글로벌 외분비 췌장 기능 부전(EPI) 치료 및 진단 시장, 진단(영상 검사 및 췌장 기능 검사), 치료(영양 관리, 췌장 효소 대체 요법 (PERT), 약물 유형(제네릭 및 브랜드), 최종 사용자(병원, 전문 클리닉, 재택 치료, 진단 센터, 연구 및 학술 기관 및 기타), 유통 채널(직접 입찰, 소매 약국, 제3자 유통업체 및 기타) - 업계 동향 및 2030년까지의 예측.
외분비 췌장 부전(EPI) 치료제 및 진단 시장 분석 및 통찰력
전 세계적으로 고급 진단 기술에 대한 인식이 증가함에 따라 시장 수요가 증가했습니다. 더 나은 건강 서비스를 위한 의료비 지출 증가도 시장 성장에 기여합니다. 주요 시장 참여자는 이 중요한 기간 동안 다양한 서비스 출시와 승인에 집중합니다. 또한 프로세스와 기술의 향상된 발전 증가도 외분비 췌장 부전(EPI) 치료제 및 진단에 대한 수요 증가에 기여합니다.
글로벌 외분비 췌장 부전(EPI) 치료 및 진단 시장은 시장 참여자의 증가와 고급 서비스의 가용성으로 인해 예측 연도에 성장할 것으로 예상됩니다. 이와 함께 제조업체는 시장에서 새로운 서비스를 출시하기 위한 개발 활동에 참여하고 있습니다. 고급 의료 기술의 개발이 증가함에 따라 시장 성장이 더욱 촉진되고 있습니다. 그러나 외분비 췌장 부전(EPI) 치료 및 진단 분야를 방해하는 엄격한 규정, 췌장 효소 대체 요법(PERT)과 관련된 부작용 및 종교적 장애물과 같은 어려움이 예측 기간 동안 글로벌 외분비 췌장 부전(EPI) 치료 및 진단 시장의 성장을 방해할 수 있습니다.
진보와 개발에 대한 의료 지출 증가는 시장에 기회를 제공할 것으로 예상됩니다. 그러나 EPI 진단 및 치료와 관련된 높은 비용은 시장 성장에 도전이 될 수 있습니다.
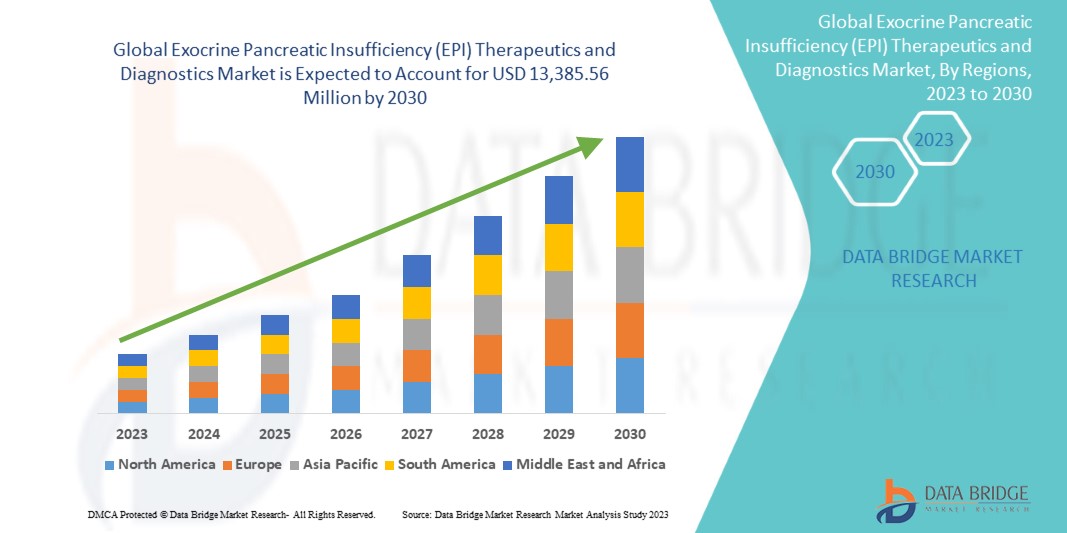
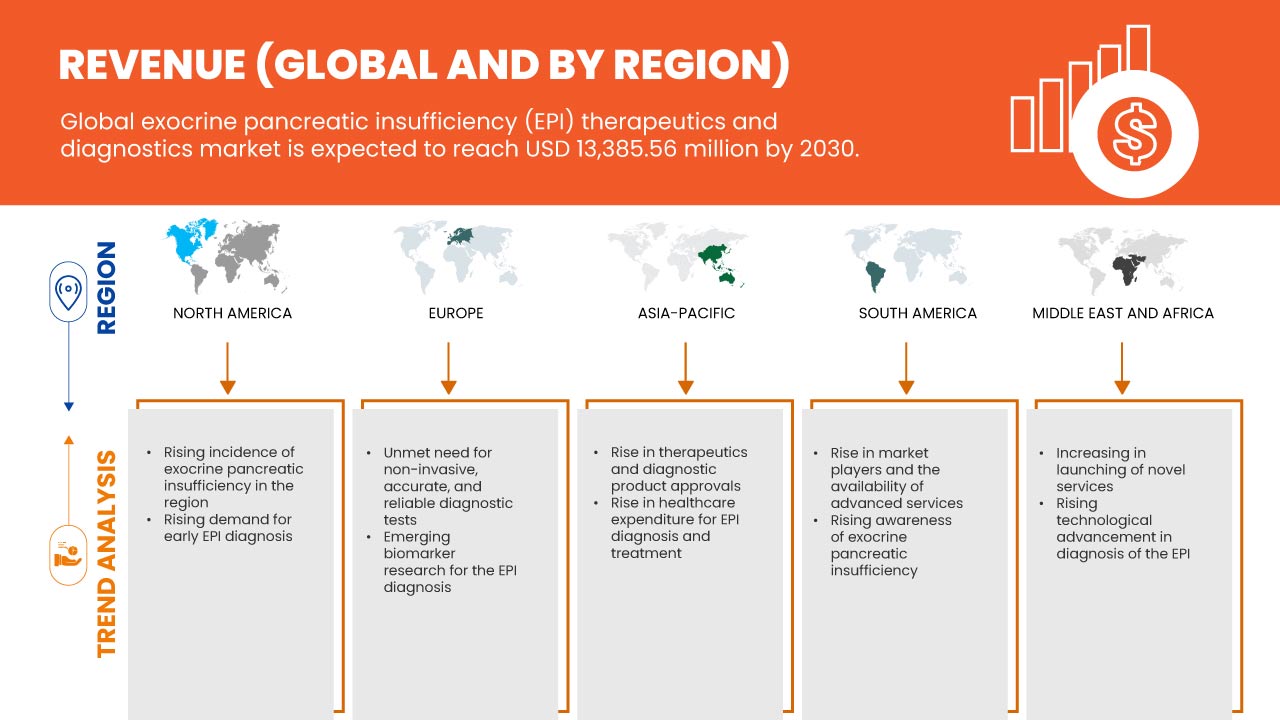
Data Bridge Market Research에 따르면, 글로벌 외분비 췌장 기능 부전(EPI) 치료 및 진단 시장은 2030년까지 13,385.56백만 달러 규모에 도달할 것으로 예상되며, 이는 예측 기간 동안 7.2%의 CAGR로 성장할 것으로 예상됩니다.
|
보고서 메트릭 |
세부 |
|
예측 기간 |
2023년부터 2030년까지 |
|
기준 연도 |
2022 |
|
역사적 연도 |
2021 (2015-2020까지 사용자 정의 가능) |
|
양적 단위 |
매출은 백만, 볼륨은 단위, 가격은 USD로 표시 |
|
다루는 세그먼트 |
진단(영상 검사 및 췌장 기능 검사), 치료(영양 관리, 췌장 효소 대체 요법(PERT), 약물 유형(제네릭 및 브랜드), 최종 사용자(병원, 전문 클리닉, 홈 케어, 진단 센터, 연구 및 학술 기관 및 기타), 유통 채널(직접 입찰, 소매 약국, 제3자 유통업체 및 기타) |
|
적용 국가 |
U.S., Canada, Mexico, Germany, France, U.K, Italy, Spain, Netherlands, Russia, Switzerland, Turkey, Belgium, Denmark, Finland, Norway, Sweden, Poland, and Rest of Europe, China, Japan, India, South Korea, Australia, New Zealand, Singapore, Thailand, Malaysia, Indonesia, Philippines, Vietnam, Taiwan, and Rest of Asia-Pacific, Brazil, Argentina, and Rest of South America, South Africa, Saudi Arabia, Bahrain, Kuwait, Qatar, Oman, U.A.E., Egypt, Israel, and Rest of Middle East and Africa |
|
Market Players Covered |
EagleBio, AbbVie., Nordmark Arzneimittel GmbH & Co., Digestive Care, Inc., Cilian AG, Alcresta Therapeutics, Inc., ChiRhoClin, Abbott, Bioserv Diagnostics, Laboratory Corporation of America, Organon group of companies Metagenics LLC, Janssen, Nestlé., VIVUS LLC., and ScheBo Biotech AG, among others |
