Globaler Markt für die Behandlung der Muskeldystrophie Duchenne, nach Behandlungstyp (molekularbasierte Therapien, Steroidtherapie und andere), Therapie (Exon-Skipping-Ansatz, Mutationsunterdrückung und Dystrophin-zielgerichtete Therapien), Verabreichungsweg (oral, parenteral und andere), Endbenutzer (Krankenhäuser, Fachkliniken, häusliche Pflege und andere), Vertriebskanal (Krankenhausapotheke, Einzelhandelsapotheke und Online-Apotheke) – Branchentrends und Prognose bis 2030.
Marktanalyse und -größe für die Behandlung der Muskeldystrophie Duchenne
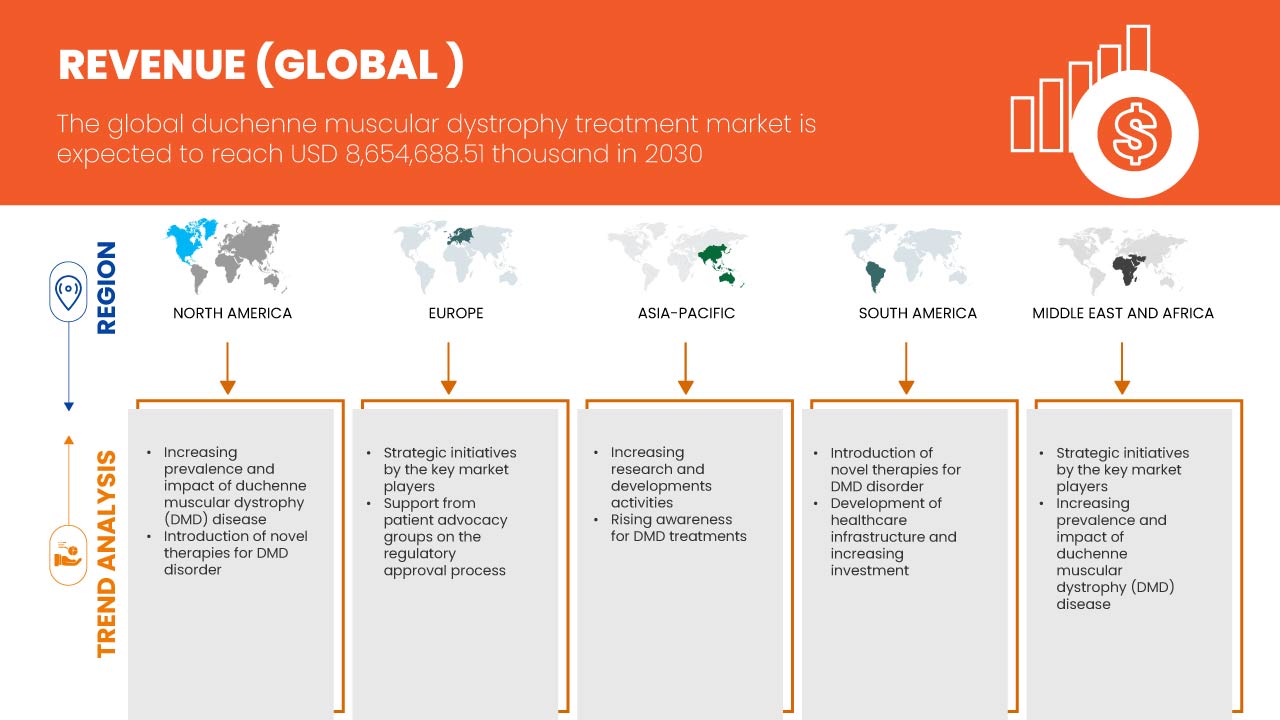
Die wichtigsten Faktoren, die das Marktwachstum antreiben dürften, sind das steigende Bewusstsein für die Behandlung von DMD und die Einführung neuer Therapien für DMD. Darüber hinaus sind die zunehmende Prävalenz und Auswirkung von DMD ein weiterer wichtiger Treiber, der das Marktwachstum ankurbeln dürfte. Ein Anstieg der Anzahl klinischer Studien ist ein aktueller Trend, der das Marktwachstum ebenfalls ankurbeln dürfte. Die begrenzte Verfügbarkeit von DMD-Behandlungen aufgrund fortschrittlicher Technologie und die hohen Behandlungskosten von DMD dürften das Marktwachstum jedoch bremsen.
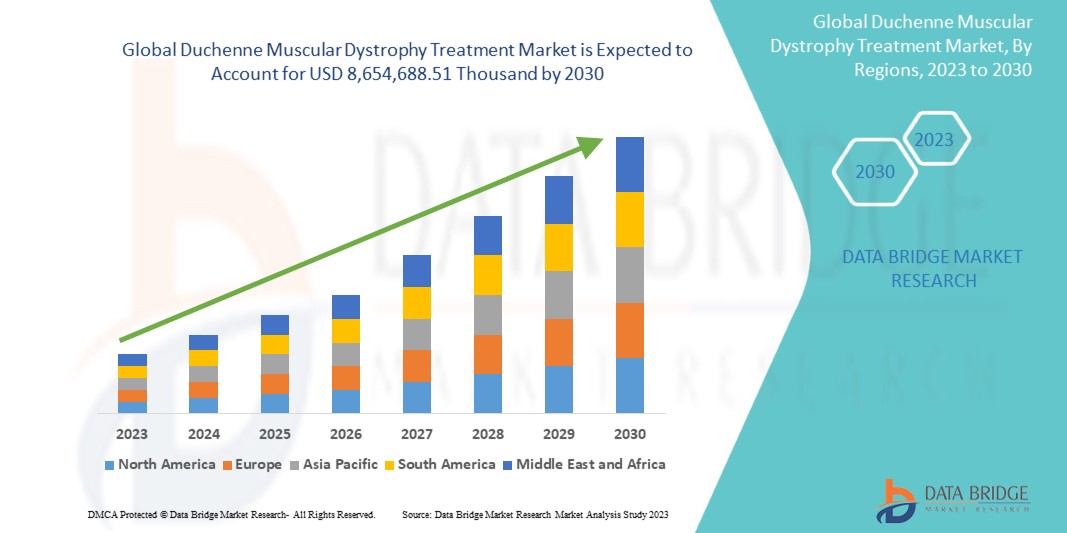
Data Bridge Market Research analysiert, dass der globale Markt für Behandlungen der Muskeldystrophie Duchenne bis 2030 voraussichtlich 8.654.688,51.000 USD erreichen wird, was einer durchschnittlichen jährlichen Wachstumsrate von 16,8 % im Prognosezeitraum 2023–2030 entspricht. Dieser Marktbericht behandelt auch ausführlich Preisanalysen und technologische Fortschritte.
