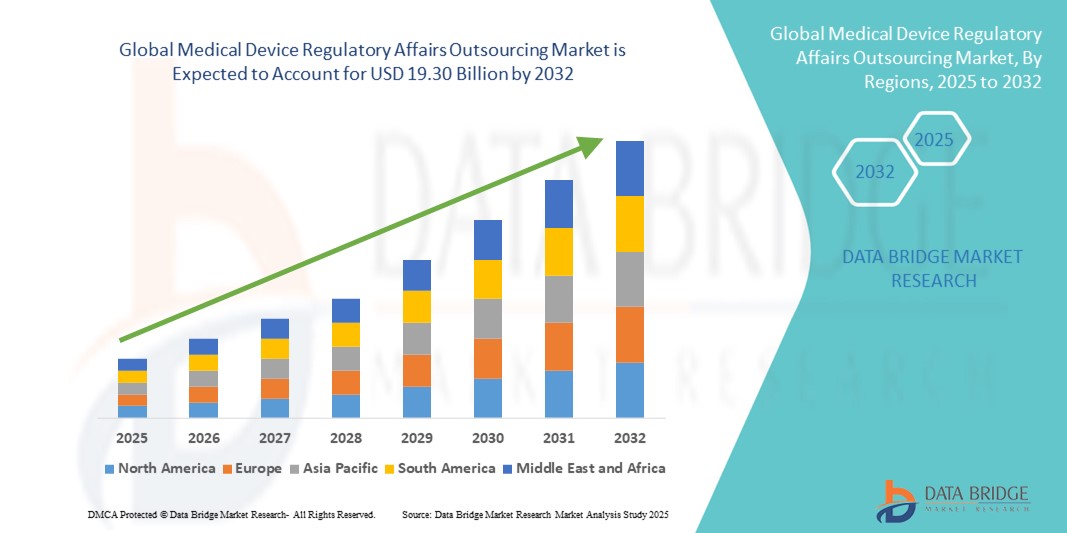
- Der globale Markt für das Outsourcing von regulatorischen Angelegenheiten im Bereich Medizinprodukte wurde im Jahr 2024 auf 7,36 Milliarden US-Dollar geschätzt und dürfte bis 2032 19,30 Milliarden US-Dollar erreichen , bei einer CAGR von 12,8 % im Prognosezeitraum.
- Das Marktwachstum ist vor allem auf die zunehmende Komplexität globaler regulatorischer Anforderungen und die steigende Nachfrage nach schnelleren Produktzulassungen zurückzuführen. Hersteller medizinischer Geräte verlassen sich daher zunehmend auf spezialisierte regulatorische Outsourcing-Dienstleistungen.
- Darüber hinaus zwingen wachsende Forschungs- und Entwicklungsaktivitäten, häufige Aktualisierungen der Compliance-Standards und der Druck, die Markteinführungszeit zu verkürzen, Unternehmen dazu, mit externen Regulierungsexperten zusammenzuarbeiten. Diese Faktoren beschleunigen die Einführung von Outsourcing-Lösungen und tragen maßgeblich zur Expansion des Marktes bei.
Marktanalyse für das Outsourcing regulatorischer Angelegenheiten im Bereich Medizinprodukte
- Das Outsourcing von regulatorischen Angelegenheiten für Medizinprodukte beinhaltet die Delegierung von regulatorischen Aufgaben wie Produktregistrierung, Konformitätsdokumentation und der Zusammenarbeit mit Behörden an spezialisierte Drittunternehmen. Dies wird zu einer entscheidenden Strategie für Hersteller, die sich effizient und kostengünstig in zunehmend komplexen globalen Regulierungsumgebungen zurechtfinden möchten.
- Die steigende Nachfrage nach regulatorischem Outsourcing ist vor allem auf die ständigen Aktualisierungen der globalen Compliance-Anforderungen, die Notwendigkeit einer beschleunigten Marktzulassung und den Mangel an interner regulatorischer Expertise bei kleinen und mittleren Geräteherstellern zurückzuführen.
- Nordamerika dominierte den Outsourcing-Markt für regulatorische Angelegenheiten im Bereich Medizinprodukte mit dem größten Umsatzanteil von 39,2 % im Jahr 2024, unterstützt durch die ausgereifte Medizinprodukteindustrie der Region, einen starken regulatorischen Rahmen und eine hohe Outsourcing-Durchdringung bei US-amerikanischen Herstellern, die schnellere FDA-Zulassungen anstreben.
- Der asiatisch-pazifische Raum wird im Prognosezeitraum voraussichtlich das schnellste Marktwachstum verzeichnen. Dies ist auf die wachsende Produktionsbasis für Medizinprodukte in der Region, sich entwickelnde regulatorische Rahmenbedingungen und Kostenvorteile zurückzuführen, die lokale CROs und Regulierungsberatungen bieten.
- Das Segment des Verfassens und Veröffentlichens von regulatorischen Dokumenten dominierte den Markt mit einem Anteil von 42 % im Jahr 2024. Dies ist auf seine entscheidende Rolle bei der Sicherstellung klarer und konformer Einreichungen bei Aufsichtsbehörden zurückzuführen, was sich direkt auf die Genehmigungszeiträume und den Erfolg der Produkteinführung auswirkt.
Berichtsumfang und Marktsegmentierung für das Outsourcing regulatorischer Angelegenheiten im Bereich Medizinprodukte
|
Eigenschaften |
Wichtige Markteinblicke im Outsourcing regulatorischer Angelegenheiten für Medizinprodukte |
|
Abgedeckte Segmente |
|
|
Abgedeckte Länder |
Nordamerika
Europa
Asien-Pazifik
Naher Osten und Afrika
Südamerika
|
|
Wichtige Marktteilnehmer |
|
|
Marktchancen |
|
|
Wertschöpfungsdaten-Infosets |
Zusätzlich zu den Einblicken in Marktszenarien wie Marktwert, Wachstumsrate, Segmentierung, geografische Abdeckung und wichtige Akteure enthalten die von Data Bridge Market Research kuratierten Marktberichte auch ausführliche Expertenanalysen, Preisanalysen, Markenanteilsanalysen, Verbraucherumfragen, demografische Analysen, Lieferkettenanalysen, Wertschöpfungskettenanalysen, eine Übersicht über Rohstoffe/Verbrauchsmaterialien, Kriterien für die Lieferantenauswahl, PESTLE-Analysen, Porter-Analysen und regulatorische Rahmenbedingungen. |
Markttrends für das Outsourcing regulatorischer Angelegenheiten im Bereich Medizinprodukte
„Digitale Transformation und KI-gestützte regulatorische Lösungen“
- Ein bedeutender und sich beschleunigender Trend im globalen Outsourcing-Markt für Medizinprodukte-Zulassungen ist die zunehmende Nutzung digitaler Technologien und künstlicher Intelligenz (KI) zur Verbesserung regulatorischer Prozesse und der Compliance-Effizienz. Dieser digitale Wandel rationalisiert die Abläufe und verändert die Art und Weise, wie Hersteller komplexe, länderübergreifende Zulassungsanträge bearbeiten.
- So setzen beispielsweise Regulierungsdienstleister wie Freyr und IQVIA KI-gestützte Plattformen für die Automatisierung regulatorischer Dokumente, die Echtzeit-Verfolgung von Einreichungen und prädiktive regulatorische Intelligenz ein, um schnellere und präzisere Entscheidungen zu ermöglichen.
- Die KI-Integration ermöglicht die automatisierte Zusammenstellung technischer Unterlagen, die Identifizierung regulatorischer Lücken und die proaktive Einhaltung sich ändernder globaler Standards. Diese Funktionen reduzieren die Markteinführungszeit und den Ressourcenaufwand für Hersteller erheblich.
- Die Nutzung zentralisierter digitaler Plattformen verbessert die Zusammenarbeit zwischen Outsourcing-Partnern und internen Regulierungsteams weiter und gewährleistet Transparenz, Konsistenz und einfachen Zugriff auf aktualisierte Einreichungsmaterialien.
- Dieser Trend zu intelligenten regulatorischen Lösungen verändert die Erwartungen innerhalb der Branche. Die Kunden fordern schnellere Bearbeitungszeiten, verbesserte Datengenauigkeit und skalierbare Compliance-Unterstützung.
- Mit der zunehmenden Komplexität globaler Vorschriften steigt die Nachfrage nach intelligenten, automatisierten und technologiegetriebenen regulatorischen Outsourcing-Dienstleistungen sowohl in etablierten als auch in aufstrebenden Märkten für Medizinprodukte. Unternehmen betrachten diese Partnerschaften zunehmend als entscheidend für ihre globalen Expansionsstrategien.