Market Definition
Exocrine pancreatic insufficiency (EPI) is a medical condition where the pancreas fails to produce enough digestive enzymes to digest fats, proteins, and carbohydrates in the intestine. EPI can be caused by various conditions, such as chronic pancreatitis, cystic fibrosis, and pancreatic cancer. Therapeutics for EPI involve using pancreatic enzyme replacement therapy (PERT), which provides the necessary digestive enzymes that the pancreas is not producing. PERT is usually taken with meals and snacks to help break down food and absorb nutrients properly; several PERT products are available, such as pancrelipase, pancreatin, and lipase.
Diagnostics for EPI may involve blood tests to measure the levels of digestive enzymes in the blood, fecal elastase test to check the level of pancreatic elastase in the stool, and imaging tests, such as CT scans, MRI, or endoscopic ultrasound to visualize the pancreas and detect any abnormalities. Additionally, breath tests can measure the levels of hydrogen and methane in the breath, indicating EPI.
Global Exocrine Pancreatic Insufficiency (EPI) Therapeutics and Diagnostics Market Dynamics
This section deals with understanding the market drivers, opportunities, restraints, and challenges. All of this is discussed in detail below:
Drivers
- Increasing prevalence of exocrine pancreatic insufficiency (EPI) due to chronic pancreatitis and cystic fibrosis
Exocrine pancreatic insufficiency (EPI) is a condition caused when the pancreas fails to make sufficient amounts of digestive enzymes to help digestion and absorption of nutrients. Chronic pancreatitis is the main cause of EPI in adults. Pancreatitis causes inflammation and swelling of the pancreas. Over time, it can damage the pancreatic cells that make digestive enzymes. Similarly, cystic fibrosis is one of the main causes of EPI in children. It is inherited from parents and causes thick mucus to build up in the lungs, creating difficulty breathing. Mucus also accumulates in the pancreas and prevents digestive enzymes from reaching the small intestine. People with EPI can’t absorb enough fats, proteins, and carbohydrates from foods which is called malabsorption.
