北米の分子診断サービス市場規模、シェア、トレンド分析レポート
Market Size in USD Billion
CAGR :
% 
 USD
66.14 Million
USD
122.42 Million
2024
2032
USD
66.14 Million
USD
122.42 Million
2024
2032
| 2025 –2032 | |
| USD 66.14 Million | |
| USD 122.42 Million | |
|
|
|
|
北米の分子診断サービス市場の細分化、サービスタイプ別(機器修理サービス、トレーニングサービス、コンプライアンスサービス、 校正 サービス、メンテナンスサービス、スケーラブルオートメーションサービス、ターンキーサービス、機器移転サービス、ハードウェアカスタマイズ、パフォーマンス保証サービス、設計開発サービス、サプライチェーンソリューション、新製品導入サービス、製造サービス、環境および規制サービス、医療管理システム認証および監査、臨床研究サービス、コンサルティングサービス、その他のサービス)、テクノロジー別(PCR、リアルタイムPCR、次世代シーケンシング、その他のテクノロジー)、エンドユーザー別(病院、診断センター、学術研究機関、その他) - 2032年までの業界動向と予測
北米の分子診断サービス市場規模
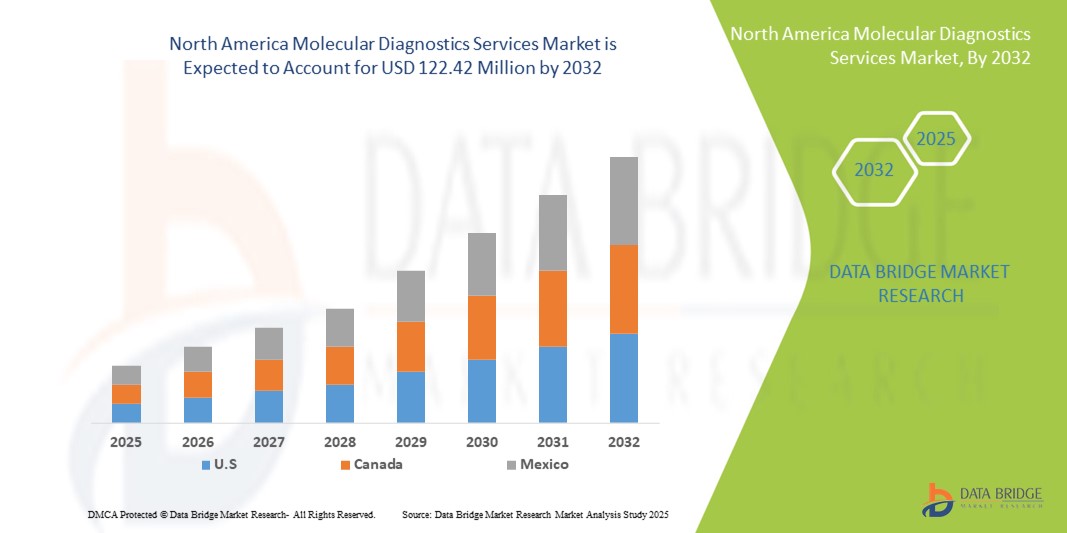
- 北米の分子診断サービス市場規模は2024年に6,614万米ドルと評価され、予測期間中に8.00%のCAGRで成長し、2032年には1億2,242万米ドル に達すると予想されています。
- 市場の成長は、感染症、腫瘍学、遺伝性疾患など、迅速で正確かつ個別化された検査へのニーズの高まりを受け、病院、診断センター、研究機関における分子診断サービスの導入拡大に大きく牽引されています。PCR、リアルタイムPCR、次世代シーケンシング(NGS)、その他の分子アッセイといった技術の進歩により、サービス提供能力がさらに向上し、迅速な結果提供、患者アウトカムの改善、そして臨床的有用性の向上が実現しています。
- さらに、疾患の早期発見、精密医療、ハイスループット診断ソリューションへの需要の高まりにより、分子診断サービスは現代の医療インフラの重要な構成要素として確立されつつあります。これらの要因が重なり合い、高度な分子診断サービスの導入が加速し、市場の成長を大きく促進しています。
北米の分子診断サービス市場分析
- 分子診断サービスは、病気の検出、遺伝子検査、個別化医療のための高度なツールを提供しており、その正確性、迅速な処理時間、標的治療戦略を導く能力により、病院と臨床検査室の両方でますます重要になっています。
- 分子診断サービスに対する需要の高まりは、感染症や癌の罹患率の上昇、病気の早期発見の重要性の高まり、医療システム全体にわたる精密医療アプローチの採用拡大によって主に促進されている。
- 米国は、先進的な医療インフラ、高い診断検査件数、そして大手サービスプロバイダーの存在に支えられ、2024年には北米の分子診断サービス市場において88.7%という最大の収益シェアを獲得し、市場を席巻しました。腫瘍学、感染症、遺伝子スクリーニングにおける分子検査の普及に加え、既存の診断企業と新興バイオテクノロジー企業による継続的なイノベーションも、成長を牽引しています。
- カナダは、予測期間中に北米の分子診断サービス市場で最も急速に成長する国になると予想されており、2025年から2032年にかけて11.5%のCAGRで拡大すると予測されています。この成長は、医療インフラへの投資の増加、がんや感染症の分子検査の採用の増加、および早期の病気の発見を支援する政府の取り組みに起因しています。
- PCRセグメントは、診断技術として比類のない信頼性、スピード、費用対効果により、2024年には北米の分子診断サービス市場において41.7%の収益シェアを占め、市場を席巻しました。PCRは、感染症診断、腫瘍学検査、遺伝子スクリーニング、法医学分析など、多様な用途における分子検査の基盤となっています。
レポートの範囲と分子診断サービス市場のセグメンテーション