|
Berichtsmetrik |
Details |
|
Prognosezeitraum |
2023 bis 2030 |
|
Basisjahr |
2022 |
|
Historische Jahre |
2021 (Anpassbar auf 2015 – 2020) |
|
Quantitative Einheiten |
Umsatz in Tausend USD |
|
Abgedeckte Segmente |
Behandlungstyp (molekularbasierte Therapien, Steroidtherapie und andere), Therapie (Exon-Skipping-Ansatz, Mutationsunterdrückung und Dystrophin-zielgerichtete Therapien), Verabreichungsweg (oral, parenteral und andere), Endbenutzer (Krankenhäuser, Fachkliniken, häusliche Pflege und andere), Vertriebskanal (Krankenhausapotheke, Einzelhandelsapotheke und Online-Apotheke) |
|
Abgedeckte Länder |
USA, Kanada, Mexiko, Deutschland, Frankreich, Großbritannien, Italien, Spanien, Russland, Türkei, Belgien, Niederlande, Schweiz, Restliches Europa, Japan, China, Südkorea, Indien, Australien, Singapur, Thailand, Malaysia, Indonesien, Philippinen, Restlicher Asien-Pazifik-Raum, Brasilien, Argentinien, Restliches Südamerika, Südafrika, Saudi-Arabien, Vereinigte Arabische Emirate, Ägypten, Israel und Restlicher Naher Osten und Afrika |
|
Abgedeckte Marktteilnehmer |
Sarepta Therapeutics, Inc., GSK plc., Capricor Therapeutics, Inc., Dyne Therapeutics, Solid Biosciences Inc., BioMarin, Stealth BioTherapeutics Inc., Avidity Biosciences, ReveraGen BioPharma, Inc. PTC Therapeutics., NS Pharma, Inc, ITALFARMACO SpA, FibroGen, Inc, SANTHERA PHARMACEUTICALS, Pfizer Inc., F. Hoffmann-La Roche Ltd, Akashi RX und TAIHO PHARMACEUTICAL CO., LTD und unter anderem |
Marktdefinition
Die Muskeldystrophie Duchenne (DMD) ist eine seltene genetische Erkrankung, die durch fortschreitende Muskeldegeneration und -schwäche gekennzeichnet ist. Die primäre Behandlung von DMD umfasst die Verwendung von Kortikosteroiden wie Prednison oder Deflazacort. Diese Medikamente helfen, Entzündungen zu reduzieren und die Muskeldegeneration zu verzögern, wodurch letztendlich die Gehfähigkeit verlängert und die Muskelfunktion erhalten bleibt. Es hat sich gezeigt, dass die Behandlung mit Kortikosteroiden die Muskelkraft, die Atemfunktion und die allgemeine Lebensqualität von Personen mit DMD verbessert.
Globale Marktdynamik für die Behandlung der Muskeldystrophie Duchenne
In diesem Abschnitt geht es um das Verständnis der Markttreiber, Vorteile, Einschränkungen und Herausforderungen. All dies wird im Folgenden ausführlich erläutert:
Treiber
- Zunehmende Prävalenz und Auswirkung der Duchenne-Muskeldystrophie (DMD )
Die Muskeldystrophie Duchenne (DMD) ist eine seltene und schwächende genetische Erkrankung, die durch fortschreitende Muskelschwäche und Funktionsverlust gekennzeichnet ist. Sie betrifft hauptsächlich Männer, wobei die Symptome typischerweise in der frühen Kindheit auftreten. Die Zahl der Menschen, bei denen DMD diagnostiziert wird, nimmt ebenfalls zu, und diese steigende Prävalenz hat einen Bedarf an wirksamen Behandlungen und Therapien geschaffen, da die Weltbevölkerung weiterhin wächst.
Die steigende Prävalenz von DMD hat auch die Bemühungen von Patienten und medizinischem Fachpersonal verstärkt, sich für DMD einzusetzen. Diese Gruppen spielen eine wichtige Rolle bei der Sensibilisierung für DMD, bei der Befürwortung von Forschungsgeldern und bei der schnelleren Zulassung potenzieller Behandlungen. Die DMD-Gemeinschaft wächst, daher ist auch die Weiterentwicklung von Technologien und Investitionen in diesem Bereich der Gesundheitsversorgung von Bedeutung.
- Einführung neuer Therapien für die DMD- Störung
Das Aufkommen innovativer Behandlungsansätze führt zu einem Anstieg der DMD-Behandlung und trägt erheblich zur Marktexpansion bei. Traditionelle Behandlungsoptionen konzentrieren sich in erster Linie auf Symptommanagement und unterstützende Pflege. Die Einführung neuartiger Therapien stellt jedoch einen Paradigmenwechsel dar, da sie auf die für DMD verantwortlichen genetischen Grundmutationen abzielen.
Darüber hinaus haben sich Exon-Skipping-Medikamente als eine weitere vielversprechende Therapieklasse für DMD herausgestellt. Diese Medikamente sind so konzipiert, dass sie bestimmte Exons im Dystrophin-Gen „überspringen“, wodurch die Produktion eines verkürzten, aber teilweise funktionsfähigen Dystrophin-Proteins ermöglicht wird. Diese Medikamente können das Fortschreiten der Krankheit deutlich verlangsamen und die Lebensqualität von Menschen mit DMD verbessern. Die Entwicklung und Zulassung dieser Therapien hat die Erwartungen von DMD-Patienten geweckt und wird voraussichtlich das Marktwachstum weiter vorantreiben.
Gelegenheit
- Ausbau der Gesundheitsinfrastruktur und steigende Investitionen
Patienten mit DMD können dank einer besseren Gesundheitsinfrastruktur, zu der gut ausgestattete Krankenhäuser und Diagnoseeinrichtungen gehören, früher diagnostiziert werden. Eine frühe Diagnose ermöglicht ein rechtzeitiges Eingreifen und den Beginn der Behandlung, was möglicherweise das Fortschreiten der Krankheit verlangsamt und die Behandlungsergebnisse verbessert. Entwickelte Gesundheitssysteme verfügen häufig über spezialisierte Zentren und Kliniken, die sich auf seltene Krankheiten wie DMD spezialisiert haben. Diese Zentren bieten umfassende Betreuung, einschließlich Zugang zu spezialisiertem medizinischem Fachpersonal, Physiotherapie und unterstützenden Diensten, die die Lebensqualität von DMD-Patienten verbessern können.
Entwickelte Gesundheitssysteme bieten oft eine breite Palette unterstützender Dienstleistungen wie Physio- und Ergotherapie , Hilfsmittel und Beratung an, die die Lebensqualität von DMD-Patienten deutlich verbessern können. Regierungen, private Investoren und philanthropische Organisationen investieren eher in die Arzneimittelentwicklung für seltene Krankheiten wie DMD, wenn eine gut etablierte Gesundheitsinfrastruktur vorhanden ist. Diese Investitionen können Forschung, klinische Studien und die Entwicklung innovativer Therapien unterstützen.
Einschränkung/ Herausforderung
- Eingeschränkte Verfügbarkeit von DMD-Behandlungen aufgrund fehlender fortschrittlicher Technologie
Für DMD stehen nur begrenzte Behandlungsmöglichkeiten zur Verfügung, sodass den Patienten und ihren Angehörigen kaum Möglichkeiten bleiben, das Fortschreiten der Krankheit zu verlangsamen oder ihr allgemeines Wohlbefinden zu verbessern.
Der technologische Fortschritt führt häufig zur Entwicklung hochspezialisierter und innovativer Behandlungen für DMD, wie Gentherapien oder personalisierte Medizinansätze. Die Entwicklung, Herstellung und Verabreichung dieser hochmodernen Therapien ist komplex. Insbesondere die Entwicklung von Gentherapien erfordert komplexe Prozesse und spezielle Herstellungsverfahren. Diese Therapien erfordern die Modifikation oder den Ersatz fehlerhafter Gene, um die zugrunde liegende Ursache von DMD zu bekämpfen, was zu hohen Behandlungskosten führt. Darüber hinaus trägt die begrenzte Anzahl von Produktionsanlagen, die Gentherapien herstellen können, zu ihrer begrenzten Verfügbarkeit bei.
Aufgrund ihrer Komplexität können Herstellung und Verabreichung moderner DMD-Behandlungen eine Herausforderung darstellen, was zu ihrer begrenzten Verfügbarkeit beiträgt. Für die Herstellung können spezielle Einrichtungen und Fachkenntnisse erforderlich sein, und die Verabreichung dieser Behandlungen kann spezialisiertes medizinisches Fachpersonal erfordern, das in bestimmten Regionen knapp sein kann.
Jüngste Entwicklungen
- Im März 2023 gab Dyne Therapeutics bekannt, dass DYNE-251, ein Prüfpräparat für Mutationen der Muskeldystrophie Duchenne (DMD), die durch Überspringen von Exon 51 beeinflusst werden, von der US-amerikanischen FDA als Orphan-Arzneimittel und seltene pädiatrische Krankheit zugelassen wurde. DYNE-251 wird in der klinischen Phase-1/2-Studie DELIVER evaluiert. Dies hilft dem Unternehmen, seine Produktkategorie und seinen Gesamtumsatz zu steigern.