Therefore, the increasing prevalence of EPI due to chronic pancreatitis and cystic fibrosis is driving the growth of the market in the forecast period.
- Rising technological advancements in the diagnosis and treatment of EPI
There is a lack of consensus regarding the best diagnostic approach, and experts have noted the non-reliability and non-specificity of available diagnostic tests. Available diagnostic tests for EPI include fecal fat quantification, the fecal elastase-1 test, and the C-mixed triglyceride breath test. Developing a machine learning model that identifies patients in a commercial medical claims database who likely have EPI but are undiagnosed is one of the recent objectives to advance the diagnosis and treatment framework of EPI.
Therefore, the rising technological advancements in the diagnosis and treatment of EPI is driving the market's growth in the forecast period.
Restraint
- Stringent regulations to hamper the exocrine pancreatic insufficiency (EPI) therapeutic and diagnostic sector
The use of therapies and medications for EPI disorders across the globe is rapidly increasing, with the growth of the aged population and several chronic diseases which are preventable by early diagnosis. At the same time, the players of the EPI disorders products manufacturers in the market must follow certain regulations to get approval from the upper authorities for launching the product in the market. These stringent guidelines need to be followed and this is one of the most difficult tasks of all the steps. The U.S. Food and Drug Administration (FDA) regulates the U.S. and the European Union (EU) regulates Europe. However, the rapid development of privacy policies and regulations is being made in the Asia-Pacific and EMEA, including India, Russia, China, South Korea, Singapore, Hong Kong, and Australia. Some drug regulatory agencies, including the U.S. Food and Drug Administration (FDA), have instructed that manufacturers obtain regulatory approvals before marketing their products because of possible adverse effects and inadequate effectiveness. These stringent FDA regulations have made approval of pancrelipase products difficult.
Hence, from the above instances, concluded that stringent regulations may restrain the growth of the exocrine therapeutic insufficiency therapeutics and diagnostics market in the forecast period.
Opportunity
-
Increasing prevalence of pancreatic disorders and associated condition
EPI is a prominent side effect of pancreatic diseases like cancer, cystic fibrosis, and chronic pancreatitis. These ailments can harm the pancreas, impairing its capacity to generate digestive enzymes and result in EPI. The rising prevalence of pancreatic diseases and related ailments worldwide is driving the demand for EPI therapies and diagnostics. These conditions can cause damage to the pancreas, leading to a reduced ability to produce digestive enzymes, which can result in EPI. Cystic fibrosis's damage to the pancreas makes it another disorder that can result in EPI. The genetic disease known as cystic fibrosis impacts the reproductive, digestive, and respiratory systems. Cystic fibrosis is more common in Caucasians, particularly those with a Northern European ancestry, than worldwide. As the incidence of these conditions continues to increase, the demand for EPI therapeutics and diagnostics is also expected to grow. This presents a significant opportunity for pharmaceutical companies and manufacturers to develop new and effective treatments and diagnostic tests for EPI.
Thus, the increasing prevalence of pancreatic disorders and associated conditions is expected to act as an opportunity for market growth.
Challenge
- High cost associated with EPI diagnosis and treatment
The complexity of the illness and the requirement for specialized tests make the diagnosis of EPI expensive. Faecal elastase testing, secretin-stimulated pancreatic function testing, and imaging tests like magnetic resonance imaging (MRI) and endoscopic ultrasonography (EUS) are the most frequently used diagnostic procedures for EPI. These tests can be pricey, and not all medical professionals can access the tools and training needed to carry them out. The high expense of identifying EPI can also delay a diagnosis, which can exacerbate symptoms and raise healthcare costs over time. Untreated EPI can cause continuing digestive problems, nutrient malabsorption, and weight loss in patients, all of which can increase medical expenses and difficulties.
Thus, the high cost associated with EPI diagnosis and treatment is expected to act as a challenge in the market growth.
Recent Developments
- In August 2021, AzurRx announced that it had engaged in the development of yeast-derived lipase, MS1819, which has been engineered to have superior enzymatic activity as compared to current treatments.
- In February 2023, Codexis, Inc. and Nestlé Health Science announced the interim findings from a Phase 1 trial examining the safety, tolerability, pharmacokinetics (PK), and pharmacodynamics of CDX-7108. A lipase variation called CDX-7108 was created expressly to get around the drawbacks of the current pancreatic enzyme replacement treatment (PERT). This has aided the business in marketing the product.