Marktdynamik für das Outsourcing regulatorischer Angelegenheiten im Bereich Medizinprodukte
Treiber
„Zunehmende Komplexität globaler Regulierungsrahmen und Produktinnovationen“
- Die zunehmende Komplexität internationaler Regulierungsstandards und die kontinuierliche Innovation in der Medizinproduktetechnologie sind die Haupttreiber, die die Nachfrage nach Outsourcing-Dienstleistungen im Bereich der Regulierungsfragen beschleunigen.
- So erweiterte Parexel im Januar 2024 seine globalen regulatorischen Beratungsdienste, um Unternehmen bei der Bewältigung der neuen EU-MDR- und IVDR-Rahmenbedingungen zu unterstützen, und verdeutlichte damit den zunehmenden Bedarf an fachkundiger Beratung in komplexen Compliance-Umgebungen.
- Hersteller medizinischer Geräte stehen unter dem Druck, mit den sich entwickelnden globalen Anforderungen Schritt zu halten und gleichzeitig einen zeitnahen Marktzugang sicherzustellen. Externe Regulierungspartner sind daher für die Aufrechterhaltung von Geschwindigkeit und Genauigkeit von entscheidender Bedeutung.
- Der Aufstieg digitaler Gesundheitslösungen, KI-fähiger Geräte und Kombinationsprodukte hat die regulatorische Kontrolle weiter verschärft und Unternehmen dazu gezwungen, sich bei strategischen Einreichungen, der laufenden Einhaltung von Vorschriften und der Marktüberwachung auf externe Experten zu verlassen.
- Durch das Outsourcing dieser Aufgaben können sich Hersteller auf Innovation und Kommerzialisierung konzentrieren und gleichzeitig die Belastung interner Ressourcen reduzieren, Genehmigungsverfahren beschleunigen und effektiver in neue Märkte expandieren.
Einschränkung/Herausforderung
„Datenschutzbedenken und Fachkräftemangel“
- Wachsende Bedenken hinsichtlich Datenschutz und Cybersicherheit stellen eine große Herausforderung für den globalen Outsourcing-Markt für Medizinprodukte dar. Da sensible Patientendaten und proprietäre Produktinformationen grenzüberschreitend übertragen werden, müssen Unternehmen die vollständige Einhaltung von Datenschutzbestimmungen wie DSGVO und HIPAA sicherstellen.
- Beispielsweise kann jeder Verstoß oder jede falsche Handhabung der behördlichen Dokumentation durch Drittanbieter für Hersteller erhebliche rechtliche, finanzielle und Reputationsrisiken bedeuten, was möglicherweise zu Verzögerungen bei der Produktzulassung oder zu behördlichen Sanktionen führen kann.
- Darüber hinaus herrscht auf dem Markt ein anhaltender Mangel an qualifizierten Regulierungsexperten mit fundierten Kenntnissen der regionsspezifischen Gerätevorschriften und Qualitätsstandards, was zu Engpässen und Herausforderungen bei der Qualitätskontrolle für Outsourcing-Unternehmen führt.
- Während einige Unternehmen in die Personalentwicklung und die digitale Compliance-Infrastruktur investieren, ist die Überbrückung dieser Talent- und Sicherheitslücke entscheidend, um das Vertrauen der Kunden zu gewinnen und zu behalten.
- Die Lösung dieser Probleme durch verbesserte Datenverwaltungsprotokolle, kontinuierliche Weiterbildung und strategische regionale Partnerschaften wird der Schlüssel zur Gewährleistung eines sicheren, effizienten und konformen Outsourcings von regulatorischen Angelegenheiten auf lange Sicht sein.
Outsourcing des Marktes für regulatorische Angelegenheiten im Bereich Medizinprodukte
Der Markt ist nach Diensten, Produkten, Gerätetypen, Kategorien, Anwendungen und Endbenutzern segmentiert.
- Nach Dienstleistungen
Der Markt für das Outsourcing von Zulassungsangelegenheiten für Medizinprodukte ist nach Dienstleistungen segmentiert in Zulassungsdienstleistungen, Qualitätsberatung, Verfassen und Veröffentlichen von Zulassungsanträgen, Zulassungsanträge für klinische Studien, Produktregistrierungen, Rechtsvertretung und medizinisches Schreiben. Das Segment „Verfassen und Veröffentlichen von Zulassungsanträgen“ dominierte den Markt mit dem größten Umsatzanteil von 42 % im Jahr 2024, was auf seine wesentliche Rolle bei der Erstellung und Einreichung konformer, detaillierter Zulassungsdokumente bei globalen Behörden zurückzuführen ist. Das Verfassen von Zulassungsanträgen gewährleistet Klarheit, Genauigkeit und Konsistenz bei der Einreichung, was entscheidend für die rechtzeitige Produktzulassung und die Einhaltung der Konformität während des gesamten Produktlebenszyklus ist.
Das Segment der klinischen Studienanwendungen wird voraussichtlich zwischen 2025 und 2032 das höchste Wachstum verzeichnen, angetrieben durch die steigende Zahl innovativer Geräte in der klinischen Entwicklung. Angesichts strengerer regulatorischer Rahmenbedingungen trägt das Outsourcing klinischer Anwendungsaufgaben an erfahrene Regulierungsdienstleister dazu bei, Genauigkeit und Compliance zu gewährleisten und Verzögerungen im Genehmigungsprozess zu reduzieren.
- Nach Produkt
Der Markt für das Outsourcing von Zulassungsangelegenheiten für Medizinprodukte ist produktbezogen in Fertigprodukte, Elektronik und Rohstoffe segmentiert. Das Segment Fertigprodukte hatte 2024 den größten Marktanteil, was auf die hohe Anzahl fertiger Medizinprodukte zurückzuführen ist, die vor der Vermarktung umfangreiche Dokumentation, Tests und behördliche Genehmigungen erfordern. Diese Produkte erfordern oft umfassende globale Zulassungsstrategien und Unterstützung bei der Marktüberwachung nach der Markteinführung, was die Nachfrage nach Outsourcing ankurbelt.
Der Elektroniksektor wird voraussichtlich von 2025 bis 2032 die höchste durchschnittliche jährliche Wachstumsrate verzeichnen, was auf die zunehmende Integration digitaler Technologien, KI und Softwarekomponenten in Medizinprodukte zurückzuführen ist. Die Einhaltung gesetzlicher Vorschriften für elektronische Komponenten, insbesondere in vernetzten und tragbaren Geräten, erfordert spezielles Fachwissen in den Bereichen Cybersicherheit und technische Dateivorbereitung, was die Nachfrage nach Outsourcing erhöht.
- Nach Gerätetyp
Der Markt für das Outsourcing von Zulassungsangelegenheiten für Medizinprodukte ist nach Gerätetyp in Klasse I, Klasse II und Klasse III unterteilt. Das Klasse-II-Segment dominierte den Markt mit dem größten Umsatzanteil im Jahr 2024 aufgrund der großen Anzahl mittelkomplexer Medizinprodukte, die unter diese Klassifizierung fallen. Diese Geräte erfordern umfangreiche regulatorische Dokumentation und die Einhaltung von Qualitätsstandards, weshalb Hersteller externe Unterstützung für effiziente und zeitnahe Zulassungen suchen.
Das Segment der Klasse III wird voraussichtlich von 2025 bis 2032 das höchste Wachstum verzeichnen, da diese Hochrisikogeräte umfangreiche Zulassungen vor der Markteinführung, strenge klinische Studien und Langzeitüberwachungsdaten erfordern. Aufgrund der Komplexität und des Risikos dieser Geräte ist Outsourcing eine strategische Entscheidung für Hersteller, die die globalen regulatorischen Erwartungen erfüllen möchten.
- Nach Kategorie
Der Markt für das Outsourcing von regulatorischen Angelegenheiten für Medizinprodukte ist nach Kategorien in Biologika, Arzneimittel und Medizinprodukte unterteilt. Das Segment Medizinprodukte dominierte den Markt mit dem größten Umsatzanteil im Jahr 2024, angetrieben durch die zunehmende Innovation und das weltweite Volumen neuer Geräteentwicklungen. Die ständige Weiterentwicklung der regulatorischen Anforderungen für Medizinprodukte ermutigt Hersteller, ihre Arbeit an spezialisierte Dienstleister auszulagern, um die Compliance zu verbessern und die Effizienz zu steigern.
Das Segment der Biologika dürfte zwischen 2025 und 2032 das schnellste Wachstum verzeichnen, angetrieben durch die zunehmende Verbreitung von Kombinationsprodukten und personalisierten Therapien. Diese Produkte erfordern multidisziplinäre regulatorische Ansätze, wodurch Outsourcing für die Bewältigung komplexer Zulassungsverfahren und die Gewährleistung einer koordinierten Compliance unerlässlich wird.