|
属性 |
分子診断サービス主要市場インサイト |
|
対象セグメント |
|
|
対象国 |
北米
|
|
主要な市場プレーヤー |
|
|
市場機会 |
|
|
付加価値データ情報セット |
データブリッジマーケットリサーチがまとめた市場レポートには、市場価値、成長率、セグメンテーション、地理的範囲、主要プレーヤーなどの市場シナリオに関する洞察に加えて、専門家による詳細な分析、価格設定分析、ブランドシェア分析、消費者調査、人口統計分析、サプライチェーン分析、バリューチェーン分析、原材料/消耗品の概要、ベンダー選択基準、PESTLE分析、ポーター分析、規制の枠組みも含まれています。 |
北米の分子診断サービス市場動向
AIとデータ統合による診断精度の向上
- 北米の分子診断サービス市場において、人工知能(AI)、機械学習、そして高度なバイオインフォマティクスと既存の分子検査ワークフローとの統合が加速しており、その重要性はますます高まっています。こうした技術の融合により、PCRや次世代シーケンシングからデジタルPCRやマルチプレックスパネルに至るまで、幅広い検査において分析感度と特異性が向上し、検査室では同じデータからより臨床的に意義のある情報を抽出することが可能になっています。
- 例えば、AIを活用したアルゴリズムは、複雑なNGSパイプライン(リードアライメント、バリアントコール、アノテーションなどのタスク)の自動化にますます活用されています。これにより、実作業時間の短縮、人的ミスの最小化、生データから臨床的に実用的なレポートへの結果の移行の迅速化が実現します。これらの自動化されたパイプラインは、複数の研究室間での解釈の標準化にも役立ち、腫瘍学、希少疾患検査、感染症サーベイランスにおける再現性の高い意思決定をサポートします。
- AI統合により、デジタル病理学における自動画像解析、疾患進行および治療反応の予測モデル、専門家によるレビューが必要な非定型結果のインテリジェントなフラグ付けといった高度な機能が可能になります。機械学習モデルは、臨床的に重要な変異の優先順位付け、機能的影響の可能性の示唆、確認検査が必要な症例のトリアージを支援し、診断スループットと臨床的関連性の両方を向上させます。
- 分子診断プラットフォームと臨床検査情報管理システム(LIMS)、電子健康記録(EHR)、そして安全なクラウドインフラとのシームレスな連携により、リアルタイムの結果提供、遠隔専門医による診察、そして長期的な患者モニタリングが促進されます。一元化されたダッシュボードと相互運用可能なデータパイプラインにより、臨床医と公衆衛生チームは、傾向を可視化し、アウトブレイクを追跡し、分子診断結果をケアパスウェイに効率的に統合することが可能になります。
- よりインテリジェントで自動化され、相互接続された診断ワークフローへの移行は、臨床検査室と医療提供者への期待を再構築しています。サービスプロバイダーは、サンプル追跡、自動化されたウェットラボ機器、AI支援分析、そして臨床医向けレポートを組み合わせたエンドツーエンドのソリューションを開発しており、ルーチン検査や複雑な分子検査における処理時間の短縮、スループットの向上、そして費用対効果の向上を実現しています。
- AIとデータに基づく知見を統合的に提供する分子診断サービスへの需要は、病院、リファレンスラボ、公衆衛生機関において急速に高まっています。ステークホルダーは、個別化された治療方針の決定、抗菌薬適正使用、そして新たな感染症の脅威への迅速な対応を支援する、実用的でタイムリーな結果をますます重視しており、AIを活用した分子診断サービスは、多くのラボや医療システムにとって戦略的な優先事項となっています。
北米の分子診断サービス市場の動向
ドライバ
疾病負担の増加と精密医療の進歩によるニーズの高まり
- 北米における慢性疾患および感染症の罹患率の上昇と、早期発見および予防への関心の高まりが相まって、分子診断サービスの需要が大きく高まっています。