- Im August 2022 gab FibroGen, Inc. Topline-Daten aus der Phase-3-Studie LELANTOS-2 mit Pamrevlumab zur Behandlung ambulanter Patienten mit DMD unter systemischer Kortikosteroid-Hintergrundtherapie bekannt. Dies half dem Unternehmen, sein Pipeline-Portfolio zu stärken.
Globaler Marktumfang für die Behandlung der Muskeldystrophie Duchenne
Der globale Markt für Behandlungen der Muskeldystrophie Duchenne ist in fünf wichtige Segmente unterteilt, basierend auf Behandlungsart, Therapie, Verabreichungsweg, Endverbraucher und Vertriebskanal. Das Wachstum dieser Segmente hilft Ihnen bei der Analyse schwacher Wachstumssegmente in den Branchen und bietet den Benutzern einen wertvollen Marktüberblick und Markteinblicke, die ihnen bei der strategischen Entscheidungsfindung zur Identifizierung der wichtigsten Marktanwendungen helfen.
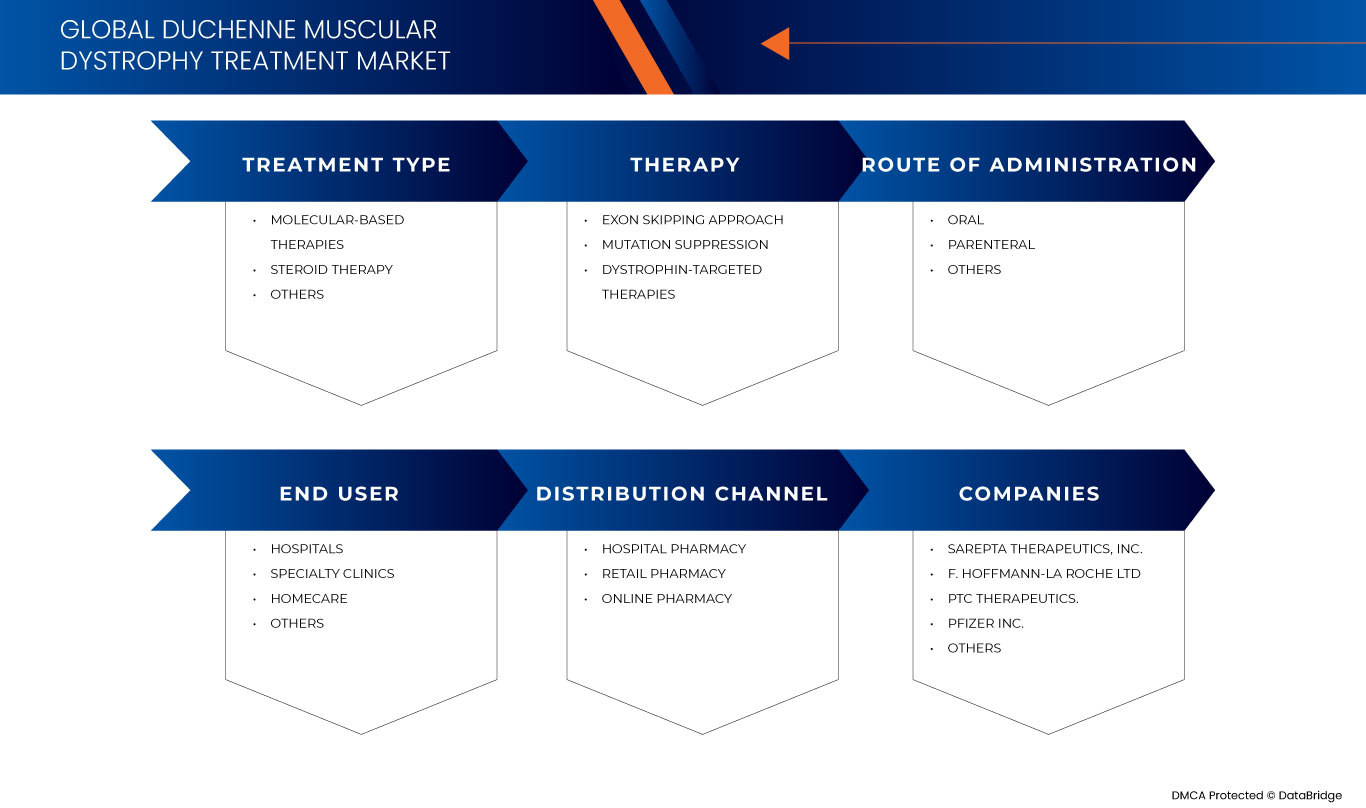
Behandlungstyp
- Molekularbasierte Therapien
- Steroidtherapie
- Sonstiges
Auf der Grundlage der Behandlungsart ist der Markt in molekularbasierte Therapien, Steroidtherapie und andere segmentiert.
Therapie
- Exon-Skipping-Ansatz
- Unterdrückung von Mutationen
- Dystrophin-gezielte Therapien
Auf der Grundlage der Therapie ist der Markt in die Ansätze Exon-Skipping, Mutationsunterdrückung und Dystrophin-gezielte Therapien segmentiert.
Verabreichungsweg
- Oral
- Parenterale
- Sonstiges
Auf der Grundlage der Verabreichungsart wird der Markt in oral, parenteral und sonstige Verabreichung unterteilt.
Endbenutzer
- Krankenhäuser
- Häusliche Gesundheitspflege
- Spezialkliniken
- Sonstiges
Auf der Grundlage des Endbenutzers ist der Markt in Krankenhäuser, häusliche Pflege, Fachkliniken und Sonstige segmentiert.
Vertriebskanal
- Krankenhausapotheke
- Online-Apotheke
- Einzelhandelsapotheke
Auf der Grundlage der Vertriebskanäle ist der Markt in Krankenhausapotheken, Online-Apotheken und Einzelhandelsapotheken segmentiert .
Globale Marktanalyse/Einblicke zur Behandlung der Muskeldystrophie Duchenne
Der globale Markt für die Behandlung der Muskeldystrophie Duchenne ist basierend auf Behandlungsart, Therapie, Verabreichungsweg, Endverbraucher und Vertriebskanal in fünf wichtige Segmente unterteilt.
Die in diesem globalen Marktbericht zur Behandlung der Muskeldystrophie Duchenne abgedeckten Länder sind die USA, Kanada, Mexiko, Deutschland, Frankreich, Großbritannien, Italien, Spanien, Russland, Türkei, Belgien, Niederlande, Schweiz, übriges Europa, Japan, China, Südkorea, Indien, Australien, Singapur, Thailand, Malaysia, Indonesien, Philippinen, übriger asiatisch-pazifischer Raum, Brasilien, Argentinien, übriges Südamerika, Südafrika, Saudi-Arabien, Vereinigte Arabische Emirate, Ägypten, Israel sowie der restliche Nahe Osten und Afrika.