- Nach Anwendung
Der Markt für das Outsourcing von regulatorischen Angelegenheiten für Medizinprodukte ist nach Anwendungsgebieten in Kardiologie, diagnostische Bildgebung, Orthopädie, IVD, Augenheilkunde, allgemeine und plastische Chirurgie, Arzneimittelverabreichung, Zahnmedizin, Endoskopie, Diabetesversorgung und weitere Bereiche unterteilt. Das IVD-Segment hatte 2024 den größten Marktanteil, unterstützt durch den Anstieg diagnostischer Tests und die Umsetzung strenger regulatorischer Rahmenbedingungen wie der EU-IVDR. Regulatorisches Outsourcing ist entscheidend, um eine genaue Klassifizierung, technische Dokumentation und Leistungsbewertungsdaten für diese Produkte zu gewährleisten.
Das Segment Diabetesversorgung wird im Prognosezeitraum voraussichtlich die höchste jährliche Wachstumsrate verzeichnen, angetrieben durch die Verbreitung innovativer Blutzuckermessgeräte und digitaler Gesundheitslösungen. Diese Technologien erfordern spezielle Regulierungsstrategien und eine kontinuierliche Compliance-Überwachung, was die Abhängigkeit von Outsourcing-Partnern erhöht.
- Nach Endbenutzer
Der Markt für das Outsourcing von regulatorischen Angelegenheiten im Bereich Medizinprodukte ist nach Endnutzern segmentiert: kleine Medizinproduktehersteller, mittlere Medizinproduktehersteller, Pharma- und Biotechnologieunternehmen, Medizinproduktehersteller und große Medizinproduktehersteller. Das Segment der großen Medizinproduktehersteller dominierte den Markt mit dem größten Umsatzanteil im Jahr 2024, da diese Unternehmen vielfältige Portfolios in mehreren globalen Märkten verwalten und auf regulatorisches Outsourcing angewiesen sind, um ihre betriebliche Effizienz und regulatorische Konsistenz zu gewährleisten.
Das Segment der kleinen Medizintechnikunternehmen wird voraussichtlich von 2025 bis 2032 das stärkste Wachstum verzeichnen. Grund dafür sind begrenzte interne Regulierungsressourcen und der Bedarf an Expertenberatung zur Erfüllung komplexer Compliance-Standards. Diese Unternehmen lagern häufig umfassende Regulierungsaufgaben aus, um den Markteintritt zu beschleunigen und den operativen Aufwand zu reduzieren.
Regionale Analyse des Outsourcing-Marktes für regulatorische Angelegenheiten im Medizinproduktebereich
- Nordamerika dominierte den Outsourcing-Markt für regulatorische Angelegenheiten im Bereich Medizinprodukte mit dem größten Umsatzanteil von 39,2 % im Jahr 2024, unterstützt durch die ausgereifte Medizinprodukteindustrie der Region, einen starken regulatorischen Rahmen und eine hohe Outsourcing-Durchdringung bei US-amerikanischen Herstellern, die schnellere FDA-Zulassungen anstreben.
- Unternehmen in der Region legen Wert auf zeitnahe Produktzulassungen und eine strategische globale Expansion. Dies führt dazu, dass sie sich bei der Verwaltung komplexer Einreichungen und sich entwickelnder Vorschriften wie der QMSR der US-amerikanischen FDA und der Übergangsunterstützung zur EU-MDR auf Outsourcing-Partner verlassen.
- Diese starke Marktposition wird durch ein gut etabliertes Ökosystem von Regulierungsberatungsfirmen, robuste F&E-Investitionen und eine hohe Konzentration von Herstellern medizinischer Geräte weiter unterstützt, was Nordamerika zu einem wichtigen Zentrum für ausgelagerte Regulierungsdienstleistungen in allen Phasen der Produktentwicklung und -vermarktung macht.
Einblicke in den US-Markt für das Outsourcing regulatorischer Angelegenheiten im Bereich Medizinprodukte
Der US-Markt für das Outsourcing von Zulassungsangelegenheiten für Medizinprodukte erzielte 2024 mit 84 % den größten Umsatzanteil in Nordamerika. Dies ist auf die Komplexität der FDA-Zulassungsprozesse und die zunehmende Abhängigkeit von externer Expertise zur Beschleunigung von Zulassungen zurückzuführen. Angesichts des hohen Innovationsvolumens bei Medizinprodukten und des wachsenden Drucks, den Markteintritt zu beschleunigen, lagern US-Hersteller Aufgaben wie die Vorbereitung von Einreichungen, die Regulierungsstrategie und die Einhaltung der Vorschriften nach der Markteinführung aus. Darüber hinaus verstärkt die kontinuierliche Weiterentwicklung der FDA-Vorschriften, einschließlich der Aktualisierung des QMSR, den Bedarf an spezialisierter regulatorischer Beratung für Unternehmen aller Größen.
Einblicke in den europäischen Markt für das Outsourcing regulatorischer Angelegenheiten im Bereich Medizinprodukte
Der europäische Markt für das Outsourcing von regulatorischen Angelegenheiten im Bereich Medizinprodukte wird im Prognosezeitraum voraussichtlich mit einer deutlichen jährlichen Wachstumsrate wachsen, vor allem aufgrund der Umsetzung der EU-MDR- und IVDR-Rahmenwerke. Diese Vorschriften haben die Anforderungen an Dokumentation, klinische Nachweise und Überwachung deutlich erhöht und damit die Nachfrage nach Outsourcing-Partnern mit EU-spezifischer regulatorischer Expertise erhöht. Die starke Präsenz kleiner und mittelständischer Gerätehersteller in der Region trägt zusätzlich zur Akzeptanz des Outsourcings bei, insbesondere in wichtigen Ländern wie Deutschland, Frankreich und den Niederlanden, wo eine rechtzeitige CE-Kennzeichnung für den Geschäftserfolg entscheidend ist.
Einblicke in den britischen Outsourcing-Markt für Medizinprodukteregulierung
Der britische Markt für das Outsourcing von regulatorischen Angelegenheiten im Bereich Medizinprodukte wird im Prognosezeitraum voraussichtlich mit einer bemerkenswerten jährlichen Wachstumsrate wachsen. Dies wird durch regulatorische Veränderungen nach dem Brexit und die zunehmende Notwendigkeit, sowohl die UKCA- als auch die internationalen Compliance-Prozesse zu meistern, unterstützt. Hersteller in Großbritannien lagern zunehmend regulatorische Aufgaben aus, um die Einhaltung der MHRA-Anforderungen sicherzustellen und gleichzeitig den globalen Marktzugang zu sichern. Die Präsenz hochspezialisierter Regulierungsberatungen und ein starkes Medtech-Ökosystem machen Großbritannien zu einem wichtigen Wachstumstreiber der Region.
Markteinblicke für das Outsourcing regulatorischer Angelegenheiten im Medizinproduktebereich in Deutschland
Der deutsche Markt für das Outsourcing von regulatorischen Angelegenheiten im Bereich Medizinprodukte wird im Prognosezeitraum voraussichtlich mit einer deutlichen jährlichen Wachstumsrate wachsen. Dies ist auf die Position des Unternehmens als führender Standort für die Herstellung von Medizinprodukten und das strenge regulatorische Umfeld zurückzuführen. Da sich Unternehmen auf die Erfüllung der komplexen EU-MDR-Konformitätsanforderungen konzentrieren, wenden sich deutsche Hersteller an Outsourcing-Unternehmen, um Unterstützung bei Dokumentation, Gap-Analyse und klinischen Bewertungen zu erhalten. Der Schwerpunkt auf Präzision, Qualität und Nachhaltigkeit in der deutschen Medizintechnik fördert zudem die Zusammenarbeit mit erfahrenen Regulierungspartnern, um die globale Wettbewerbsfähigkeit zu sichern.
Markteinblicke für das Outsourcing regulatorischer Angelegenheiten im Bereich Medizinprodukte im asiatisch-pazifischen Raum
Der Markt für das Outsourcing von regulatorischen Angelegenheiten im Bereich Medizinprodukte im asiatisch-pazifischen Raum dürfte im Prognosezeitraum von 2025 bis 2032 mit einer durchschnittlichen jährlichen Wachstumsrate von 25 % wachsen. Dies ist auf die wachsende Produktion von Medizinprodukten, steigende internationale Exporte und immer anspruchsvollere regulatorische Rahmenbedingungen in Ländern wie China, Japan und Indien zurückzuführen. Regionale Regierungen verstärken die Aufsicht und orientieren sich an globalen Standards, was lokale und globale Unternehmen dazu veranlasst, Compliance-Aufgaben auszulagern. Darüber hinaus ziehen die Kosteneffizienz und die schnelle Skalierbarkeit von Outsourcing-Lösungen im asiatisch-pazifischen Raum multinationale Gerätehersteller an, die eine regionale Zulassung und Distribution anstreben.
