アジア太平洋ワクチン市場
Market Size in USD Billion
CAGR :
% 
 USD
9,048.51 Million
USD
18,031.69 Million
2022
2030
USD
9,048.51 Million
USD
18,031.69 Million
2022
2030
| 2023 –2030 | |
| USD 9,048.51 Million | |
| USD 18,031.69 Million | |
|
|
|
アジア太平洋地域のワクチン市場、構成別(混合ワクチンおよび単一ワクチン)、タイプ別(サブユニット、組み換え、多糖類、および結合ワクチン、生弱毒化ワクチン、不活化ワクチン、およびトキソイドワクチン)、種類別(定期ワクチン、推奨ワクチン、および必須ワクチン)、接種年齢別(小児ワクチンおよび成人ワクチン)、疾患別(肺炎球菌性疾患、麻疹、おたふく風邪および水痘、三種混合ワクチン、肝炎、インフルエンザ、腸チフス、髄膜炎菌性、狂犬病、日本脳炎、黄熱病、およびその他)、接種経路別(注射、経口、および経鼻)、エンドユーザー別(地域病院、病院、専門センター、診療所、およびその他)、流通チャネル別(病院薬局、小売薬局、およびオンライン薬局) - 2030 年までの業界動向と予測。
アジア太平洋ワクチン市場の分析と洞察
細菌性疾患やウイルス性疾患を含む感染症の蔓延の増加は、市場に有利な成長をもたらします。これに加えて、政府の支援の増加と新しいワクチンの発売もワクチン市場を後押ししています。ワクチン市場の成長を後押しするもう1つの要因は、ワクチン接種の認知度と効果的なCOVID-19ワクチンの需要の高まりです。
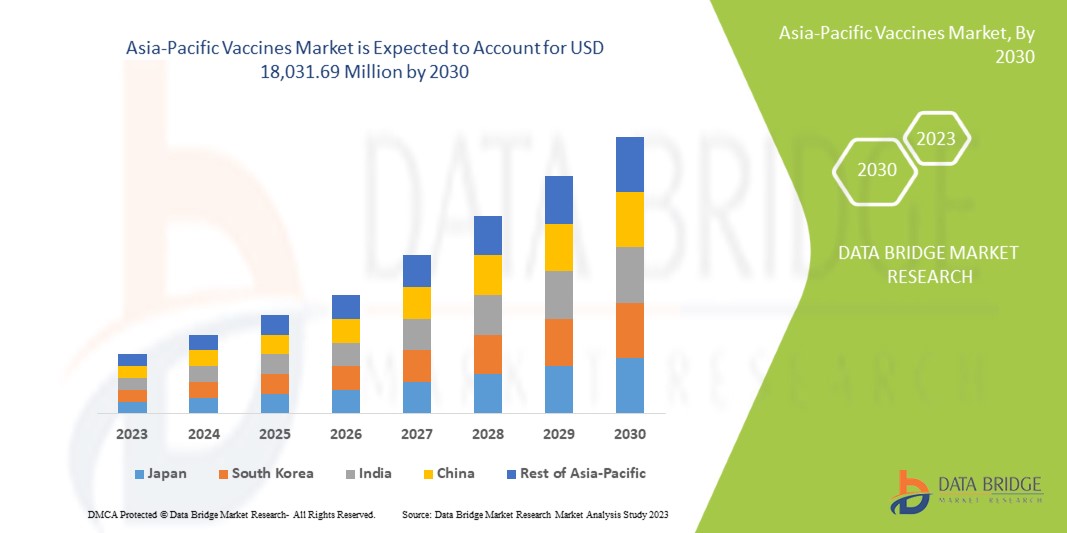
アジア太平洋地域のワクチン市場は、2023年から2030年の予測期間に成長すると予想されています。データブリッジマーケットリサーチは、市場は2023年から2030年の予測期間に9.4%のCAGRで成長し、2022年の90億4,851万米ドルから2030年には180億3,169万米ドルに達すると分析しています。
|
レポートメトリック |
詳細 |
|
予測期間 |
2023年から2030年 |
|
基準年 |
2022 |
|
歴史的な年 |
2021 (2015~2020年にカスタマイズ可能) |
|
定量単位 |
収益(百万米ドル) |
|
対象セグメント |
成分別(混合ワクチンおよび単一ワクチン)、タイプ別(サブユニットワクチン、組み換えワクチン、多糖体ワクチン、結合ワクチン、生弱毒化ワクチン、不活化ワクチン、トキソイドワクチン)、種類別(定期接種ワクチン、推奨ワクチン、必須ワクチン)、接種年齢別(小児用ワクチンおよび成人用ワクチン)、疾患別(肺炎球菌感染症、麻疹、おたふく風邪および水痘、三種混合ワクチン、肝炎、インフルエンザ、腸チフス、髄膜炎菌性疾患、狂犬病、日本脳炎、黄熱病、その他)、接種経路別(注射、経口、経鼻)、エンドユーザー別(地域病院、病院、専門センター、診療所、その他)、流通チャネル別(病院薬局、小売薬局、オンライン薬局) |