Global Exocrine Pancreatic Insufficiency (EPI) Therapeutics and Diagnostics Market Scope
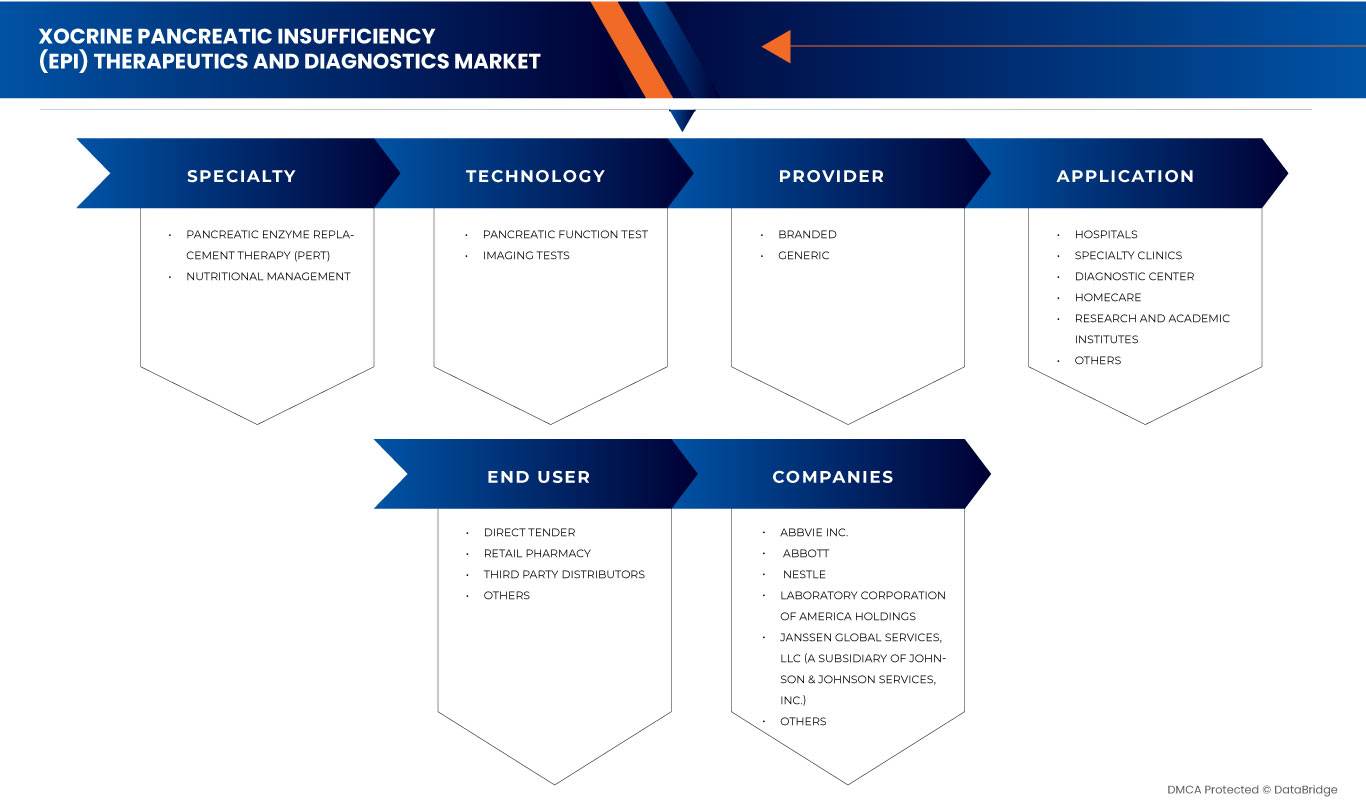
The global exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market is categorized into five notable segments such as diagnosis, treatment, drug type, end user, and distribution channel. The growth among segments helps you analyze niche pockets of growth and strategies to approach the market and determine your core application areas and the difference in your target markets.
Diagnosis
- Imaging Tests
- Pancreatic Function Test
On the basis of diagnosis, the global exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market is segmented into imaging test and pancreatic function test.
Treatment
- Nutritional Management
- Pancreatic Enzyme Replacement Therapy (PERT)
On the basis of treatment, the global exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market is segmented into nutritional management and pancreatic enzyme replacement therapy (PERT).
Drug Type
- Generic
- Branded
On the basis of drug type, the global exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market is segmented into generic and branded.
End User
- Hospitals
- Specialty Clinics
- Homecare
- Diagnostic Centre
- Research and Academic Institutes
- Others
On the basis of end user, the global exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market is segmented into Hospitals, Specialty Clinics, Homecare, Diagnostic Centers, Research and Academic Institute, and others.
Distribution Channel
- Direct Tender
- Retail Pharmacy
- Third-Party Distributor
- Others
On the basis of distribution channel, the global exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market is segmented into direct tender, retail pharmacy, third party distributor, and others.
Global Exocrine Pancreatic Insufficiency (EPI) Therapeutics and Diagnostics Market Regional Analysis/Insights
The global exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market is categorized into five notable segments such as diagnosis, treatment, drug type, end user, and distribution channel.
The countries covered in this market report U.S., Canada, Mexico, Germany, France, U.K, Italy, Spain, Netherlands, Russia, Switzerland, Turkey, Belgium, Denmark, Finland, Norway, Sweden, Poland, and Rest of Europe, China, Japan, India, South Korea, Australia, New Zealand, Singapore, Thailand, Malaysia, Indonesia, Philippines, Vietnam, Taiwan, and Rest of Asia-Pacific, Brazil, Argentina, and Rest of South America, South Africa, Saudi Arabia, Bahrain, Kuwait, Qatar, Oman, U.A.E., Egypt, Israel, and Rest of Middle East and Africa.
North America is dominating due to the presence of key market players in the largest consumer market with high GDP. The U.S is expected to grow due to the rise in technological advancement in the market.
North America is dominating the market due to the increasing investment in healthcare is expected to boost the market growth. The U.S. dominates the North America region due to the strong presence of key players. Germany dominates Europe due to the increasing demand from emerging markets and expansion. Japan dominates the Asia-Pacific region due to increasing customer inclinations towards advanced technological processes.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impact the current and future trends of the market. Data points such as new sales, replacement sales, country demographics, regulatory acts, and import-export tariffs are some of the major pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, and impact of sales channels are considered while providing forecast analysis of the country data.
Competitive Landscape and Global Exocrine Pancreatic Insufficiency (EPI) Therapeutics and Diagnostics Market Share Analysis
Global exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market competitive landscape provide details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in R&D, new market initiatives, production sites and facilities, company strengths and weaknesses, product launch, product approvals, product width and breath, application dominance, and product type lifeline curve. The above data points provided are only related to the company’s focus on the global nurse call systems market.