Einblicke in den japanischen Markt für das Outsourcing regulatorischer Angelegenheiten im Bereich Medizinprodukte
Der japanische Markt für das Outsourcing von Zulassungsangelegenheiten für Medizinprodukte gewinnt aufgrund der komplexen PMDA-Zulassungsverfahren und der hohen technologischen Innovationsrate im Medizintechniksektor des Landes an Dynamik. Japanische Hersteller nutzen zunehmend regulatorisches Outsourcing für die Produktklassifizierung, die Unterstützung klinischer Studien und die zweisprachige Dokumentation, um zeitnahe lokale und internationale Zulassungen zu gewährleisten. Die Nachfrage nach regulatorischer Expertise in Bereichen wie KI-fähigen Geräten und Kombinationsprodukten treibt das Marktwachstum in Japan weiter voran.
Einblicke in den indischen Markt für das Outsourcing regulatorischer Angelegenheiten im Bereich Medizinprodukte
Der indische Markt für das Outsourcing von regulatorischen Angelegenheiten im Bereich Medizinprodukte erzielte 2024 den größten Umsatzanteil im asiatisch-pazifischen Raum. Dies ist auf die wachsende Produktionsbasis für Medizinprodukte, die zunehmende Einhaltung der CDSCO-Richtlinien und die zunehmende Präsenz inländischer Regulierungsberatungen zurückzuführen. Die Förderung von „Make in India“ und intelligenter Gesundheitsinfrastruktur hat zu einem Anstieg der Produktentwicklung geführt, der eine effiziente regulatorische Unterstützung erfordert. Die Verfügbarkeit qualifizierter Fachkräfte und die Kostenvorteile machen Indien zu einem zentralen Knotenpunkt für lokales und internationales regulatorisches Outsourcing.
Marktanteil im Outsourcing von regulatorischen Angelegenheiten für Medizinprodukte
Die Outsourcing-Branche für regulatorische Angelegenheiten im Bereich Medizinprodukte wird hauptsächlich von etablierten Unternehmen geführt, darunter:
- Parexel International (MA) Corporation (USA)
- North American Science Associates, LLC (USA)
- SGS Société Générale de Surveillance SA. (Schweiz)
- Tempo (USA)
- Trilogy Writing & Consulting GmbH (Deutschland)
- Creganna (Irland)
- Intertek Group plc (Großbritannien)
- WuXi AppTec (China)
- Charles River Laboratories (USA)
- Celestica Inc. (Kanada)
- Freyr (USA)
- Cactus Communications (Indien)
- In.Corp Indonesia (Indonesien)
- Eurofins Scientific (Luxemburg)
- Plexus Corp. (USA)
- Sanmina Corporation (USA)
- OMRON Corporation (Japan)
Was sind die jüngsten Entwicklungen auf dem globalen Outsourcing-Markt für regulatorische Angelegenheiten im Bereich Medizinprodukte?
- Im April 2023 erweiterte die Parexel International Corporation ihre globalen regulatorischen Beratungsleistungen, um Entwickler von Medizinprodukten und Kombinationsprodukten bei der Bewältigung der zunehmend komplexen Anforderungen der EU-Medizinprodukteverordnung (EU-MDR) und der In-vitro-Diagnostikaverordnung (IVDR) zu unterstützen. Diese strategische Erweiterung spiegelt Parexels Engagement wider, Kunden durch umfassende regulatorische Expertise und globale Präsenz bei der Beschleunigung des Marktzugangs zu unterstützen, insbesondere angesichts der zunehmenden europäischen Compliance-Landschaft.
- Im März 2023 kündigte ICON plc die Einführung einer neuen Regulatory Intelligence-Plattform an, die Medizinprodukteunternehmen bei der Bewältigung sich entwickelnder globaler Vorschriften und Einreichungsstrategien unterstützen soll. Diese digitale Lösung bietet Echtzeitzugriff auf regulatorische Updates, Compliance-Zeitpläne und Dokumentvorlagen und ermöglicht so optimierte Vorbereitungs- und Genehmigungsprozesse. Die Innovation unterstreicht ICONs Fokus auf die Integration von Technologie in die regulatorische Beratung, um Effizienz und Genauigkeit für seine Kunden zu steigern.
- Im März 2023 eröffnete Freyr Solutions ein neues globales Zentrum für regulatorische Dienstleistungen im asiatisch-pazifischen Raum. Ziel ist es, Medizinprodukteunternehmen bei der umfassenden Einhaltung länderspezifischer Zulassungen zu unterstützen. Das neue Zentrum stärkt Freyrs Kapazitäten, die wachsende Nachfrage aus Schwellenländern zu bewältigen und stärkt seine Position als führender Anbieter von Regulatory Affairs Outsourcing durch skalierbare, regional zugeschnittene Lösungen.
- Im Februar 2023 gab IQVIA eine strategische Partnerschaft mit einem europäischen Hersteller von Medizinprodukten bekannt, um die Erstellung von Zulassungsunterlagen und die Berichterstattung über klinische Bewertungen gemäß der EU-MDR zu unterstützen. Diese Zusammenarbeit unterstreicht IQVIAs Stärke bei der Bereitstellung umfassender, maßgeschneiderter regulatorischer Dienstleistungen und spiegelt einen breiteren Trend wider, dass Hersteller auf Outsourcing setzen, um die Komplexität der MDR zu bewältigen und Risiken zu minimieren.
- Im Januar 2023 führte Medistri SA, ein Schweizer Medizintechnik-Dienstleister, eine integrierte Beratungslösung für Regulatory Affairs und Qualitätsmanagement für kleine und mittelständische Gerätehersteller ein. Dieses Angebot soll Start-ups und jungen Unternehmen den Weg zur Compliance erleichtern und bietet kostengünstige Unterstützung bei der CE-Kennzeichnung, der Erstellung technischer Unterlagen und der Marktüberwachung. Dieser Schritt unterstreicht den wachsenden Fokus der Branche auf skalierbare, zugängliche Outsourcing-Dienstleistungen, die auf Unternehmen mit begrenzten internen Regulierungsressourcen zugeschnitten sind.
SKU-
Erhalten Sie Online-Zugriff auf den Bericht zur weltweit ersten Market Intelligence Cloud
- Interaktives Datenanalyse-Dashboard
- Unternehmensanalyse-Dashboard für Chancen mit hohem Wachstumspotenzial
- Zugriff für Research-Analysten für Anpassungen und Abfragen
- Konkurrenzanalyse mit interaktivem Dashboard
- Aktuelle Nachrichten, Updates und Trendanalyse
- Nutzen Sie die Leistungsfähigkeit der Benchmark-Analyse für eine umfassende Konkurrenzverfolgung
Inhaltsverzeichnis
- INTRODUCTION
- OBJECTIVES OF THE STUDY
- MARKET DEFINITION
- OVERVIEW OF GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET
- LIMITATIONS
- MARKETS COVERED
- MARKET SEGMENTATION
- MARKETS COVERED
- GEOGRAPHICAL SCOPE
- YEARS CONSIDERED FOR THE STUDY
- CURRENCY AND PRICING
- DBMR TRIPOD DATA VALIDATION MODEL
- MULTIVARIATE MODELLING
- SERVICES LIFELINE CURVE
- PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
- DBMR MARKET POSITION GRID
- VENDOR SHARE ANALYSIS
- MARKET APPLICATION COVERAGE GRID
- SECONDARY SOURCES
- ASSUMPTIONS
- EXECUTIVE SUMMARY
- PREMIUM INSIGHT
- MARKET OVERVIEW
- DRIVERS
- INCREASING GEOGRAPHICAL EXPANSION ACTIVITIES BY THE MEDICAL DEVICE COMPANIES
- INCREASING ADOPTION OF OUTSOURCING MODELS FOR REGULATORY SERVICES
- INCREASE IN R&D ACTIVITIES BY THE MEDICAL DEVICE COMPANIES
- RISING NUMBER OF CLINICAL TRIALS
- INCREASING NUMBER OF PATENT EXPIRATIONS
- RESTRAINTS
- FLUCTUATION IN PRICES
- HIGH COMPETITION AMONG KEY MARKET PLAYERS
- OPPORTUNITIES
- INCREASING SPENDING ON HEALTHCARE
- END-TO-END REGULATORY EXPERTISE
- AWARDS AND RECOGNITION
- CHALLENGES
- LACK OF ACCESSIBILITY
- PANDEMIC OUTBREAK OF COVID-19
- IMPACT OF COVID-19 ON THE GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET
- IMPACT ON PRICE
- IMPACT ON DEMAND
- IMPACT ON SUPPLY CHAIN
- STRATEGIC DECISIONS OF GOVERNMENT AND MANUFACTURERS
- CONCLUSION
- GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES
- OVERVIEW
- REGULATORY AFFAIRS SERVICES
- CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS
- CERTIFICATION SERVICES
- ISO 13485 AUDITS
- NOTIFIED BODY (EU)
- GLOBAL MARKET ACCESS
- CB SCHEME
- NRL & SCC (US & CAN)
- MDSAP
- REGULATORY WRITING AND PUBLISHING
- LEGAL REPRESENTATION
- OTHERS
- QUALITY CONSULTING
- MEDICAL WRITING
- CLINICAL WRITING
- REGULATORY WRITING
- SCIENTIFIC WRITING
- OTHERS
- GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY PRODUCT
- OVERVIEW
- FINISHED GOODS
- ELECTRONICS
- RAW MATERIALS
- GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY DEVICE TYPE
- OVERVIEW
- CLASS I
- CLASS II
- CLASS III
- GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY APPLICATION
- OVERVIEW
- CARDIOLOGY
- CLASS III
- CLASS I
- CLASS II
- GENERAL AND PLASTIC SURGERY
- CLASS I
- CLASS II
- CLASS III
- IVD
- CLASS I
- CLASS III
- CLASS II
- ORTHOPAEDIC
- CLASS III
- CLASS I
- CLASS II
- DIAGNOSTIC IMAGING
- CLASS II
- CLASS I
- CLASS III
- DENTAL
- CLASS I
- CLASS III
- CLASS II
- OPHTHALMIC
- CLASS I
- CLASS II
- CLASS III
- ENDOSCOPY
- CLASS II
- CLASS I
- CLASS III
- DIABETES CARE
- CLASS I
- CLASS II
- CLASS III
- DRUG DELIVERY
- CLASS II
- CLASS III
- CLASS I
- OTHERS
- CLASS I
- CLASS II
- CLASS III
- GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY END USER
- OVERVIEW
- MEDIUM MEDICAL DEVICE COMPANY
- SMALL MEDICAL DEVICE COMPANY
- LARGE MEDICAL DEVICE COMPANY
- GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY GEOGRAPHY
- OVERVIEW
- NORTH AMERICA
- U.S.