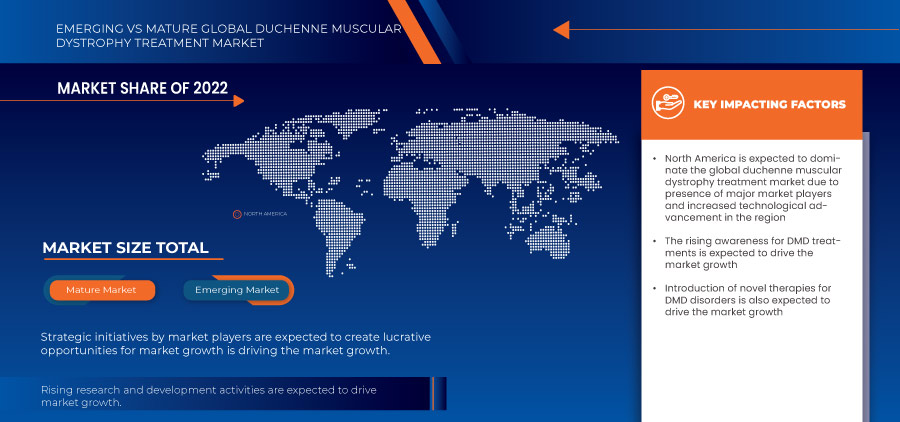
Aufgrund des wachsenden Bewusstseins und der zunehmenden Vorsorgeuntersuchungen für DMD werden die USA in Nordamerika voraussichtlich dominieren. In Europa wird Deutschland aufgrund der steigenden Prävalenz von Muskeldystrophie und der Verbreitung neuer technologischer Fortschritte voraussichtlich dominieren. China wird voraussichtlich den asiatisch-pazifischen Raum dominieren, da die strategischen Initiativen der wichtigsten Marktteilnehmer erheblich expandieren.
Der Länderabschnitt des Berichts enthält auch einzelne marktbeeinflussende Faktoren und Änderungen der Marktregulierung, die sich auf die aktuellen und zukünftigen Trends des Marktes auswirken. Datenpunkte wie die Analyse der nachgelagerten und vorgelagerten Wertschöpfungskette, technische Trends, Porters Fünf-Kräfte-Analyse und Fallstudien sind einige der Hinweise, die zur Prognose des Marktszenarios für einzelne Länder verwendet werden. Bei der Bereitstellung einer Prognoseanalyse der Länderdaten werden auch die Präsenz und Verfügbarkeit globaler Marken und ihre Herausforderungen aufgrund großer oder geringer Konkurrenz durch lokale und inländische Marken, die Auswirkungen inländischer Zölle und Handelsrouten berücksichtigt.
Wettbewerbsumfeld und globale Analyse der Marktanteile zur Behandlung der Muskeldystrophie Duchenne
Das Wettbewerbsumfeld auf dem globalen Markt für die Behandlung der Muskeldystrophie Duchenne liefert Einzelheiten zum Wettbewerber. Die enthaltenen Einzelheiten umfassen Unternehmensübersicht, Unternehmensfinanzen, erzielten Umsatz, Marktpotenzial, neue Marktinitiativen, globale Präsenz, Produktionsstandorte und -anlagen, Produktionskapazitäten, Stärken und Schwächen des Unternehmens, Produkteinführung, Produktbreite und -umfang sowie Anwendungsdominanz. Die oben angegebenen Datenpunkte beziehen sich nur auf den Fokus der Unternehmen in Bezug auf den Markt.
Zu den wichtigsten Marktteilnehmern auf dem globalen Markt für die Behandlung der Muskeldystrophie Duchenne zählen unter anderem Sarepta Therapeutics, Inc., GSK plc., Capricor Therapeutics, Inc., Dyne Therapeutics, Solid Biosciences Inc., BioMarin, Stealth BioTherapeutics Inc., Avidity Biosciences, ReveraGen BioPharma, Inc. PTC Therapeutics., NS Pharma, Inc, ITALFARMACO SpA, FibroGen, Inc, SANTHERA PHARMACEUTICALS, Pfizer Inc., F. Hoffmann-La Roche Ltd, Akashi RX und TAIHO PHARMACEUTICAL CO., LTD.
SKU-
Erhalten Sie Online-Zugriff auf den Bericht zur weltweit ersten Market Intelligence Cloud
- Interaktives Datenanalyse-Dashboard
- Unternehmensanalyse-Dashboard für Chancen mit hohem Wachstumspotenzial
- Zugriff für Research-Analysten für Anpassungen und Abfragen
- Konkurrenzanalyse mit interaktivem Dashboard
- Aktuelle Nachrichten, Updates und Trendanalyse
- Nutzen Sie die Leistungsfähigkeit der Benchmark-Analyse für eine umfassende Konkurrenzverfolgung
Inhaltsverzeichnis
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF THE GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATIONS
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 DBMR TRIPOD DATA VALIDATION MODEL
2.5 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.6 MULTIVARIATE MODELLING