Some of the major players operating in the global exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market are EagleBio, AbbVie., Nordmark Arzneimittel GmbH & Co., Digestive Care, Inc., Cilian AG, Alcresta Therapeutics, Inc., ChiRhoClin, Abbott, Bioserv Diagnostics, Laboratory Corporation of America, Organon group of companies Metagenics LLC, Janssen, Nestlé., VIVUS LLC., and ScheBo Biotech AG among others.
SKU-
세계 최초의 시장 정보 클라우드 보고서에 온라인으로 접속하세요
- 대화형 데이터 분석 대시보드
- 높은 성장 잠재력 기회를 위한 회사 분석 대시보드
- 사용자 정의 및 질의를 위한 리서치 분석가 액세스
- 대화형 대시보드를 통한 경쟁자 분석
- 최신 뉴스, 업데이트 및 추세 분석
- 포괄적인 경쟁자 추적을 위한 벤치마크 분석의 힘 활용
목차
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET
1.4 LIMITATIONS
1.5 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 CURRENCY AND PRICING
2.5 DBMR TRIPOD DATA VALIDATION MODEL
2.6 MULTIVARIATE MODELLING
2.7 PRODUCT TYPE LIFELINE CURVE
2.8 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.9 DBMR MARKET POSITION GRID
2.1 MARKET TESTING TYPE COVERAGE GRID
2.11 VENDOR SHARE ANALYSIS
2.12 SECONDARY SOURCES
2.13 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PESTEL ANALYSIS
4.2 PORTER ANALYSIS
5 INDUSTRY INSIGHTS
6 REGULATORY FRAMEWORK
7 MARKET OVERVIEW
7.1 DRIVERS
7.1.1 INCREASING PREVALENCE OF EXOCRINE PANCREATIC INSUFFICIENCY (EPI) DUE TO CHRONIC PANCREATITIS AND CYSTIC FIBROSIS
7.1.2 RISING TECHNOLOGICAL ADVANCEMENTS IN DIAGNOSIS AND TREATMENT OF EPI
7.1.3 STRATEGIC INITIATIVES BY THE KEY MARKET PLAYERS
7.1.4 SEDENTARY LIFESTYLES AND UNHEALTHY DIETS
7.2 RESTRAINTS
7.2.1 STRINGENT REGULATIONS TO HAMPER THE EXOCRINE PANCREATIC INSUFFICIENCY(EPI) THERAPEUTIC AND DIAGNOSTIC SECTOR
7.2.2 SIDE EFFECTS AND RELIGIOUS HURDLES ASSOCIATED WITH PANCREATIC ENZYME REPLACEMENT THERAPY (PERT)
7.3 OPPORTUNITIES
7.3.1 INCREASING PREVALENCE OF PANCREATIC DISORDERS AND ASSOCIATED CONDITION
7.3.2 RISING AWARENESS ABOUT THE BENEFITS OF EARLY DIAGNOSIS AND TREATMENT OF EPI
7.3.3 GROWING DEMAND FOR COMBINATION THERAPIES AND PERSONALIZED MEDICINE
7.4 CHALLENGES
7.4.1 HIGH COST ASSOCIATED WITH EPI DIAGNOSIS AND TREATMENT
7.4.2 LIMITED AVAILABILITY OF EPI THERAPEUTICS AND DIAGNOSTICS
8 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY TREATMENT
8.1 OVERVIEW
8.2 PANCREATIC ENZYME REPLACEMENT THERAPY (PERT)
8.2.1 BY ENZYME
8.2.1.1 LIPASE
8.2.1.2 PROTEASE
8.2.1.3 AMYLASE
8.2.2 BY ROUTE OF ADMINISTRATION
8.2.2.1 ORAL
8.2.2.1.1 CAPSULES
8.2.2.1.2 TABLETS
8.2.2.1.3 POWDER
8.2.2.2 INTRAVENOUS
8.3 NUTRITIONAL MANAGEMENT
8.3.1 VITAMIN D
8.3.2 VITAMIN E
8.3.3 VITAMIN A
8.3.4 VITAMIN K
8.3.5 OTHERS
9 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY DIAGNOSIS
9.1 OVERVIEW
9.2 PANCREATIC FUNCTION TEST
9.2.1 STOOL
9.2.1.1 FECAL ELASTASE TEST
9.2.1.2 FECAL FAT TEST
9.2.2 SECRETIN PANCREATIC FUNCTION TEST
9.2.3 BLOOD TESTS
9.2.4 C-MIXED TRIGLYCERIDES BREATH TEST
9.2.5 FASTING PLASMA GLUCOSE TEST
9.3 IMAGING TEST
9.3.1 CT SCANNING
9.3.2 MAGNETIC RESONANCE IMAGING TESTS (MRI)
9.3.3 ENDOSCOPIC RETROGRADE PANCREATOGRAPHY
9.3.4 ENDOSCOPIC ULTRA-SONOGRAPHY (EUS)
9.3.5 SECRETIN-STIMULATED MAGNETIC RESONANCE CHOLANGIOPANCREATOGRAPHY
10 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY DRUG TYPE
10.1 OVERVIEW
10.2 BRANDED
10.2.1 CREON
10.2.2 ZENPEP
10.2.3 PANCREAZE
10.2.4 NUTRIZYM
10.2.5 PANCREASE MT
10.2.6 PANCREX
10.2.7 COTAZYM
10.2.8 ULTERSA
10.2.9 VIOKACE
10.2.10 PERTZYE
10.2.11 OTHERS
10.3 GENERIC
11 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY END USER
11.1 OVERVIEW
11.2 HOSPITALS
11.2.1 PRIVATE
11.2.2 PUBLIC
11.3 SPECIALTY CLINICS
11.4 DIAGNOSTIC CENTER
11.5 HOMECARE
11.6 RESEARCH AND ACADEMIC INSTITUTES
11.7 OTHERS
12 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY DISTRIBUTION CHANNEL
12.1 OVERVIEW
12.2 DIRECT TENDER
12.3 RETAIL PHARMACY
12.4 THIRD PARTY DISTRIBUTORS
12.5 OTHERS
13 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION
13.1 OVERVIEW
13.2 NORTH AMERICA
13.2.1 U.S.
13.2.2 CANADA
13.2.3 MEXICO
13.3 EUROPE
13.3.1 GERMANY
13.3.2 FRANCE
13.3.3 U.K.
13.3.4 ITALY
13.3.5 SPAIN
13.3.6 RUSSIA
13.3.7 TURKEY
13.3.8 BELGIUM
13.3.9 DENMARK
13.3.10 NETHERLANDS
13.3.11 SWITZERLAND
13.3.12 SWEDEN
13.3.13 POLAND
13.3.14 NORWAY
13.3.15 FINLAND
13.3.16 REST OF EUROPE
13.4 ASIA PACIFIC
13.4.1 JAPAN
13.4.2 CHINA
13.4.3 SOUTH KOREA
13.4.4 INDIA
13.4.5 AUSTRALIA
13.4.6 NEW ZEALAND
13.4.7 SINGAPORE
13.4.8 THAILAND
13.4.9 MALAYSIA