- CANADA
- MEXICO
- EUROPE
- GERMANY
- FRANCE
- U.K.
- ITALY
- SPAIN
- RUSSIA
- TURKEY
- IRELAND
- BELGIUM
- NETHERLANDS
- SWITZERLAND
- REST OF EUROPE
- ASIA-PACIFIC
- JAPAN
- CHINA
- SOUTH KOREA
- INDIA
- AUSTRALIA
- SINGAPORE
- THAILAND
- MALAYSIA
- INDONESIA
- PHILIPPINES
- REST OF ASIA-PACIFIC
- SOUTH AMERICA
- BRAZIL
- ARGENTINA
- REST OF SOUTH AMERICA
- MIDDLE EAST AND AFRICA
- SOUTH AFRICA
- SAUDI ARABIA
- U.A.E.
- EGYPT
- ISRAEL
- REST OF MIDDLE EAST AND AFRICA
- GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET: COMPANY LANDSCAPE
- COMPANY SHARE ANALYSIS: GLOBAL
- COMPANY SHARE ANALYSIS: NORTH AMERICA
- COMPANY SHARE ANALYSIS: EUROPE
- COMPANY SHARE ANALYSIS: ASIA-PACIFIC
- SWOT ANALYSIS
- COMPANY PROFILE
- PLEXUS CORP.
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- COMPANY SHARE ANLYSIS
- SERVICE PORTFOLIO
- RECENT DEVELOPMENTS
- CELESTICA INC.
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- COMPANY SHARE ANLYSIS
- SERVICE PORTFOLIO
- RECENT DEVELOPMENTS
- FLEX LTD.
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- COMPANY SHARE ANALYSIS
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENTS
- TE CONNECTIVITY
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- COMPANY SHARE ANLYSIS
- INDUSTRY & SOLUTION PORTFOLIO
- RECENT DEVELOPMENTS
- TECOMET, INC.
- COMPANY SNAPSHOT
- COMPANY SHARE ANLYSIS
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- PAREXEL INTERNATIONAL CORPORATION
- COMPANY SNAPSHOT
- SOLUTION PORTFOLIO
- RECENT DEVELOPMENTS
- SANMINA CORPORATION
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- INDUSTRY PORTFOLIO
- RECENT DEVELOPMENTS
- EUROFINS SCIENTIFIC
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- SERVICE PORTFOLIO
- RECENT DEVELOPMENT
- AMERICAN PRECLINICAL SERVICES, LLC
- COMPANY SNAPSHOT
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- CACTUS COMMUNICATIONS
- COMPANY SNAPSHOT
- SERVICE PORTFOLIO
- RECENT DEVELOPMENTS
- CEKINDO BUSINESS INTERNATIONAL
- COMPANY SNAPSHOT
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- CHARLES RIVER LABORATORIES
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- PRODUCT & SERVICE PORTFOLIO
- RECENT DEVELOPMENTS
- COVANCE (A SUBSIDIARY OF LABORATORY CORPORATION OF AMERICA HOLDINGS)
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- SERVICE PORTFOLIO
- RECENT DEVELOPMENTS
- CREGANNA (A SUBSIDIARY OF TE CONNECTIVITY)
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- PRODUCT & SERVICE PORTFOLIO
- RECENT DEVELOPMENTS
- EAST WEST MANUFACTURING
- COMPANY SNAPSHOT
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- FREYR
- COMPANY SNAPSHOT
- SERVICE PORTFOLIO
- RECENT DEVELOPMENTS
- HERAEUS HOLDING
- COMPANY SNAPSHOT
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- INTEGER HOLDINGS CORPORATION
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENTS
- INTERTEK GROUP PLC
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- IQVIA
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENTS
- NORTH AMERICAN SCIENCE ASSOCIATES, INC.
- COMPANY SNAPSHOT
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- NORTECH SYSTEMS, INC.
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- OMICS INTERNATIONAL
- COMPANY SNAPSHOT
- SERVICE PORTFOLIO
- RECENT DEVELOPMENTS
- OMRON CORPORATION
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- PACE ANALYTICAL SERVICES, LLC
- COMPANY SNAPSHOT
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENTS
- JABIL INC.
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- SGS SA
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- STERIGENICS U.S., LLC – A SOTERA HEALTH COMPANY
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- SERVICE PORTFOLIO
- RECENT DEVELOPMENTS
- TRILOGY WRITING & CONSULTING GMBH
- COMPANY SNAPSHOT
- PRODUCT PORTFOLIO
- RECENT DEVELOPMENT
- TÜV SÜD
- COMPANY SNAPSHOT
- SERVICE PORTFOLIO
- RECENT DEVELOPMENT
- WUXI APPTEC
- COMPANY SNAPSHOT
- REVENUE ANALYSIS
- SERVICE PORTFOLIO
- RECENT DEVELOPMENTS
- QUESTIONNAIRE
- RELATED REPORTS
Tabellenverzeichnis
TABLE 1 Global Medical device regulatory affairs outsourcing market, By services, 2019-2028 (USD million)
TABLE 2 Global Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD Million)
TABLE 3 Global Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 4 Global Clinical Trials Applications and Product Registrations IN Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 5 Global Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 6 Global Quality Consulting service in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD Million)
TABLE 7 Global Medical Writing service in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD Million)
TABLE 8 Global Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 9 Global Medical device regulatory affairs outsourcing market, By product, 2019-2028 (USD million)
TABLE 10 Global Finished Goods products in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 11 Global Electronics products in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 12 Global Raw Materials products in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 13 Global Medical device regulatory affairs outsourcing market, By device type, 2019-2028 (USD million)
TABLE 14 Global class i Device in Medical device regulatory affairs outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 15 Global Class II Device in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 16 Global Class III Device in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 17 Global Medical device regulatory affairs outsourcing market, By application, 2019-2028 (USD million)
TABLE 18 Global Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 19 Global Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By device type, 2019-2028 (USD million)
TABLE 20 Global General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 21 Global General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By device type, 2019-2028 (USD million)
TABLE 22 Global IVD in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 23 Global IVD in Medical Device Regulatory Affairs Outsourcing Market, By device type, 2019-2028 (USD million)
TABLE 24 Global Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 25 Global Orthopedic in Medical Device Regulatory Affairs Outsourcing Market, By device type, 2019-2028 (USD million)
TABLE 26 Global Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 27 Global Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By device type, 2019-2028 (USD million)
TABLE 28 Global Dental in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 29 Global dental in Medical device regulatory affairs outsourcing market, By device type, 2019-2028 (USD million)
TABLE 30 Global Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 31 Global Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By device type, 2019-2028 (USD million)
TABLE 32 Global Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 33 Global Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By device type, 2019-2028 (USD million)
TABLE 34 Global Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 35 Global Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By device type, 2019-2028 (USD million)
TABLE 36 Global Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 37 Global Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By device type, 2019-2028 (USD million)
TABLE 38 Global Others in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 39 Global Others in Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD million)
TABLE 40 Global Medical device regulatory affairs outsourcing market, By end user, 2019-2028 (USD million)
TABLE 41 Global Medium Medical Device Company in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 42 Global Small Medical Device Company in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 43 Global Large Medical Device Company in Medical Device Regulatory Affairs Outsourcing Market, By Region, 2019-2028 (USD million)
TABLE 44 GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY REGION, 2019-2028 (USD MILLION)
TABLE 45 North America medical device regulatory affairs outsourcing Market, By COUNTRY, 2019-2028 (USD million)
TABLE 46 North America Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 47 North America Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 48 North America CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 49 North America CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 50 North America Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 51 North America Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 52 North America Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 53 North America Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 54 North America Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By device type, 2019-2028 (USD Million)
TABLE 55 North America General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 56 North America IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 57 North America Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 58 North America Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 59 North America Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 60 North America Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 61 North America Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 62 North America Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 63 North America Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 64 North America Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 65 North America Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 66 U.S. Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 67 U.S. Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 68 U.S. CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 69 U.S. CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 70 U.S. Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 71 U.S. Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 72 U.S. Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 73 U.S. Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 74 U.S. Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 75 U.S. General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 76 U.S. IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 77 U.S. Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 78 U.S. Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 79 U.S. Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 80 U.S. Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 81 U.S. Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 82 U.S. Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 83 U.S. Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 84 U.S. Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 85 U.S. Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 86 CANADA Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 87 CANADA Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 88 CANADA CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 89 CANADA CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 90 CANADA Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 91 CANADA Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 92 CANADA Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 93 CANADA Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 94 CANADA Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 95 CANADA General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 96 CANADA IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 97 CANADA Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 98 CANADA Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 99 CANADA Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 100 CANADA Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 101 CANADA Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 102 CANADA Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 103 CANADA Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 104 CANADA Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 105 CANADA Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 106 MEXICO Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 107 MEXICO Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 108 MEXICO CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 109 MEXICO CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 110 MEXICO Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 111 MEXICO Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 112 MEXICO Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 113 MEXICO Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 114 MEXICO Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 115 MEXICO General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 116 MEXICO IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 117 MEXICO Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 118 MEXICO Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 119 MEXICO Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 120 MEXICO Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 121 MEXICO Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 122 MEXICO Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 123 MEXICO Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 124 MEXICO Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 125 MEXICO Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 126 Europe medical device regulatory affairs outsourcing Market, By COUNTRY, 2021-2028 (USD million)
TABLE 127 Europe Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 128 Europe Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 129 Europe CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 130 Europe CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 131 Europe Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 132 Europe Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 133 Europe Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 134 Europe Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 135 Europe Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 136 Europe General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 137 Europe IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 138 Europe Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 139 Europe Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 140 Europe Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 141 Europe Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 142 Europe Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 143 Europe Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 144 Europe Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 145 Europe Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 146 Europe Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 147 Germany Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 148 Germany Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 149 Germany CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 150 Germany CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 151 Germany Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 152 Germany Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 153 Germany Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 154 Germany Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 155 Germany Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 156 Germany General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 157 Germany IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 158 Germany Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 159 Germany Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 160 Germany Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 161 Germany Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 162 Germany Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 163 Germany Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 164 Germany Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 165 Germany Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 166 Germany Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 167 France Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 168 France Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 169 France CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 170 France CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 171 France Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 172 France Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 173 France Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 174 France Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 175 France Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 176 France General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 177 France IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 178 France Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 179 France Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 180 France Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 181 France Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 182 France Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 183 France Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 184 France Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 185 France Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 186 France Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 187 U.K. Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 188 U.K. Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 189 U.K. CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 190 U.K. CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 191 U.K. Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 192 U.K. Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 193 U.K. Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 194 U.K. Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 195 U.K. Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 196 U.K. General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 197 U.K. IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 198 U.K. Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 199 U.K. Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 200 U.K. Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 201 U.K. Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 202 U.K. Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 203 U.K. Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 204 U.K. Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 205 U.K. Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 206 U.K. Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 207 Italy Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 208 Italy Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 209 Italy CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 210 Italy CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 211 Italy Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 212 Italy Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 213 Italy Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 214 Italy Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 215 Italy Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 216 Italy General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 217 Italy IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 218 Italy Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 219 Italy Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 220 Italy Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 221 Italy Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 222 Italy Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 223 Italy Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 224 Italy Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 225 Italy Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 226 Italy Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 227 Spain Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 228 Spain Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 229 Spain CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 230 Spain CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 231 Spain Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 232 Spain Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 233 Spain Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 234 Spain Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 235 Spain Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 236 Spain General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 237 Spain IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 238 Spain Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 239 Spain Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 240 Spain Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 241 Spain Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 242 Spain Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 243 Spain Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 244 Spain Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 245 Spain Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 246 Spain Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 247 Russia Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 248 Russia Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 249 Russia CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 250 Russia CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 251 Russia Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 252 Russia Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 253 Russia Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 254 Russia Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 255 Russia Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 256 Russia General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 257 Russia IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 258 Russia Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 259 Russia Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 260 Russia Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 261 Russia Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 262 Russia Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 263 Russia Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 264 Russia Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 265 Russia Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 266 Russia Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 267 Turkey Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 268 Turkey Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 269 Turkey CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 270 Turkey CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 271 Turkey Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 272 Turkey Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 273 Turkey Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 274 Turkey Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 275 Turkey Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 276 Turkey General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 277 Turkey IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 278 Turkey Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 279 Turkey Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 280 Turkey Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 281 Turkey Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 282 Turkey Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 283 Turkey Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 284 Turkey Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 285 Turkey Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 286 Turkey Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 287 Ireland Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 288 Ireland Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 289 Ireland CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 290 Ireland CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 291 Ireland Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 292 Ireland Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 293 Ireland Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 294 Ireland Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 295 Ireland Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 296 Ireland General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 297 Ireland IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 298 Ireland Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 299 Ireland Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 300 Ireland Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 301 Ireland Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 302 Ireland Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 303 Ireland Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 304 Ireland Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 305 Ireland Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 306 Ireland Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 307 Belgium Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 308 Belgium Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 309 Belgium CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 310 Belgium CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 311 Belgium Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 312 Belgium Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 313 Belgium Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 314 Belgium Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 315 Belgium Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 316 Belgium General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 317 Belgium IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 318 Belgium Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 319 Belgium Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 320 Belgium Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 321 Belgium Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 322 Belgium Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 323 Belgium Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 324 Belgium Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 325 Belgium Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 326 Belgium Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 327 Netherlands Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 328 Netherlands Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 329 Netherlands CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 330 Netherlands CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 331 Netherlands Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 332 Netherlands Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 333 Netherlands Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 334 Netherlands Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 335 Netherlands Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 336 Netherlands General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 337 Netherlands IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 338 Netherlands Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 339 Netherlands Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 340 Netherlands Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 341 Netherlands Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 342 Netherlands Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 343 Netherlands Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 344 Netherlands Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 345 Netherlands Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 346 Netherlands Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 347 Switzerland Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 348 Switzerland Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 349 Switzerland CLINICAL TRIALS APPLICATIONS AND PRODUCT REGISTRATIONS IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 350 Switzerland CERTIFICATION SERVICES IN MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET, BY SERVICES, 2019-2028 (USD MILLION)
TABLE 351 Switzerland Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 352 Switzerland Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 353 Switzerland Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 354 Switzerland Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 355 Switzerland Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 356 Switzerland General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 357 Switzerland IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 358 Switzerland Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 359 Switzerland Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 360 Switzerland Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 361 Switzerland Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 362 Switzerland Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 363 Switzerland Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 364 Switzerland Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 365 Switzerland Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 366 Switzerland Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 367 REST OF EUROPE Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 368 Asia-Pacific medical device regulatory affairs outsourcing Market, By COUNTRY, 2019-2028 (USD MILLION)
TABLE 369 Asia-Pacific Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 370 Asia-Pacific Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 371 Asia-Pacific Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 372 Asia-Pacific Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 373 Asia-Pacific Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 374 Asia-Pacific Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 375 Asia-Pacific Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 376 Asia-Pacific Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 377 Asia-Pacific Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By device type, 2019-2028 (USD Million)
TABLE 378 Asia-Pacific General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 379 Asia-Pacific IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 380 Asia-Pacific Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 381 Asia-Pacific Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 382 Asia-Pacific Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 383 Asia-Pacific Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 384 Asia-Pacific Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 385 Asia-Pacific Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 386 Asia-Pacific Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 387 Asia-Pacific Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 388 Asia-Pacific Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 389 Japan Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 390 Japan Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 391 Japan Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 392 Japan Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 393 Japan Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 394 Japan Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 395 Japan Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 396 Japan Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 397 Japan Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 398 Japan General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 399 Japan IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 400 Japan Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 401 Japan Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 402 Japan Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 403 Japan Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 404 Japan Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 405 Japan Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 406 Japan Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 407 Japan Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 408 Japan Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 409 China Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 410 China Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 411 China Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 412 China Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 413 China Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 414 China Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 415 China Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 416 China Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 417 China Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 418 China General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 419 China IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 420 China Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 421 China Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 422 China Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 423 China Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 424 China Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 425 China Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 426 China Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 427 China Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 428 China Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 429 South Korea Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 430 South