2.7 TREATMENT TYPE SEGMENT LIFELINE CURVE
2.8 MARKET END USER COVERAGE GRID
2.9 DBMR MARKET POSITION GRID
2.1 VENDOR SHARE ANALYSIS
2.11 SECONDARY SOURCES
2.12 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PESTEL'S MODEL
4.2 PORTER'S FIVE FORCES MODEL
4.3 PRICING ANALYSIS
5 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: REGULATIONS
5.1 REGULATIONS IN U.S.
5.2 REGULATTIONS IN EUROPE
5.3 REGULATTIONS IN AUSTRALIA
5.4 REGULATIONS IN SOUTH AFRICA
5.5 REGULATIONS IN BRAZIL
6 MARKET OVERVIEW
6.1 DRIVERS
6.1.1 INCREASING PREVALENCE AND IMPACT OF DUCHENNE MUSCULAR DYSTROPHY (DMD) DISEASE
6.1.2 INTRODUCTION OF NOVEL THERAPIES FOR DMD DISORDER
6.1.3 RISING AWARENESS FOR DMD TREATMENTS
6.1.4 INCREASE IN THE NUMBER OF CLINICAL TRIALS IS A RECENT TREND
6.2 RESTRAINTS
6.2.1 LIMITED AVAILABILITY OF DMD TREATMENTS DUE TO LACK OF ADVANCE TECHNOLOGY
6.2.2 HIGH TREATMENT COST OF DMD DISORDER
6.3 OPPORTUNITIES
6.3.1 DEVELOPMENT OF HEALTHCARE INFRASTRUCTURE AND INCREASING INVESTMENT
6.3.2 RISING PATIENT INCLINATION TOWARDS PERSONALIZED AND EFFECTIVE THERAPIES
6.3.3 STRATEGIC INITIATIVES BY THE KEY MARKET PLAYERS
6.3.4 SUPPORT FROM PATIENT ADVOCACY GROUPS ON THE REGULATORY APPROVAL PROCESS
6.4 CHALLENGES
6.4.1 LACK OF STANDARDIZATION IN DMD DIAGNOSIS
6.4.2 ETHICAL CONSIDERATIONS RELATED TO PERMANENT ALTERATION OF A PATIENT'S GENETIC CODE
7 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE
7.1 OVERVIEW
7.2 MOLECULAR-BASED THERAPIES
7.2.1 ANTISENSE OLIGONUCLEOTIDE THERAPY
7.2.1.1 EXONDYS 51
7.2.1.2 AMONDYS 45
7.2.1.3 VYONDYS 53
7.3 NONSENSE MUTATION
7.3.1.1 Translarna
7.4 STEROID THERAPY
7.4.1 PREDNISONE
7.4.2 DEFLAZACORT
7.5 OTHERS
8 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY
8.1 OVERVIEW
8.2 EXON SKIPPING APPROACH
8.2.1 MULTI-EXON SKIPPING APPROACH
8.2.2 SINGLE-EXON SKIPPING APPROACH
8.3 MUTATION SUPPRESSION
8.4 DYSTROPHIN-TARGETED THERAPIES
8.4.1 GENE THERAPIES
8.4.2 CELL THERAPIES
8.4.2.1 Gene Editing
8.4.2.2 Gene Addition
9 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION
9.1 OVERVIEW
9.2 PARENTERAL
9.3 ORAL
9.4 OTHERS
10 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER
10.1 OVERVIEW
10.2 HOSPITALS
10.3 SPECIALTY CLINICS
10.4 HOMECARE
10.5 OTHERS
11 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL
11.1 OVERVIEW
11.2 HOSPITAL PHARMACY
11.3 RETAIL PHARMACY
11.4 ONLINE PHARMACY
12 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION
12.1 OVERVIEW
12.2 NORTH AMERICA
12.2.1 U.S.
12.2.2 CANADA
12.2.3 MEXICO
12.3 EUROPE
12.3.1 GERMANY
12.3.2 FRANCE
12.3.3 U.K.
12.3.4 ITALY
12.3.5 SPAIN
12.3.6 RUSSIA
12.3.7 TURKEY
12.3.8 BELGIUM
12.3.9 NETHERLANDS
12.3.10 SWITZERLAND
12.3.11 REST OF EUROPE
12.4 ASIA-PACIFIC
12.4.1 CHINA
12.4.2 JAPAN
12.4.3 INDIA
12.4.4 SOUTH KOREA
12.4.5 AUSTRALIA
12.4.6 SINGAPORE
12.4.7 THAILAND
12.4.8 MALAYSIA
12.4.9 INDONESIA
12.4.10 PHILIPPINES
12.4.11 REST OF ASIA-PACIFIC
12.5 SOUTH AMERICA
12.5.1 BRAZIL
12.5.2 ARGENTINA
12.5.3 REST OF SOUTH AMERICA
12.6 MIDDLE EAST AND AFRICA
12.6.1 SOUTH AFRICA
12.6.2 SAUDI ARABIA
12.6.3 U.A.E
12.6.4 EGYPT
12.6.5 ISRAEL
12.6.6 REST OF MIDDLE EAST AND AFRICA
13 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, COMPANY LANDSCAPE
13.1 COMPANY SHARE ANALYSIS: GLOBAL
14 NORTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, COMPANY LANDSCAPE
14.1 COMPANY SHARE ANALYSIS: NORTH AMERICA
15 EUROPE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, COMPANY LANDSCAPE
15.1 COMPANY SHARE ANALYSIS: EUROPE
16 ASIA-PACIFIC DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, COMPANY LANDSCAPE
16.1 COMPANY SHARE ANALYSIS: ASIA-PACIFIC
17 SWOT ANALYSIS
18 COMPANY PROFILES
18.1 SAREPTA THERAPEUTICS, INC.
18.1.1 COMPANY SNAPSHOT
18.1.1 REVENUE ANALYSIS
18.1.2 COMPANY SHARE ANALYSIS
18.1.3 PRODUCT PORTFOLIO
18.1.4 PIPELINE PORTFOLIO
18.1.5 RECENT DEVELOPMENTS
18.2 F. HOFFMANN-LA ROCHE LTD
18.2.1 COMPANY SNAPSHOT
18.2.2 REVENUE ANALYSIS
18.2.3 COMPANY SHARE ANALYSIS
18.2.4 PRODUCT PORTFOLIO
18.2.5 PIPELINE PORTFOLIO
18.2.6 RECENT DEVELOPMENTS
18.3 PTC THERAPEUTICS.
18.3.1 COMPANY SNAPSHOT
18.3.2 REVENUE ANALYSIS
18.3.3 COMPANY SHARE ANALYSIS
18.3.4 PRODUCT PORTFOLIO
18.3.5 RECENT DEVELOPMENT
18.4 PFIZER INC.
18.4.1 COMPANY SNAPSHOT
18.4.2 REVENUE ANALYSIS
18.4.3 COMPANY SHARE ANALYSIS
18.4.4 PIPELINE PORTFOLIO
18.4.5 PRODUCT PORTFOLIO
18.4.6 RECENT DEVELOPMENT
18.5 AKASHI RX
18.5.1 COMPANY SNAPSHOT
18.5.2 PIPELINE PORTFOLIO
18.5.3 RECENT DEVELOPMENTS
18.6 AVIDITY BIOSCIENCES
18.6.1 COMPANY SNAPSHOT
18.6.2 REVENUE ANALYSIS
18.6.3 PIPELINE PORTFOLIO
18.6.4 RECENT DEVELOPMENTS
18.7 BIOMARIN
18.7.1 COMPANY SNAPSHOT
18.7.2 REVENUE ANALYSIS
18.7.3 PIPELINE PORTFOLIO
18.7.4 RECENT DEVELOPMENTS
18.8 CAPRICOR THERAPEUTICS, INC.
18.8.1 COMPANY SNAPSHOT
18.8.2 PIPELINE PORTFOLIO
18.8.3 RECENT DEVELOPMENTS
18.9 DYNE THERAPEUTICS
18.9.1 COMPANY SNAPSHOT
18.9.2 PIPELINE PORTFOLIO
18.9.3 RECENT DEVELOPMENTS
18.1 FIBROGEN, INC.
18.10.1 COMPANY SNAPSHOT
18.10.2 REVENUE ANALYSIS
18.10.3 PIPELINE PORTFOLIO
18.10.4 RECENT DEVELOPMENT
18.11 ITALFARMACO S.P.A.
18.11.1 COMPANY SNAPSHOT
18.11.2 PIPELINE PORTFOLIO
18.11.3 RECENT DEVELOPMENT
18.12 NS PHARMA, INC.
18.12.1 COMPANY SNAPSHOT
18.12.2 PIPELINE PORTFOLIO
18.12.3 RECENT DEVELOPMENTS
18.13 REVERAGEN BIOPHARMA, INC.
18.13.1 COMPANY SNAPSHOT
18.13.2 PIPELINE PORTFOLIO
18.13.3 RECENT DEVELOPMENT
18.14 SANTHERA PHARMACEUTICALS
18.14.1 COMPANY SNAPSHOT
18.14.2 REVENUE ANALYSIS
18.14.3 PIPELINE PORTFOLIO
18.14.4 RECENT DEVELOPMENT
18.15 TAIHO PHARMACEUTICAL CO., LTD.
18.15.1 COMPANY SNAPSHOT
18.15.2 PIPELINE PORTFOLIO
18.15.3 RECENT DEVELOPMENTS
18.16 SOLID BIOSCIENCES INC.
18.16.1 COMPANY SNAPSHOT
18.16.2 PIPELINE PORTFOLIO
18.16.3 RECENT DEVELOPMENT
18.17 STEALTH BIOTHERAPEUTICS INC
18.17.1 COMPANY SNAPSHOT
18.17.2 PIPELINE PORTFOLIO
18.17.3 RECENT DEVELOPMENT
19 QUESTIONNAIRE
20 RELATED REPORTS
Tabellenverzeichnis
TABLE 1 LIST OF PRICES FOR APPROVED DRUGS OF THE GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET
TABLE 2 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE , 2021-2030 (USD THOUSAND)
TABLE 3 GLOBAL MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 4 GLOBAL MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 5 GLOBAL ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 6 GLOBAL NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 7 GLOBAL STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 8 GLOBAL STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 9 GLOBAL OTHERS IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 10 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 11 GLOBAL EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 12 GLOBAL EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 13 GLOBAL MUTATION SUPPRESSION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 14 GLOBAL DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 15 GLOBAL DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 16 GLOBAL CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 17 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION , 2021-2030 (USD THOUSAND)
TABLE 18 GLOBAL PARENTERAL IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 19 GLOBAL ORAL IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 20 GLOBAL OTHERS IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 21 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USERS, 2021-2030 (USD THOUSAND)
TABLE 22 GLOBAL HOSPITALS IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 23 GLOBAL SPECIALITY CLINICS IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 24 GLOBAL HOMECARE IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 25 GLOBAL OTHERS IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 26 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 27 GLOBAL HOSPITAL PHARMACY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 28 GLOBAL RETAIL PHARMACY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 29 GLOBAL ONLINE PHARMACY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 30 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY REGION, 2021-2030 (USD THOUSAND)
TABLE 31 NORTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY COUNTRY, 2021-2030 (USD THOUSAND) COUNTRY
TABLE 32 NORTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 33 NORTH AMERICA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 34 NORTH AMERICA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 35 NORTH AMERICA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 36 NORTH AMERICA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 37 NORTH AMERICA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 38 NORTH AMERICA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 39 NORTH AMERICA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 40 NORTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 41 NORTH AMERICA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 42 NORTH AMERICA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 43 NORTH AMERICA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 44 NORTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 45 NORTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 46 NORTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 47 U.S. DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 48 U.S. MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 49 U.S. ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 50 U.S. ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 51 U.S. ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 52 U.S. NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 53 U.S. NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 54 U.S. NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 55 U.S. STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 56 U.S. STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 57 U.S. STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 58 U.S.DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 59 U.S. EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 60 U.S. DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 61 U.S. CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 62 U.S. DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 63 U.S. DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 64 U.S. DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 65 CANADA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 66 CANADA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 67 CANADA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 68 CANADA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 69 CANADA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 70 CANADA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 71 CANADA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 72 CANADA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 73 CANADA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 74 CANADA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 75 CANADA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 76 CANADA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 77 CANADA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 78 CANADA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 79 CANADA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 80 CANADA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 81 CANADA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 82 CANADA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 83 MEXICO DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 84 MEXICO MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 85 MEXICO ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 86 MEXICO ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 87 MEXICO ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 88 MEXICO NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 89 MEXICO NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 90 MEXICO NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 91 MEXICO STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 92 MEXICO STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 93 MEXICO STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 94 MEXICO DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 95 MEXICO EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 96 MEXICO DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 97 MEXICO CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 98 MEXICO DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 99 MEXICO DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 100 MEXICO DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 101 EUROPE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY COUNTRY, 2021-2030 (USD THOUSAND)