13.4.10 VIETNAM
13.4.11 TAIWAN
13.4.12 INDONESIA
13.4.13 PHILIPPINES
13.4.14 REST OF ASIA-PACIFIC
13.5 SOUTH AMERICA
13.5.1 BRAZIL
13.5.2 ARGENTINA
13.5.3 REST OF SOUTH AMERICA
13.6 MIDDLE EAST AND AFRICA
13.6.1 SOUTH AFRICA
13.6.2 SAUDI ARABIA
13.6.3 BAHRAIN
13.6.4 U.A.E.
13.6.5 KUWAIT
13.6.6 OMAN
13.6.7 QATAR
13.6.8 EGYPT
13.6.9 ISRAEL
13.6.10 REST OF MIDDLE EAST AND AFRICA
14 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: COMPANY LANDSCAPE
14.1 COMPANY SHARE ANALYSIS: GLOBAL
14.2 COMPANY SHARE ANALYSIS: NORTH AMERICA
14.3 COMPANY SHARE ANALYSIS: EUROPE
14.4 COMPANY SHARE ANALYSIS: ASIA-PACIFIC
15 SWOT ANALYSIS
16 COMPANY PROFILE
16.1 ABBVIE INC.
16.1.1 COMPANY SNAPSHOT
16.1.2 REVENUE ANALYSIS
16.1.3 COMPANY SHARE ANALYSIS
16.1.4 PRODUCT PORTFOLIO
16.1.5 RECENT DEVELOPMENT
16.2 ABBOTT
16.2.1 COMPANY SNAPSHOT
16.2.2 REVENUE ANALYSIS
16.2.3 COMPANY SHARE ANALYSIS
16.2.4 PRODUCT PORTFOLIO
16.2.5 RECENT DEVELOPMENT
16.3 NESTLE
16.3.1 COMPANY SNAPSHOT
16.3.2 REVENUE ANALYSIS
16.3.3 COMPANY SHARE ANALYSIS
16.3.4 PRODUCT PORTFOLIO
16.3.5 RECENT DEVELOPMENTS
16.4 LABORATORY CORPORATION OF AMERICA HOLDINGS (2022)
16.4.1 COMPANY SNAPSHOT
16.4.2 REVENUE ANALYSIS
16.4.3 COMPANY SHARE ANALYSIS
16.4.4 PRODUCT PORTFOLIO
16.4.5 RECENT DEVELOPMENT
16.5 JANSSEN GLOBAL SERVICES, LLC
16.5.1 COMPANY SNAPSHOT
16.5.2 REVENUE ANALYSIS
16.5.3 COMPANY SHARE ANALYSIS
16.5.4 PRODUCT PORTFOLIO
16.5.5 RECENT DEVELOPMENT
16.6 ALCRESTA THERAPEUTICS, INC.
16.6.1 COMPANY SNAPSHOT
16.6.2 COMPANY SHARE ANALYSIS
16.6.3 PRODUCT PORTFOLIO
16.6.4 RECENT DEVELOPMENT
16.7 BIOSERV DIAGNOSTICS GMBH
16.7.1 COMPANY SNAPSHOT
16.7.2 PRODUCT PORTFOLIO
16.7.3 RECENT DEVELOPMENT
16.8 CILIAN AG
16.8.1 COMPANY SNAPSHOT
16.8.2 PRODUCT PORTFOLIO
16.8.3 RECENT DEVELOPMENT
16.9 CHIRHOCLIN
16.9.1 COMPANY SNAPSHOT
16.9.2 COMPANY SHARE ANALYSIS
16.9.3 PRODUCT PORTFOLIO
16.9.4 RECENT DEVELOPMENT
16.1 DIGESTIVE CARE, INC.
16.10.1 COMPANY SNAPSHOT
16.10.2 PRODUCT PORTFOLIO
16.10.3 RECENT DEVELOPMENT
16.11 EAGLEBIO
16.11.1 COMPANY SNAPSHOT
16.11.2 PRODUCT PORTFOLIO
16.11.3 RECENT DEVELOPMENT
16.12 METAGENICS LLC
16.12.1 COMPANY SNAPSHOT
16.12.2 PRODUCT PORTFOLIO
16.12.3 RECENT DEVELOPMENT
16.13 NORDMARK
16.13.1 COMPANY SNAPSHOT
16.13.2 COMPANY SHARE ANALYSIS
16.13.3 PRODUCT PORTFOLIO
16.13.4 RECENT DEVELOPMENT
16.14 ORGANON GROUP OF COMPANIES.
16.14.1 COMPANY SNAPSHOT
16.14.2 REVENUE ANALYSIS
16.14.3 PRODUCT PORTFOLIO
16.14.4 RECENT DEVELOPMENT
16.15 SCHEBO BIOTECH AG
16.15.1 COMPANY SNAPSHOT
16.15.2 PRODUCT PORTFOLIO
16.15.3 RECENT DEVELOPMENT
16.16 VIVUS LLC
16.16.1 COMPANY SNAPSHOT
16.16.2 COMPANY SHARE ANALYSIS
16.16.3 PRODUCT PORTFOLIO
16.16.4 RECENT DEVELOPMENT
17 QUESTIONNAIRE
18 RELATED REPORTS
표 목록
TABLE 1 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY TREATMENT, 2021-2030 (USD MILLION)
TABLE 2 GLOBAL PANCREATIC ENZYME REPLACEMENT THERAPY (PERT) IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 3 GLOBAL PANCREATIC ENZYME REPLACEMENT THERAPY (PERT) IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY ENZYME, 2021-2030 (USD MILLION)
TABLE 4 GLOBAL PANCREATIC ENZYME REPLACEMENT THERAPY (PERT) IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 5 GLOBAL ORAL IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 6 GLOBAL NUTRITIONAL MANAGEMENT IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 7 GLOBAL NUTRITIONAL MANAGEMENT IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY TREATMENT, 2021-2030 (USD MILLION)
TABLE 8 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY DIAGNOSIS, 2021-2030 (USD MILLION)
TABLE 9 GLOBAL PANCREATIC FUNCTION TEST IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 10 GLOBAL PANCREATIC FUNCTION TEST IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY DIAGNOSIS, 2021-2030 (USD MILLION)
TABLE 11 GLOBAL STOOL IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY DIAGNOSIS, 2021-2030 (USD MILLION)
TABLE 12 GLOBAL IMAGING TESTS IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 13 GLOBAL IMAGING TESTS IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY DIAGNOSIS, 2021-2030 (USD MILLION)
TABLE 14 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY DRUG TYPE, 2021-2030 (USD MILLION)
TABLE 15 GLOBAL BRANDED IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 16 GLOBAL BRANDED IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY DRUG TYPE, 2021-2030 (USD MILLION)