Korea Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 431 South Korea Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 432 South Korea Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 433 South Korea Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 434 South Korea Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 435 South Korea Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 436 South Korea Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 437 South Korea Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 438 South Korea General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 439 South Korea IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 440 South Korea Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 441 South Korea Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 442 South Korea Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 443 South Korea Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 444 South Korea Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 445 South Korea Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 446 South Korea Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 447 South Korea Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 448 South Korea Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 449 India Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 450 India Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 451 India Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 452 India Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 453 India Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 454 India Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 455 India Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 456 India Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 457 India Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 458 India General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 459 India IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 460 India Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 461 India Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 462 India Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 463 India Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 464 India Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 465 India Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 466 India Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 467 India Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 468 India Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 469 Australia Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 470 Australia Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 471 Australia Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 472 Australia Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 473 Australia Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 474 Australia Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 475 Australia Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 476 Australia Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 477 Australia Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 478 Australia General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 479 Australia IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 480 Australia Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 481 Australia Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 482 Australia Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 483 Australia Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 484 Australia Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 485 Australia Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 486 Australia Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 487 Australia Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 488 Australia Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 489 Singapore Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 490 Singapore Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 491 Singapore Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 492 Singapore Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 493 Singapore Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 494 Singapore Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 495 Singapore Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 496 Singapore Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 497 Singapore Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 498 Singapore General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 499 Singapore IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 500 Singapore Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 501 Singapore Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 502 Singapore Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 503 Singapore Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 504 Singapore Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 505 Singapore Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 506 Singapore Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 507 Singapore Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 508 Singapore Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 509 Thailand Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 510 Thailand Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 511 Thailand Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 512 Thailand Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 513 Thailand Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 514 Thailand Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 515 Thailand Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 516 Thailand Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 517 Thailand Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 518 Thailand General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 519 Thailand IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 520 Thailand Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 521 Thailand Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 522 Thailand Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 523 Thailand Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 524 Thailand Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 525 Thailand Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 526 Thailand Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 527 Thailand Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 528 Thailand Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 529 Malaysia Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 530 Malaysia Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 531 Malaysia Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 532 Malaysia Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 533 Malaysia Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 534 Malaysia Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 535 Malaysia Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 536 Malaysia Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 537 Malaysia Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 538 Malaysia General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 539 Malaysia IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 540 Malaysia Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 541 Malaysia Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 542 Malaysia Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 543 Malaysia Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 544 Malaysia Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 545 Malaysia Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 546 Malaysia Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 547 Malaysia Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 548 Malaysia Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 549 Indonesia Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 550 Indonesia Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 551 Indonesia Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 552 Indonesia Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 553 Indonesia Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 554 Indonesia Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 555 Indonesia Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 556 Indonesia Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 557 Indonesia Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 558 Indonesia General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 559 Indonesia IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 560 Indonesia Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 561 Indonesia Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 562 Indonesia Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 563 Indonesia Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 564 Indonesia Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 565 Indonesia Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 566 Indonesia Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 567 Indonesia Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 568 Indonesia Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 569 Philippines Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 570 Philippines Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 571 Philippines Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 572 Philippines Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 573 Philippines Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 574 Philippines Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 575 Philippines Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 576 Philippines Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 577 Philippines Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 578 Philippines General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 579 Philippines IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 580 Philippines Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 581 Philippines Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 582 Philippines Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 583 Philippines Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 584 Philippines Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 585 Philippines Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 586 Philippines Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 587 Philippines Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 588 Philippines Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 589 Rest of Asia-Pacific Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 590 South America medical device regulatory affairs outsourcing Market, By COUNTRY, 2019-2028 (USD million)
TABLE 591 South America Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 592 South America Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 593 South America Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 594 South America Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 595 South America Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 596 South America Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 597 South America Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 598 South America Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 599 South America Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 600 South America General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 601 South America IVD in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 602 South America Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 603 South America Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 604 South America Dental in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 605 South America Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 606 South America Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 607 South America Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 608 South America Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 609 South America Others in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 610 South America Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 611 Brazil Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 612 Brazil Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 613 Brazil Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 614 Brazil Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 615 Brazil Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 616 Brazil Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 617 Brazil Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 618 Brazil Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 619 Brazil Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 620 Brazil General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 621 Brazil IVD in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 622 Brazil Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 623 Brazil Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 624 Brazil Dental in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 625 Brazil Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 626 Brazil Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 627 Brazil Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 628 Brazil Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 629 Brazil Others in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 630 Brazil Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 631 Argentina Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 632 Argentina Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 633 Argentina Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 634 Argentina Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 635 Argentina Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 636 Argentina Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 637 Argentina Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 638 Argentina Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 639 Argentina Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 640 Argentina General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 641 Argentina IVD in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 642 Argentina Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 643 Argentina Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 644 Argentina Dental in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 645 Argentina Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 646 Argentina Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 647 Argentina Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 648 Argentina Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 649 Argentina Others in Medical Device Regulatory Affairs Outsourcing Market, By DEVICE TYPE, 2019-2028 (USD Million)
TABLE 650 Argentina Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 651 Rest of South America Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 652 Middle East and Africa medical device regulatory affairs outsourcing Market, By COUNTRY, 2019-2028 (USD million)