TABLE 102 EUROPE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 103 EUROPE MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 104 EUROPE ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 105 EUROPE ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 106 EUROPE NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 107 EUROPE NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 108 EUROPE STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 109 EUROPE STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 110 EUROPE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 111 EUROPE EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 112 EUROPE DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 113 EUROPE CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 114 EUROPE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 115 EUROPE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 116 EUROPE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 117 GERMANY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 118 GERMANY MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 119 GERMANY ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 120 GERMANY ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 121 GERMANY ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 122 GERMANY NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 123 GERMANY NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 124 GERMANY NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 125 GERMANY STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 126 GERMANY STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 127 GERMANY STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 128 GERMANY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 129 GERMANY EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 130 GERMANY DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 131 GERMANY CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 132 GERMANY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 133 GERMANY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 134 GERMANY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 135 FRANCE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 136 FRANCE MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 137 FRANCE ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 138 FRANCE ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 139 FRANCE ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 140 FRANCE NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 141 FRANCE NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 142 FRANCE NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 143 FRANCE STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 144 FRANCE STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 145 FRANCE STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 146 FRANCE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 147 FRANCE EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 148 FRANCE DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 149 FRANCE CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 150 FRANCE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 151 FRANCE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 152 FRANCE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 153 U.K. DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 154 U.K. MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 155 U.K. ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 156 U.K. ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 157 U.K. ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 158 U.K. NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 159 U.K. NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 160 U.K. NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 161 U.K. STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 162 U.K. STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 163 U.K. STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 164 U.K. DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 165 U.K. EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 166 U.K. DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 167 U.K. CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 168 U.K. DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 169 U.K. DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 170 U.K. DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 171 ITALY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 172 ITALY MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 173 ITALY ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 174 ITALY ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 175 ITALY ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 176 ITALY NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 177 ITALY NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 178 ITALY NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 179 ITALY STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 180 ITALY STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 181 ITALY STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 182 ITALYDUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 183 ITALY EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 184 ITALY DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 185 ITALY CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 186 ITALY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 187 ITALY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 188 ITALY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 189 SPAIN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 190 SPAIN MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 191 SPAIN ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 192 SPAIN ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 193 SPAIN ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 194 SPAIN NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 195 SPAIN NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 196 SPAIN NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 197 SPAIN STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 198 SPAIN STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 199 SPAIN STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 200 SPAIN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 201 SPAIN EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 202 SPAIN DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 203 SPAIN CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 204 SPAIN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 205 SPAIN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 206 SPAIN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 207 RUSSIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 208 RUSSIA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 209 RUSSIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 210 RUSSIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 211 RUSSIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 212 RUSSIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 213 RUSSIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 214 RUSSIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 215 RUSSIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 216 RUSSIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 217 RUSSIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 218 RUSSIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 219 RUSSIA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 220 RUSSIA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 221 RUSSIA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 222 RUSSIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 223 RUSSIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 224 RUSSIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 225 TURKEY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 226 TURKEY MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 227 TURKEY ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 228 TURKEY ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 229 TURKEY ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 230 TURKEY NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 231 TURKEY NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 232 TURKEY NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 233 TURKEY STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 234 TURKEY STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 235 TURKEY STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 236 TURKEY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 237 TURKEY EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 238 TURKEY DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 239 TURKEY CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 240 TURKEY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 241 TURKEY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 242 TURKEY DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 243 BELGIUM DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 244 BELGIUM MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 245 BELGIUM ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 246 BELGIUM ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 247 BELGIUM ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 248 BELGIUM NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 249 BELGIUM NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 250 BELGIUM NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 251 BELGIUM STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 252 BELGIUM STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 253 BELGIUM STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 254 BELGIUM DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 255 BELGIUM EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 256 BELGIUM DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 257 BELGIUM CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 258 BELGIUM DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 259 BELGIUM DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 260 BELGIUM DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 261 NETHERLANDS DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 262 NETHERLANDS MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 263 NETHERLANDS ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 264 NETHERLANDS ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 265 NETHERLANDS ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 266 NETHERLANDS NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 267 NETHERLANDS NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 268 NETHERLANDS NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 269 NETHERLANDS STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 270 NETHERLANDS STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 271 NETHERLANDS STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 272 NETHERLANDS DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 273 NETHERLANDS EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 274 NETHERLANDS DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 275 NETHERLANDS CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 276 NETHERLANDS DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 277 NETHERLANDS DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 278 NETHERLANDS DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 279 SWITZERLAND DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 280 SWITZERLAND MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 281 SWITZERLAND ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 282 SWITZERLAND ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 283 SWITZERLAND ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 284 SWITZERLAND NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 285 SWITZERLAND NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 286 SWITZERLAND NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 287 SWITZERLAND STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 288 SWITZERLAND STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 289 SWITZERLAND STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 290 SWITZERLANDDUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 291 SWITZERLAND EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 292 SWITZERLAND DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 293 SWITZERLAND CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 294 SWITZERLAND DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 295 SWITZERLAND DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 296 SWITZERLAND DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 297 REST OF EUROPE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 298 ASIA-PACIFIC DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY COUNTRY, 2021-2030 (USD THOUSAND)
TABLE 299 ASIA-PACIFIC DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 300 ASIA-PACIFIC MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 301 ASIA-PACIFIC ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 302 ASIA-PACIFIC ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 303 ASIA-PACIFIC NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 304 ASIA-PACIFIC NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 305 ASIA-PACIFIC STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 306 ASIA-PACIFIC STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 307 ASIA-PACIFIC DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 308 ASIA-PACIFIC EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 309 ASIA-PACIFIC DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 310 ASIA-PACIFIC CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 311 ASIA-PACIFIC DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 312 ASIA-PACIFIC DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 313 ASIA-PACIFIC DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 314 CHINA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 315 CHINA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 316 CHINA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 317 CHINA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 318 CHINA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 319 CHINA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 320 CHINA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 321 CHINA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 322 CHINA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 323 CHINA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 324 CHINA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 325 CHINA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 326 CHINA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 327 CHINA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 328 CHINA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 329 CHINA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 330 CHINA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 331 CHINA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 332 JAPAN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 