- 例えば、2024年3月、QIAGENは次世代シーケンシング(NGS)ベースの検査ポートフォリオの拡充を発表しました。これは、プレシジョン・メディシン(精密医療)の取り組みを支援し、より迅速かつ正確な疾患診断を可能にするものです。このような大手企業による戦略は、予測期間中に分子診断サービス業界の成長を加速させると予想されます。
- 医療システムが正確かつタイムリーな疾患特定をますます優先するにつれて、分子診断サービスは、従来の診断アプローチと比較してより高い感度と特異性を実現するリアルタイムPCR、次世代シーケンシング、バイオマーカーベースの検査などの高度なソリューションを提供します。
- さらに、分子診断とデジタルヘルスプラットフォームおよび臨床意思決定支援システムの統合により、病院、診断センター、研究機関全体での有用性が向上し、これらのサービスは現代の医療提供に不可欠な要素となっています。
- パーソナライズ医療の採用の増加と、ユーザーフレンドリーで拡張可能な分子診断ソリューションの利用可能性の増加が相まって、特に腫瘍学、感染症管理、遺伝性疾患スクリーニングの分野で市場拡大にさらに貢献しています。
抑制/挑戦
高額なサービスコストとデータプライバシーの問題に関する懸念
- NGSや特殊なバイオマーカー検査を含む高度な分子診断サービスに関連する比較的高いコストは、特に手頃な価格が限られている低所得および中所得地域では、広く普及するための大きな障壁となっています。
- 例えば、機器の校正、メンテナンス、コンプライアンスサービスに関連する費用は、診断センターと患者の全体的なコスト負担を増加させ、より広範な利用を制限する可能性があります。
- 費用対効果の高い検査ソリューション、革新的な価格モデル、政府資金による償還プログラムの開発を通じてこれらの課題に対処することは、市場浸透を促進する上で極めて重要です。さらに、分子診断サービスはクラウドベースのプラットフォームや統合データシステムに依存することが多いため、患者データのプライバシーと機密性の高いゲノム情報の取り扱いに関する懸念も課題となっています。
- 医療ITシステムのデータセキュリティの脆弱性に関する報告により、分子診断の利点にもかかわらず、一部の機関は分子診断の全面導入に慎重になっている。
- 堅牢なデータ暗号化の確保、GDPRやHIPAAなどの規制への確実なコンプライアンス、そしてプライバシー保護に関する継続的な消費者教育は、信頼を築く上で不可欠なステップです。さらに、より手頃な価格で利用しやすい分子診断サービスの開発は、世界市場の持続的な成長に不可欠です。
北米の分子診断サービス市場の範囲
市場は、サービスタイプ、テクノロジー、エンドユーザーに基づいてセグメント化されています。
- サービスタイプ別
サービスタイプに基づいて、分子診断サービス市場は、機器修理サービス、トレーニングサービス、コンプライアンスサービス、校正サービス、保守サービス、スケーラブルオートメーションサービス、ターンキーサービス、機器移転サービス、ハードウェアカスタマイズ、性能保証サービス、設計開発サービス、サプライチェーンソリューション、新製品導入サービス、製造サービス、環境・規制サービス、医療管理システム認証・監査、臨床研究サービス、コンサルティングサービス、その他のサービスに分類されます。臨床研究サービスセグメントは、高度な医薬品開発、バイオマーカー発見、検証プロセスをサポートする分子診断への依存度の高まりにより、2024年には28.4%と最大の市場収益シェアを占めました。臨床研究サービスは、信頼性の高いデータ生成と世界的な規制枠組みへの準拠を確保することにより、精密医療の開発を加速させる上で重要な役割を果たしています。がん、感染症、希少遺伝性疾患の罹患率の上昇により、製薬会社と分子診断サービスプロバイダー間の臨床研究パートナーシップの需要が大幅に増加しています。さらに、コンパニオン診断の進歩と臨床試験におけるリアルワールドエビデンスの採用の増加により、こうしたサービスに対する需要がさらに高まり、この分野が業界の基幹となっています。