TABLE 17 GLOBAL GENERIC IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 18 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 19 GLOBAL HOSPITALS IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 20 GLOBAL HOSPITALS IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 21 GLOBAL SPECIALTY CLINICS IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 22 GLOBAL DIAGNOSTIC CENTER IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 23 GLOBAL HOMECARE IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 24 GLOBAL RESRACH AND ACADEMIC INSTITUTES IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 25 GLOBAL OTHERS IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 26 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 27 GLOBAL DIRECT TENDER IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 28 GLOBAL RETAIL PHARMACY IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 29 GLOBAL THIRD PARTY DISTRIBUTORS IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
TABLE 30 GLOBAL OTHERS IN EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, BY REGION, 2021-2030 (USD MILLION)
그림 목록
FIGURE 1 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: SEGMENTATION
FIGURE 2 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: DATA TRIANGULATION
FIGURE 3 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: DROC ANALYSIS
FIGURE 4 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: GLOBAL VS REGIONAL MARKET ANALYSIS
FIGURE 5 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: DBMR MARKET POSITION GRID
FIGURE 8 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: MARKET TESTING TYPE COVERAGE GRID
FIGURE 9 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: VENDOR SHARE ANALYSIS
FIGURE 10 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: SEGMENTATION
FIGURE 11 INCREASING PREVALENCE OF EXOCRINE PANCREATIC INSUFFICIENCY (EPI) DUE TO CHRONIC PANCREATITIS AND CYSTIC FIBROSIS IS EXPECTED TO DRIVE THE FIGURE 10 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET IN THE FORECAST PERIOD
FIGURE 12 THE PANCREATIC ENZYME REPLACEMENT THERAPY (PERT) SEGMENT IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET IN 2023 & 2030
FIGURE 13 NORTH AMERICA IS EXPECTED TO DOMINATE THE GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET, AND ASIA-PACIFIC IS EXPECTED TO GROW WITH THE HIGHEST CAGR IN THE FORECAST PERIOD
FIGURE 14 ASIA-PACIFIC IS THE FASTEST-GROWING MARKET FOR GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET MANUFACTURERS IN THE FORECAST PERIOD
FIGURE 15 DRIVERS, RESTRAINTS, OPPORTUNITIES AND CHALLENGES OF THE GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET
FIGURE 16 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY TREATMENT, 2022
FIGURE 17 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY TREATMENT, 2023-2030 (USD MILLION)
FIGURE 18 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY TREATMENT, CAGR (2023-2030)
FIGURE 19 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY TREATMENT, LIFELINE CURVE
FIGURE 20 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DIAGNOSIS, 2022
FIGURE 21 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DIAGNOSIS, 2023-2030 (USD MILLION)
FIGURE 22 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DIAGNOSIS, CAGR (2023-2030)
FIGURE 23 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DIAGNOSIS, LIFELINE CURVE
FIGURE 24 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DRUG TYPE, 2022
FIGURE 25 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DRUG TYPE, 2023-2030 (USD MILLION)
FIGURE 26 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DRUG TYPE, CAGR (2023-2030)
FIGURE 27 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DRUG TYPE, LIFELINE CURVE
FIGURE 28 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY END USER, 2022
FIGURE 29 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY END USER, 2023-2030 (USD MILLION)
FIGURE 30 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY END USER, CAGR (2023-2030)
FIGURE 31 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY END USER, LIFELINE CURVE
FIGURE 32 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DISTRIBUTION CHANNEL, 2022
FIGURE 33 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DISTRIBUTION CHANNEL, 2023-2030 (USD MILLION)