333 JAPAN MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 334 JAPAN ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 335 JAPAN ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 336 JAPAN ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 337 JAPAN NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 338 JAPAN NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 339 JAPAN NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 340 JAPAN STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 341 JAPAN STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 342 JAPAN STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 343 JAPAN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 344 JAPAN EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 345 JAPAN DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 346 JAPAN CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 347 JAPAN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 348 JAPAN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 349 JAPAN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 350 INDIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 351 INDIA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 352 INDIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 353 INDIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 354 INDIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 355 INDIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 356 INDIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 357 INDIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 358 INDIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 359 INDIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 360 INDIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 361 INDIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 362 INDIA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 363 INDIA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 364 INDIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 365 INDIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 366 INDIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 367 SOUTH KOREA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 368 SOUTH KOREA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 369 SOUTH KOREA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 370 SOUTH KOREA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 371 SOUTH KOREA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 372 SOUTH KOREA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 373 SOUTH KOREA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 374 SOUTH KOREA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 375 SOUTH KOREA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 376 SOUTH KOREA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 377 SOUTH KOREA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 378 SOUTH KOREADUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 379 SOUTH KOREA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 380 SOUTH KOREA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 381 SOUTH KOREA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 382 SOUTH KOREA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 383 SOUTH KOREA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 384 SOUTH KOREA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 385 AUSTRALIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 386 AUSTRALIA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 387 AUSTRALIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 388 AUSTRALIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 389 AUSTRALIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 390 AUSTRALIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 391 AUSTRALIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 392 AUSTRALIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 393 AUSTRALIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 394 AUSTRALIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 395 AUSTRALIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 396 AUSTRALIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 397 AUSTRALIA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 398 AUSTRALIA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 399 AUSTRALIA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 400 AUSTRALIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 401 AUSTRALIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 402 AUSTRALIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 403 SINGAPORE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 404 SINGAPORE MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 405 SINGAPORE ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 406 SINGAPORE ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 407 SINGAPORE ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 408 SINGAPORE NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 409 SINGAPORE NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 410 SINGAPORE NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 411 SINGAPORE STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 412 SINGAPORE STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 413 SINGAPORE STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 414 SINGAPORE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 415 SINGAPORE EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 416 SINGAPORE DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 417 SINGAPORE CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 418 SINGAPORE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 419 SINGAPORE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 420 SINGAPORE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 421 THAILAND DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 422 THAILAND MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 423 THAILAND ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 424 THAILAND ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 425 THAILAND ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 426 THAILAND NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 427 THAILAND NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 428 THAILAND NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 429 THAILAND STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 430 THAILAND STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 431 THAILAND STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 432 THAILAND DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 433 THAILAND EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 434 THAILAND DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 435 THAILAND CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 436 THAILAND DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 437 THAILAND DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 438 THAILAND DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 439 MALAYSIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 440 MALAYSIA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 441 MALAYSIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 442 MALAYSIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 443 MALAYSIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 444 MALAYSIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 445 MALAYSIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 446 MALAYSIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 447 MALAYSIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 448 MALAYSIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 449 MALAYSIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 450 MALAYSIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 451 MALAYSIA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 452 MALAYSIA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 453 MALAYSIA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 454 MALAYSIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 455 MALAYSIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 456 MALAYSIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 457 INDONESIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 458 INDONESIA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 459 INDONESIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 460 INDONESIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 461 INDONESIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 462 INDONESIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 463 INDONESIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 464 INDONESIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 465 INDONESIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 466 INDONESIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 467 INDONESIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 468 INDONESIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 469 INDONESIA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 470 INDONESIA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 471 INDONESIA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 472 INDONESIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 473 INDONESIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 474 INDONESIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 475 PHILIPPINES DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 476 PHILIPPINES MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 477 PHILIPPINES ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 478 PHILIPPINES ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 479 PHILIPPINES ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 480 PHILIPPINES NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 481 PHILIPPINES NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 482 PHILIPPINES NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 483 PHILIPPINES STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 484 PHILIPPINES STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 485 PHILIPPINES STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 486 PHILIPPINESDUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 487 PHILIPPINES EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 488 PHILIPPINES DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 489 PHILIPPINES CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 490 PHILIPPINES DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 491 PHILIPPINES DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 492 PHILIPPINES DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 493 REST OF ASIA-PACIFIC DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 494 SOUTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY COUNTRY, 2021-2030 (USD THOUSAND)
TABLE 495 SOUTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 496 SOUTH AMERICA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 497 SOUTH AMERICA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 498 SOUTH AMERICA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 499 SOUTH AMERICA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 500 SOUTH AMERICA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 501 SOUTH AMERICA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 502 SOUTH AMERICA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 503 SOUTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 504 SOUTH AMERICA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 505 SOUTH AMERICA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 506 SOUTH AMERICA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 507 SOUTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 508 SOUTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 509 SOUTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 510 BRAZIL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 511 BRAZIL MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 512 BRAZIL ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 513 BRAZIL ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 514 BRAZIL ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 515 BRAZIL NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 516 BRAZIL NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 517 BRAZIL NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 518 BRAZIL STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 519 BRAZIL STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 520 BRAZIL STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 521 BRAZIL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 522 BRAZIL EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 523 BRAZIL DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 524 BRAZIL CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 525 BRAZIL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 526 BRAZIL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 527 BRAZIL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 528 ARGENTINA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 529 ARGENTINA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 530 ARGENTINA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 531 ARGENTINA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 532 ARGENTINA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 533 ARGENTINA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 534 ARGENTINA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 535 ARGENTINA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 536 ARGENTINA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 537 ARGENTINA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 538 ARGENTINA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 539 ARGENTINA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 540 ARGENTINA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 541 ARGENTINA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 542 ARGENTINA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 543 ARGENTINA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 544 ARGENTINA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 545 ARGENTINA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 546 REST OF SOUTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 547 MIDDLE EAST AND AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY COUNTRY, 2021-2030 (USD THOUSAND)