スケーラブルな自動化サービス分野は、大量の検査を正確かつ効率的に処理できるハイスループット診断ソリューションへの切迫したニーズに牽引され、2025年から2032年にかけて21.6%という最も高い成長率を達成すると予想されています。世界中の検査室が作業量の増加に直面する中、自動化サービスはワークフローの合理化、手作業による介入の削減、人的ミスの最小化に貢献するとともに、ターンアラウンドタイムの短縮も確保します。ロボット工学、AIベースのプラットフォーム、自動液体処理システムを診断パイプラインに統合することで、検査室は完全にデジタル化された環境への移行を可能にしています。さらに、COVID-19パンデミックは、迅速でスケーラブルな診断能力の重要性を浮き彫りにし、公衆衛生と臨床の両方のアプリケーションで自動化サービスの導入を加速させました。医療システムが検査室の近代化への投資を増やす中、スケーラブルな自動化サービスは、将来を見据えた診断の礎として位置付けられています。
- テクノロジー別
技術に基づいて、分子診断サービス市場は、PCR、リアルタイムPCR、次世代シーケンシング(NGS)、およびその他の技術に分類されます。PCRセグメントは、診断技術としての比類のない信頼性、速度、および費用対効果により、2024年に41.7%という最大の収益シェアを占めました。PCRは、感染症診断、腫瘍学検査、遺伝子スクリーニング、および法医学分析を含む、さまざまなアプリケーションにわたる分子検査の基盤となっています。その広範な採用は、標準化されたワークフロー、確立されたインフラストラクチャ、および世界中の医療システムにわたる規制承認の利用可能性によってさらにサポートされています。PCRは、その使いやすさとさまざまな検査環境への適応性により、日常的な分子診断の好ましい選択肢であり続けています。さらに、デジタルPCRやマルチプレックスPCRなどの継続的なイノベーションにより、感度とスループット能力が拡張され、市場における支配的な地位を強化しています。
次世代シーケンシング(NGS)分野は、この技術が精密医療と高度診断の状況を一変させていることから、2025年から2032年にかけて22.3%という最も高いCAGRを達成すると予想されています。NGSは包括的なゲノムプロファイリングを提供し、臨床医が希少変異を特定し、複雑な遺伝子変異を検出し、高度にターゲットを絞った治療戦略を設計することを可能にします。腫瘍学、特に液体生検と腫瘍プロファイリングにおけるNGSの応用は、個別化治療のための実用的な洞察を提供することで、がん診断を変革しました。腫瘍学を超えて、NGSは感染症の監視、希少疾患の診断、生殖医療の分野で勢いを増しており、サービスプロバイダーに広大な機会を生み出しています。シーケンシングコストの低下と、バイオインフォマティクスツールおよびクラウドベースのデータ分析プラットフォームの継続的な改善により、NGSは世界中の臨床検査室でより利用しやすくなり、この市場で最も急速に成長している技術としての地位を固めています。
- エンドユーザー別
エンドユーザーに基づいて、分子診断サービス市場は、病院、診断センター、学術研究機関、その他に分類されます。病院セグメントは、分子診断を患者ケアパスウェイに統合する上で重要な役割を果たしていることから、2024年には46.2%という最大の収益シェアを占めました。病院は高度な診断検査の主要な拠点として機能し、迅速な疾患特定、治療モニタリング、複雑な状態の管理を可能にしています。病院における分子診断の採用は、がん、心血管疾患、感染症の発生など、正確でタイムリーな結果が求められる慢性疾患の蔓延によって推進されています。病院はまた、強力な償還枠組みと熟練した専門家の存在から恩恵を受け、高水準のサービス提供を保証しています。院内分子研究所への投資と診断サービスプロバイダーとの連携により、病院はエンドユーザー市場において引き続き優位に立っています。
診断センターセグメントは、費用対効果が高く、利便性が高く、アクセスしやすい検査ソリューションへの需要の高まりにより、2025年から2032年にかけて20.1%という最も高い成長率を記録すると予測されています。診断センターは、患者にとって病院に代わる手頃な選択肢を提供すると同時に、迅速な検査結果のターンアラウンドタイムを提供します。特に都市部および準都市部では、増加する中流階級の人々が予防的健康診断に積極的になり、診断センターの人気が高まっています。診断チェーンと分子診断サービスプロバイダーとの提携により、その範囲が拡大し、リソースが限られた環境でも高度な検査が可能になっています。さらに、在宅検体採取、ポイントオブケア分子検査、デジタルレポートサービスの普及により、診断センターは消費者にとって魅力的な選択肢となり、急速な成長軌道を支えています。