FIGURE 34 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DISTRIBUTION CHANNEL, CAGR (2023-2030)
FIGURE 35 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 36 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: SNAPSHOT (2022)
FIGURE 37 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY REGION (2022)
FIGURE 38 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY REGION (2023 & 2030)
FIGURE 39 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY REGION (2022 & 2030)
FIGURE 40 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY TREATMENT (2023-2030)
FIGURE 41 NORTH AMERICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: SNAPSHOT (2022)
FIGURE 42 NORTH AMERICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2022)
FIGURE 43 NORTH AMERICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2023 & 2030)
FIGURE 44 NORTH AMERICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2022 & 2030)
FIGURE 45 NORTH AMERICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY TREATMENT (2023-2030)
FIGURE 46 EUROPE EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: SNAPSHOT (2022)
FIGURE 47 EUROPE EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2022)
FIGURE 48 EUROPE EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2023 & 2030)
FIGURE 49 EUROPE EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2022 & 2030)
FIGURE 50 EUROPE EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY TREATMENT (2023-2030)
FIGURE 51 ASIA-PACIFIC EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: SNAPSHOT (2022)
FIGURE 52 ASIA-PACIFIC EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2022)
FIGURE 53 ASIA-PACIFIC EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2023 & 2030)
FIGURE 54 ASIA-PACIFIC EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2022 & 2030)
FIGURE 55 ASIA-PACIFIC EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY TREATMENT (2023-2030)
FIGURE 56 SOUTH AMERICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: SNAPSHOT (2022)
FIGURE 57 SOUTH AMERICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2022)
FIGURE 58 SOUTH AMERICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2023 & 2030)
FIGURE 59 SOUTH AMERICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2022 & 2030)
FIGURE 60 SOUTH AMERICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY TREATMENT (2023-2030)
FIGURE 61 MIDDLE EAST AND AFRICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: SNAPSHOT (2022)
FIGURE 62 MIDDLE EAST AND AFRICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2022)
FIGURE 63 MIDDLE EAST AND AFRICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2023 & 2030)
FIGURE 64 MIDDLE EAST AND AFRICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY COUNTRY (2022 & 2030)
FIGURE 65 MIDDLE EAST AND AFRICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: BY TREATMENT (2023-2030)
FIGURE 66 GLOBAL EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: COMPANY SHARE 2022 (%)
FIGURE 67 NORTH AMERICA EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: COMPANY SHARE 2022 (%)
FIGURE 68 EUROPE EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICS MARKET: COMPANY SHARE 2022 (%)
FIGURE 69 AISA-PACIFIC EXOCRINE PANCREATIC INSUFFICIENCY (EPI) THERAPEUTICS AND DIAGNOSTICSMARKET: COMPANY SHARE 2022 (%)

연구 방법론
데이터 수집 및 기준 연도 분석은 대규모 샘플 크기의 데이터 수집 모듈을 사용하여 수행됩니다. 이 단계에는 다양한 소스와 전략을 통해 시장 정보 또는 관련 데이터를 얻는 것이 포함됩니다. 여기에는 과거에 수집한 모든 데이터를 미리 검토하고 계획하는 것이 포함됩니다. 또한 다양한 정보 소스에서 발견되는 정보 불일치를 검토하는 것도 포함됩니다. 시장 데이터는 시장 통계 및 일관된 모델을 사용하여 분석하고 추정합니다. 또한 시장 점유율 분석 및 주요 추세 분석은 시장 보고서의 주요 성공 요인입니다. 자세한 내용은 분석가에게 전화를 요청하거나 문의 사항을 드롭하세요.
DBMR 연구팀에서 사용하는 주요 연구 방법론은 데이터 마이닝, 시장에 대한 데이터 변수의 영향 분석 및 주요(산업 전문가) 검증을 포함하는 데이터 삼각 측량입니다. 데이터 모델에는 공급업체 포지셔닝 그리드, 시장 타임라인 분석, 시장 개요 및 가이드, 회사 포지셔닝 그리드, 특허 분석, 가격 분석, 회사 시장 점유율 분석, 측정 기준, 글로벌 대 지역 및 공급업체 점유율 분석이 포함됩니다. 연구 방법론에 대해 자세히 알아보려면 문의를 통해 업계 전문가에게 문의하세요.
사용자 정의 가능
Data Bridge Market Research는 고급 형성 연구 분야의 선두 주자입니다. 저희는 기존 및 신규 고객에게 목표에 맞는 데이터와 분석을 제공하는 데 자부심을 느낍니다. 보고서는 추가 국가에 대한 시장 이해(국가 목록 요청), 임상 시험 결과 데이터, 문헌 검토, 재생 시장 및 제품 기반 분석을 포함하도록 사용자 정의할 수 있습니다. 기술 기반 분석에서 시장 포트폴리오 전략에 이르기까지 타겟 경쟁업체의 시장 분석을 분석할 수 있습니다. 귀하가 원하는 형식과 데이터 스타일로 필요한 만큼 많은 경쟁자를 추가할 수 있습니다. 저희 분석가 팀은 또한 원시 엑셀 파일 피벗 테이블(팩트북)로 데이터를 제공하거나 보고서에서 사용 가능한 데이터 세트에서 프레젠테이션을 만드는 데 도움을 줄 수 있습니다.