TABLE 548 MIDDLE EAST AND AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 549 MIDDLE EAST AND AFRICA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 550 MIDDLE EAST AND AFRICA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 551 MIDDLE EAST AND AFRICA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 552 MIDDLE EAST AND AFRICA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 553 MIDDLE EAST AND AFRICA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 554 MIDDLE EAST AND AFRICA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 555 MIDDLE EAST AND AFRICA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 556 MIDDLE EAST AND AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 557 MIDDLE EAST AND AFRICA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 558 MIDDLE EAST AND AFRICA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 559 MIDDLE EAST AND AFRICA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 560 MIDDLE EAST AND AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 561 MIDDLE EAST AND AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 562 MIDDLE EAST AND AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 563 SOUTH AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 564 SOUTH AFRICA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 565 SOUTH AFRICA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 566 SOUTH AFRICA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 567 SOUTH AFRICA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 568 SOUTH AFRICA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 569 SOUTH AFRICA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 570 SOUTH AFRICA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 571 SOUTH AFRICA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 572 SOUTH AFRICA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 573 SOUTH AFRICA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 574 SOUTH AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 575 SOUTH AFRICA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 576 SOUTH AFRICA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 577 SOUTH AFRICA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 578 SOUTH AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 579 SOUTH AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 580 SOUTH AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 581 SAUDI ARABIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 582 SAUDI ARABIA MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 583 SAUDI ARABIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 584 SAUDI ARABIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 585 SAUDI ARABIA ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 586 SAUDI ARABIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 587 SAUDI ARABIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 588 SAUDI ARABIA NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 589 SAUDI ARABIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 590 SAUDI ARABIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 591 SAUDI ARABIA STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 592 SAUDI ARABIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 593 SAUDI ARABIA EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 594 SAUDI ARABIA DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 595 SAUDI ARABIA CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 596 SAUDI ARABIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 597 SAUDI ARABIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 598 SAUDI ARABIA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 599 U.A.E DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 600 U.A.E MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 601 U.A.E ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 602 U.A.E ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 603 U.A.E ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 604 U.A.E NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 605 U.A.E NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 606 U.A.E NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 607 U.A.E STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 608 U.A.E STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 609 U.A.E STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 610 U.A.E DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 611 U.A.E EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 612 U.A.E DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 613 U.A.E CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 614 U.A.E DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 615 U.A.E DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 616 U.A.E DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 617 EGYPT DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 618 EGYPT MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 619 EGYPT ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 620 EGYPT ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 621 EGYPT ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 622 EGYPT NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 623 EGYPT NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 624 EGYPT NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 625 EGYPT STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 626 EGYPT STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 627 EGYPT STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 628 EGYPT DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 629 EGYPT EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 630 EGYPT DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 631 EGYPT CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 632 EGYPT DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 633 EGYPT DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 634 EGYPT DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 635 ISRAEL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 636 ISRAEL MOLECULAR-BASED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 637 ISRAEL ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 638 ISRAEL ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 639 ISRAEL ANTISENSE OLIGONUCLEOTIDE THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 640 ISRAEL NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 641 ISRAEL NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 642 ISRAEL NONSENSE MUTATION IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 643 ISRAEL STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
TABLE 644 ISRAEL STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (VOLUME)
TABLE 645 ISRAEL STEROID THERAPY IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (ASP)
TABLE 646 ISRAEL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 647 ISRAEL EXON SKIPPING APPROACH IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 648 ISRAEL DYSTROPHIN-TARGETED THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 649 ISRAEL CELL THERAPIES IN DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY THERAPY, 2021-2030 (USD THOUSAND)
TABLE 650 ISRAEL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD THOUSAND)
TABLE 651 ISRAEL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY END USER, 2021-2030 (USD THOUSAND)
TABLE 652 ISRAEL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD THOUSAND)
TABLE 653 REST OF MIDDLE EAST AND AFRICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET, BY TREATMENT TYPE, 2021-2030 (USD THOUSAND)
Abbildungsverzeichnis
FIGURE 1 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: SEGMENTATION
FIGURE 2 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: DATA TRIANGULATION
FIGURE 3 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: DROC ANALYSIS
FIGURE 4 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: GLOBAL VS REGIONAL MARKET ANALYSIS
FIGURE 5 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: MARKET END USER COVERAGE GRID
FIGURE 8 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: DBMR MARKET POSITION GRID
FIGURE 9 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: VENDOR SHARE ANALYSIS
FIGURE 10 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: SEGMENTATION
FIGURE 11 THE RISING PREVALENCE AND IMPACT OF DUCHENNE MUSCULAR DYSTROPHY (DMD) AND INTRODUCTION OF NOVEL THERAPIES FOR DMD DISORDER ARE EXPECTED TO DRIVE THE GROWTH OF THE GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET FROM 2023 TO 2030
FIGURE 12 MOLECULAR-BASED THERAPIES SEGMENT IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET IN 2023 AND 2030
FIGURE 13 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF THE GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET
FIGURE 14 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY TREATMENT TYPE , 2022
FIGURE 15 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY TREATMENT TYPE, 2023-2030 (USD THOUSAND)
FIGURE 16 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY TREATMENT TYPE , CAGR (2023-2030)
FIGURE 17 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY TREATMENT TYPE, LIFELINE CURVE
FIGURE 18 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY THERAPY, 2022
FIGURE 19 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY THERAPY, 2023-2030 (USD THOUSAND)
FIGURE 20 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY THERAPY, CAGR (2023-2030)
FIGURE 21 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY THERAPY, LIFELINE CURVE
FIGURE 22 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY ROUTE OF ADMINISTRATION, 2022
FIGURE 23 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY ROUTE OF ADMINISTRATION, 2023-2030 (USD THOUSAND)
FIGURE 24 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY ROUTE OF ADMINISTRATION, CAGR (2023-2030)
FIGURE 25 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY ROUTE OF ADMINISTRATION, LIFELINE CURVE
FIGURE 26 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY END USER, 2022
FIGURE 27 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY END USER, 2023-2030 (USD THOUSAND)
FIGURE 28 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY END USER, CAGR (2023-2030)
FIGURE 29 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY END USER, LIFELINE CURVE
FIGURE 30 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY DISTRIBUTION CHANNEL, 2022
FIGURE 31 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY DISTRIBUTION CHANNEL, 2023-2030 (USD THOUSAND)
FIGURE 32 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY DISTRIBUTION CHANNEL, CAGR (2023-2030)
FIGURE 33 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 34 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET SNAPSHOT
FIGURE 35 GLOBAL DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: COMPANY SHARE 2022 (%)
FIGURE 36 NORTH AMERICA DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: COMPANY SHARE 2022 (%)
FIGURE 37 EUROPE DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: COMPANY SHARE 2022 (%)
FIGURE 38 ASIA-PACIFIC DUCHENNE MUSCULAR DYSTROPHY TREATMENT MARKET: COMPANY SHARE 2022 (%)

Forschungsmethodik
Die Datenerfassung und Basisjahresanalyse werden mithilfe von Datenerfassungsmodulen mit großen Stichprobengrößen durchgeführt. Die Phase umfasst das Erhalten von Marktinformationen oder verwandten Daten aus verschiedenen Quellen und Strategien. Sie umfasst die Prüfung und Planung aller aus der Vergangenheit im Voraus erfassten Daten. Sie umfasst auch die Prüfung von Informationsinkonsistenzen, die in verschiedenen Informationsquellen auftreten. Die Marktdaten werden mithilfe von marktstatistischen und kohärenten Modellen analysiert und geschätzt. Darüber hinaus sind Marktanteilsanalyse und Schlüsseltrendanalyse die wichtigsten Erfolgsfaktoren im Marktbericht. Um mehr zu erfahren, fordern Sie bitte einen Analystenanruf an oder geben Sie Ihre Anfrage ein.
Die wichtigste Forschungsmethodik, die vom DBMR-Forschungsteam verwendet wird, ist die Datentriangulation, die Data Mining, die Analyse der Auswirkungen von Datenvariablen auf den Markt und die primäre (Branchenexperten-)Validierung umfasst. Zu den Datenmodellen gehören ein Lieferantenpositionierungsraster, eine Marktzeitlinienanalyse, ein Marktüberblick und -leitfaden, ein Firmenpositionierungsraster, eine Patentanalyse, eine Preisanalyse, eine Firmenmarktanteilsanalyse, Messstandards, eine globale versus eine regionale und Lieferantenanteilsanalyse. Um mehr über die Forschungsmethodik zu erfahren, senden Sie eine Anfrage an unsere Branchenexperten.
Anpassung möglich
Data Bridge Market Research ist ein führendes Unternehmen in der fortgeschrittenen formativen Forschung. Wir sind stolz darauf, unseren bestehenden und neuen Kunden Daten und Analysen zu bieten, die zu ihren Zielen passen. Der Bericht kann angepasst werden, um Preistrendanalysen von Zielmarken, Marktverständnis für zusätzliche Länder (fordern Sie die Länderliste an), Daten zu klinischen Studienergebnissen, Literaturübersicht, Analysen des Marktes für aufgearbeitete Produkte und Produktbasis einzuschließen. Marktanalysen von Zielkonkurrenten können von technologiebasierten Analysen bis hin zu Marktportfoliostrategien analysiert werden. Wir können so viele Wettbewerber hinzufügen, wie Sie Daten in dem von Ihnen gewünschten Format und Datenstil benötigen. Unser Analystenteam kann Ihnen auch Daten in groben Excel-Rohdateien und Pivot-Tabellen (Fact Book) bereitstellen oder Sie bei der Erstellung von Präsentationen aus den im Bericht verfügbaren Datensätzen unterstützen.