TABLE 653 Middle East and Africa Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 654 Middle East and Africa Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 655 Middle East and Africa Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 656 Middle East and Africa Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 657 Middle East and Africa Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 658 Middle East and Africa Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 659 Middle East and Africa Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 660 Middle East and Africa Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 661 Middle East and Africa Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 662 Middle East and Africa General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 663 Middle East and Africa IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 664 Middle East and Africa Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 665 Middle East and Africa Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 666 Middle East and Africa Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 667 Middle East and Africa Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 668 Middle East and Africa Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 669 Middle East and Africa Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 670 Middle East and Africa Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 671 Middle East and Africa Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 672 Middle East and Africa Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 673 South Africa Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 674 South Africa Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 675 South Africa Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 676 South Africa Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 677 South Africa Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 678 South Africa Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 679 South Africa Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 680 South Africa Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 681 South Africa Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 682 South Africa General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 683 South Africa IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 684 South Africa Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 685 South Africa Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 686 South Africa Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 687 South Africa Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 688 South Africa Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 689 South Africa Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 690 South Africa Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 691 South Africa Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 692 South Africa Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 693 Saudi Arabia Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 694 Saudi Arabia Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 695 Saudi Arabia Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 696 Saudi Arabia Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 697 Saudi Arabia Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 698 Saudi Arabia Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 699 Saudi Arabia Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 700 Saudi Arabia Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 701 Saudi Arabia Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 702 Saudi Arabia General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 703 Saudi Arabia IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 704 Saudi Arabia Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 705 Saudi Arabia Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 706 audi Arabia Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 707 Saudi Arabia Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 708 Saudi Arabia Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 709 Saudi Arabia Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 710 Saudi Arabia Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 711 Saudi Arabia Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 712 Saudi Arabia Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 713 U.A.E. Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 714 U.A.E. Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 715 U.A.E. Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 716 U.A.E. Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 717 U.A.E. Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 718 U.A.E. Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 719 U.A.E. Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 720 U.A.E. Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 721 U.A.E. Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 722 U.A.E. General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 723 U.A.E. IVD in Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 724 U.A.E. Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 725 U.A.E. Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 726 U.A.E. Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 727 U.A.E. Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 728 U.A.E. Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 729 U.A.E. Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 730 U.A.E. Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 731 U.A.E. Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 732 U.A.E. Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 733 Egypt Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 734 Egypt Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 735 Egypt Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 736 Egypt Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 737 Egypt Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 738 Egypt Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 739 Egypt Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 740 Egypt Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 741 Egypt Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 742 Egypt General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 743 Egypt IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 744 Egypt Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 745 Egypt Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 746 Egypt Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 747 Egypt Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 748 Egypt Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 749 Egypt Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 750 Egypt Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 751 Egypt Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 752 Egypt Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 753 Israel Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 754 Israel Regulatory Affairs Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 755 Israel Clinical Trials Applications and Product Registrations in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 756 Israel Certification Services in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 757 Israel Medical Writing in Medical Device Regulatory Affairs Outsourcing Market, By Services, 2019-2028 (USD Million)
TABLE 758 Israel Medical Device Regulatory Affairs Outsourcing Market, By Product, 2019-2028 (USD Million)
TABLE 759 Israel Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 760 Israel Medical Device Regulatory Affairs Outsourcing Market, By Application, 2019-2028 (USD Million)
TABLE 761 Israel Cardiology in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 762 Israel General and Plastic Surgery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 763 Israel IVD in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 764 Israel Orthopaedic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 765 Israel Diagnostic Imaging in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 766 Israel Dental in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 767 Israel Ophthalmic in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 768 Israel Endoscopy in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 769 Israel Diabetes Care in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 770 Israel Drug Delivery in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 771 Israel Others in Medical Device Regulatory Affairs Outsourcing Market, By Device Type, 2019-2028 (USD Million)
TABLE 772 Israel Medical Device Regulatory Affairs Outsourcing Market, By End User, 2019-2028 (USD Million)
TABLE 773 Rest of Middle East and Africa Medical device regulatory affairs outsourcing Market, By Services, 2019-2028 (USD Million)
Abbildungsverzeichnis
FIGURE 1 Global MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET: SEGMENTATION
FIGURE 2 GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET: DATA TRIANGULATION
FIGURE 3 global Medical device regulatory affairs outsourcing market: DROC ANALYSIS
FIGURE 4 Global Medical device regulatory affairs outsourcing market: Global VS. REGIONAL MARKET ANALYSIS
FIGURE 5 Global Medical device regulatory affairs outsourcing market: COMPANY RESEARCH ANALYSIS
FIGURE 6 GLOBAL Medical device regulatory affairs outsourcing market: MULTIVARIATE MODELLING
FIGURE 7 GLOBAL Medical device regulatory affairs outsourcing market: INTERVIEW DEMOGRAPHICS
FIGURE 8 GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET: DBMR MARKET POSITION GRID
FIGURE 9 GLOBAL MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET: VENDOR SHARE ANALYSIS
FIGURE 10 GLOBAL medical device regulatory affairs outsourcing market: MARKET application COVERAGE GRID
FIGURE 11 Global Medical device regulatory affairs outsourcing market: SEGMENTATION
FIGURE 12 NORTH AMERICA is expected to DOMINATE THE GLOBAL Medical device regulatory affairs outsourcing market, AND ASIA-PACIFIC IS EXPECTED TO GROW WITH THE HIGHEST CAGR IN THE FORECAST PERIOD OF 2021 TO 2028
FIGURE 13 Increasing adoption of outsourcing models for regulatory services is DRIVing THE global Medical device regulatory affairs outsourcing market IN THE FORECAST PERIOD OF 2021 to 2028
FIGURE 14 Regulatory Affairs Services SEGMENT is expected to account for the largest share of the global MEDICAL DEVICE REGULATORY AFFAIRS OUTSOURCING MARKET in 2021 & 2028
FIGURE 15 ASIA-PACIFIC is the fastest growing market FOR the Medical device regulatory affairs outsourcing MANUFACTURERS IN THE FORECAST PERIOD OF 2021 TO 2028
FIGURE 16 DRIVERS, RESTRAINTS, OPPORTUNITIES AND CHALLENGEs OF global Medical Device Regulatory Affairs Outsourcing Market
FIGURE 17 Global Medical device regulatory affairs outsourcing market: By services, 2020
FIGURE 18 Global Medical device regulatory affairs outsourcing market: By services, 2021-2028 (USD Million)
FIGURE 19 Global Medical device regulatory affairs outsourcing market: By services, CAGR (2021-2028)
FIGURE 20 Global Medical device regulatory affairs outsourcing market: By services, LIFELINE CURVE
FIGURE 21 Global Medical device regulatory affairs outsourcing market: By product, 2020
FIGURE 22 Global Medical device regulatory affairs outsourcing market: By product, 2021-2028 (USD million)
FIGURE 23 Global Medical device regulatory affairs outsourcing market: By product, CAGR (2021-2028)
FIGURE 24 Global Medical device regulatory affairs outsourcing market: By product, LIFELINE CURVE
FIGURE 25 Global Medical device regulatory affairs outsourcing market: By device type, 2020
FIGURE 26 Global Medical device regulatory affairs outsourcing market: By device type, 2021-2028 (USD million)
FIGURE 27 Global Medical device regulatory affairs outsourcing market: By device type, CAGR (2021-2028)
FIGURE 28 Global Medical device regulatory affairs outsourcing market: By device type, LIFELINE CURVE
FIGURE 29 Global Medical device regulatory affairs outsourcing market: By application, 2020
FIGURE 30 Global Medical device regulatory affairs outsourcing market: By application, 2021-2028 (USD million)
FIGURE 31 Global Medical device regulatory affairs outsourcing market: By application, CAGR (2021-2028)
FIGURE 32 Global Medical device regulatory affairs outsourcing market: By application, LIFELINE CURVE
FIGURE 33 Global Medical device regulatory affairs outsourcing market: By end user, 2020
FIGURE 34 Global Medical device regulatory affairs outsourcing market: By end user, 2021-2028 (USD million)
FIGURE 35 Global Medical device regulatory affairs outsourcing market: By end user, CAGR (2021-2028)
FIGURE 36 Global Medical device regulatory affairs outsourcing market: By end user, LIFELINE CURVE
FIGURE 37 GLobal medical device regulatory affairs outsourcing MARKET: SNAPSHOT (2020)
FIGURE 38 global medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020)
FIGURE 39 global medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2022 & 2028)
FIGURE 40 global medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020 & 2028)
FIGURE 41 global medical device regulatory affairs outsourcing MARKET: BY SERVICES (2021-2028)
FIGURE 42 North America medical device regulatory affairs outsourcing MARKET: SNAPSHOT (2020)
FIGURE 43 North America medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020)
FIGURE 44 North America medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2021 & 2028)
FIGURE 45 North America medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020 & 2028)
FIGURE 46 North America medical device regulatory affairs outsourcing MARKET: BY SERVICES (2021-2028)
FIGURE 47 Europe medical device regulatory affairs outsourcing MARKET: SNAPSHOT (2020)
FIGURE 48 Europe medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020)
FIGURE 49 Europe medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2021 & 2028)
FIGURE 50 Europe medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020 & 2028)
FIGURE 51 Europe medical device regulatory affairs outsourcing MARKET: BY SERVICES (2021-2028)
FIGURE 52 Asia-Pacific medical device regulatory affairs outsourcing MARKET: SNAPSHOT (2020)
FIGURE 53 Asia-Pacific medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020)
FIGURE 54 Asia-Pacific medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2021 & 2028)
FIGURE 55 Asia-Pacific medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020 & 2028)
FIGURE 56 Asia-Pacific medical device regulatory affairs outsourcing MARKET: BY SERVICES (2021-2028)
FIGURE 57 South America medical device regulatory affairs outsourcing MARKET: SNAPSHOT (2020)
FIGURE 58 South America medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020)
FIGURE 59 South America medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2021 & 2028)
FIGURE 60 South America medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020 & 2028)
FIGURE 61 South America medical device regulatory affairs outsourcing MARKET: BY services (2021-2028)
FIGURE 62 Middle East and Africa medical device regulatory affairs outsourcing MARKET: SNAPSHOT (2020)
FIGURE 63 Middle East and Africa medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020)
FIGURE 64 Middle East and Africa medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2021 & 2028)
FIGURE 65 Middle East and Africa medical device regulatory affairs outsourcing MARKET: BY COUNTRY (2020 & 2028)
FIGURE 66 Middle East and Africa medical device regulatory affairs outsourcing MARKET: BY Services (2021-2028)
FIGURE 67 Global Medical device regulatory affairs outsourcing Market: company share 2020 (%)
FIGURE 68 North America Medical device regulatory affairs outsourcing Market: company share 2020 (%)
FIGURE 69 Europe Medical device regulatory affairs outsourcing Market: company share 2020 (%)
FIGURE 70 Asia-Pacific Medical device regulatory affairs outsourcing Market: company share 2020 (%)

Forschungsmethodik
Die Datenerfassung und Basisjahresanalyse werden mithilfe von Datenerfassungsmodulen mit großen Stichprobengrößen durchgeführt. Die Phase umfasst das Erhalten von Marktinformationen oder verwandten Daten aus verschiedenen Quellen und Strategien. Sie umfasst die Prüfung und Planung aller aus der Vergangenheit im Voraus erfassten Daten. Sie umfasst auch die Prüfung von Informationsinkonsistenzen, die in verschiedenen Informationsquellen auftreten. Die Marktdaten werden mithilfe von marktstatistischen und kohärenten Modellen analysiert und geschätzt. Darüber hinaus sind Marktanteilsanalyse und Schlüsseltrendanalyse die wichtigsten Erfolgsfaktoren im Marktbericht. Um mehr zu erfahren, fordern Sie bitte einen Analystenanruf an oder geben Sie Ihre Anfrage ein.
Die wichtigste Forschungsmethodik, die vom DBMR-Forschungsteam verwendet wird, ist die Datentriangulation, die Data Mining, die Analyse der Auswirkungen von Datenvariablen auf den Markt und die primäre (Branchenexperten-)Validierung umfasst. Zu den Datenmodellen gehören ein Lieferantenpositionierungsraster, eine Marktzeitlinienanalyse, ein Marktüberblick und -leitfaden, ein Firmenpositionierungsraster, eine Patentanalyse, eine Preisanalyse, eine Firmenmarktanteilsanalyse, Messstandards, eine globale versus eine regionale und Lieferantenanteilsanalyse. Um mehr über die Forschungsmethodik zu erfahren, senden Sie eine Anfrage an unsere Branchenexperten.
Anpassung möglich
Data Bridge Market Research ist ein führendes Unternehmen in der fortgeschrittenen formativen Forschung. Wir sind stolz darauf, unseren bestehenden und neuen Kunden Daten und Analysen zu bieten, die zu ihren Zielen passen. Der Bericht kann angepasst werden, um Preistrendanalysen von Zielmarken, Marktverständnis für zusätzliche Länder (fordern Sie die Länderliste an), Daten zu klinischen Studienergebnissen, Literaturübersicht, Analysen des Marktes für aufgearbeitete Produkte und Produktbasis einzuschließen. Marktanalysen von Zielkonkurrenten können von technologiebasierten Analysen bis hin zu Marktportfoliostrategien analysiert werden. Wir können so viele Wettbewerber hinzufügen, wie Sie Daten in dem von Ihnen gewünschten Format und Datenstil benötigen. Unser Analystenteam kann Ihnen auch Daten in groben Excel-Rohdateien und Pivot-Tabellen (Fact Book) bereitstellen oder Sie bei der Erstellung von Präsentationen aus den im Bericht verfügbaren Datensätzen unterstützen.