北米の分子診断サービス市場地域分析
- 強力な医療インフラ、高い診断検査量、大手サービスプロバイダーの存在に支えられ、北米は2024年に最大の収益シェアで分子診断サービス市場を席巻した。
- この地域は、高度な検査ネットワーク、確立された臨床研究施設、そして分子検査の日常的な臨床ワークフローへの統合の進展といった恩恵を受けています。腫瘍学、感染症検査、遺伝子スクリーニングにおける分子診断の急速な導入が、需要をさらに押し上げています。
- さらに、病院、診断サービスプロバイダー、製薬会社間の戦略的協力により、北米の市場リーダーシップが強化されています。
米国分子診断サービス市場インサイト
米国の分子診断サービス市場は、腫瘍学、感染症、遺伝子スクリーニング分野における分子検査の普及に牽引され、2024年には北米で最大の収益シェア(88.7%)を獲得しました。ワークフローの自動化、高度なバイオインフォマティクスソリューション、ハイスループット検査機能など、既存の診断企業と新興企業の両方による継続的なイノベーションが市場の成長を加速させています。さらに、個別化医療と精密治療への関心の高まりにより、臨床意思決定における分子診断の利用が拡大しており、米国が北米地域における主要市場としての地位をさらに強固なものにしています。
カナダの分子診断サービス市場に関する洞察
カナダの分子診断サービス市場は、予測期間中に北米で最も急速に成長すると予想されており、2025年から2032年にかけて年平均成長率(CAGR)11.5%で拡大すると予測されています。この成長は、医療インフラ、特に腫瘍学、感染症、遺伝性疾患の分子検査を導入している高度な診断ラボやがんセンターへの投資増加によって促進されています。早期疾患発見と全国的ながんスクリーニングプログラムを支援する政府の取り組みは、分子診断の導入をさらに加速させています。さらに、カナダでは個別化医療と精密医療への関心が高まっており、ハイスループット検査、次世代シーケンシング、バイオマーカー駆動型診断の需要が高まっています。医療費の増加、支援政策、最先端の分子技術の急速な導入が相まって、カナダは地域で最も急速に拡大する市場となっています。
北米の分子診断サービス市場シェア
分子診断サービス業界は、主に次のような定評のある企業によって牽引されています。
- F. ホフマン・ラ・ロシュ社(スイス)
- ダナハーコーポレーション(米国)
- ビオメリュー(フランス)
- QIAGEN(ドイツ)
- サーモフィッシャーサイエンティフィック社(米国)
- バイオ・ラッド・ラボラトリーズ社(米国)
- アボット(米国)
- DiaSorin SpA(イタリア)
- ホロジック社(米国)
- ラボコープ(米国)
- クエスト・ダイアグノスティクス・インコーポレーテッド(米国)
- ネオジェノミクス・ラボラトリーズ(米国)
- ミリアド・ジェネティクス社(米国)
- エグザクト・サイエンシズ・コーポレーション(米国)
- ガーダント・ヘルス社(米国)
- フルジェント・ジェネティクス(米国)
北米の分子診断サービス市場の最新動向
- 2022年12月、英国を拠点とするPOC(Point of Care)診断企業であるLumiraDxは、ビル&メリンダ・ゲイツ財団から1,420万ドルの助成金を授与されました。この資金は、資源が限られた環境における結核診断の改善を目的としたPOC(Point of Care)分子状結核検査システムの継続的な開発を支援します。この取り組みは、革新的な診断ソリューションを通じて北米の健康を向上させるという財団の目標と一致しています。
- 2024年3月、セフェイド社は、分娩中のB群連鎖球菌(GBS)の定性検出を目的とした二重標的分子診断検査「Xpert Xpress GBS」について、米国食品医薬品局(FDA)の承認を取得したと発表しました。この検査は、セフェイド社のGeneXpertシステムでの使用を想定して設計されており、GBSゲノムの高度に保存された領域に新たな二重標的を組み込むことで、感度と細菌株のカバー率を向上させています。
- 2024年3月、ビオメリューは、BIOFIRE SPOTFIRE呼吸器/咽喉炎(R/ST)パネルが米国食品医薬品局(FDA)のSpecial 510(k)承認および臨床検査改善法(CLIA)の適用免除を取得したと発表しました。このパネルはBIOFIRE SPOTFIREシステムで使用するために設計されており、呼吸器および咽喉炎の病原体を迅速に検出し、より情報に基づいた臨床判断を可能にします。
- 2024年8月、がんの早期発見を専門とするグレイル社は、従業員の約30%に相当する約350人の人員削減を発表しました。この組織再編は、1回の採血で複数の種類のがんを検出できる主力のがん検出検査「ガレリ」の開発にリソースを集中させる戦略的な転換の一環です。
- 2025年4月、パーキンエルマー社のライフサイエンス・診断部門として知られていたRevvityは、研究開発費の大幅な増加を発表しました。これは、ライフサイエンス・診断分野への注力強化を目的として、2023年3月に分社化とリブランディングを行ったことを受けた措置です。Revvityは、分子診断市場におけるイノベーションを推進するため、ソフトウェアと研究への投資を含め、製品ラインナップの拡充を計画しています。
SKU-
世界初のマーケットインテリジェンスクラウドに関するレポートにオンラインでアクセスする
- インタラクティブなデータ分析ダッシュボード
- 成長の可能性が高い機会のための企業分析ダッシュボード
- カスタマイズとクエリのためのリサーチアナリストアクセス
- インタラクティブなダッシュボードによる競合分析
- 最新ニュース、更新情報、トレンド分析
- 包括的な競合追跡のためのベンチマーク分析のパワーを活用
調査方法
データ収集と基準年分析は、大規模なサンプル サイズのデータ収集モジュールを使用して行われます。この段階では、さまざまなソースと戦略を通じて市場情報または関連データを取得します。過去に取得したすべてのデータを事前に調査および計画することも含まれます。また、さまざまな情報ソース間で見られる情報の不一致の調査も含まれます。市場データは、市場統計モデルと一貫性モデルを使用して分析および推定されます。また、市場シェア分析と主要トレンド分析は、市場レポートの主要な成功要因です。詳細については、アナリストへの電話をリクエストするか、お問い合わせをドロップダウンしてください。
DBMR 調査チームが使用する主要な調査方法は、データ マイニング、データ変数が市場に与える影響の分析、および一次 (業界の専門家) 検証を含むデータ三角測量です。データ モデルには、ベンダー ポジショニング グリッド、市場タイムライン分析、市場概要とガイド、企業ポジショニング グリッド、特許分析、価格分析、企業市場シェア分析、測定基準、グローバルと地域、ベンダー シェア分析が含まれます。調査方法について詳しくは、お問い合わせフォームから当社の業界専門家にご相談ください。
カスタマイズ可能
Data Bridge Market Research は、高度な形成的調査のリーダーです。当社は、既存および新規のお客様に、お客様の目標に合致し、それに適したデータと分析を提供することに誇りを持っています。レポートは、対象ブランドの価格動向分析、追加国の市場理解 (国のリストをお問い合わせください)、臨床試験結果データ、文献レビュー、リファービッシュ市場および製品ベース分析を含めるようにカスタマイズできます。対象競合他社の市場分析は、技術ベースの分析から市場ポートフォリオ戦略まで分析できます。必要な競合他社のデータを、必要な形式とデータ スタイルでいくつでも追加できます。当社のアナリスト チームは、粗い生の Excel ファイル ピボット テーブル (ファクト ブック) でデータを提供したり、レポートで利用可能なデータ セットからプレゼンテーションを作成するお手伝いをしたりすることもできます。