|
対象国 |
日本、中国、オーストラリア、インド、韓国、シンガポール、インドネシア、タイ、マレーシア、フィリピン、ベトナム、その他アジア太平洋地域 |
|
対象となる市場プレーヤー |
バーラト・バイオテック、バイオロジカル・E・リミテッド、バイオ・ファーマ、セラム・インスティテュート・オブ・インディア・プライベート・リミテッド、武田薬品工業株式会社、メルク・シャープ・アンド・ドーム社(メルク・アンド・カンパニー社の子会社)、アボット、アストラゼネカ、サノフィ、ファイザー社、ヤンセン・グローバル・サービス社(ジョンソン・エンド・ジョンソン・サービスの子会社)、F・ホフマン・ラ・ロシュ社、パナセア・バイオテック社、バクスター・ヴァッキネス社(バクスターの子会社)など |
アジア太平洋ワクチン市場の市場定義
ワクチンは、特定の病気に対する免疫を誘発するために個々の免疫系を刺激する製品です。ワクチンは記憶と認識の原理に基づいて機能します。弱毒化または殺菌された微生物が体内に注入されると、これらの微生物は免疫系の記憶細胞である B 細胞に病原体を認識させます。将来、同じ病原体が体を攻撃した場合、ワクチンはそれらに対抗します。肺炎球菌感染症、麻疹、おたふく風邪、風疹、肝炎、インフルエンザ、腸チフス、水痘、狂犬病などの感染症に対するワクチンが発見されています。
ワクチンには、混合ワクチン(病原体の異なる株を含む)と単一ワクチン(病原体の単一の株を含む)の 2 種類があります。病原体から抽出された材料に基づいて、多糖類の殻、DNA、RNA、不活化または生の生物全体など、さまざまな種類のワクチンが開発されています。
これらは、ポリオなどの病気の根絶に役立ったワクチンです。ワクチンの好みや効率に応じて、注射、経口、経鼻など、いくつかの投与経路で投与できます。ただし、全身反応を誘発するため、注射によるワクチン投与経路が特に好まれます。ワクチン接種は、病院、地域の診療所、専門診療所などで、ワクチン投与装置に関する適切な知識を持つ訓練を受けた人員によって実施できます。
細菌性疾患やウイルス性疾患を含む感染症の蔓延の増加は、市場に有利な成長をもたらします。これに加えて、政府の支援の増加と新しいワクチンの発売もワクチン市場を後押ししています。ワクチン市場の成長を後押しするもう1つの要因は、ワクチン接種の認知度と効果的なCOVID-19ワクチンの需要の高まりです。
アジア太平洋ワクチン市場の動向
このセクションでは、市場の推進要因、利点、機会、制約、課題について理解します。これらについては、以下で詳しく説明します。
ドライバー
予防接種プログラムとキャンペーンの拡大
慢性疾患の増加に伴い、世界中で予防接種プログラムやキャンペーンが増加しています。肝炎、ジフテリア、百日咳、ポリオなどの感染症が蔓延しているため、ワクチン接種に関する意識を高めることが急務であり、いくつかのキャンペーンやプログラムを立ち上げることでそれを達成できます。感染症の増加に伴い、予防接種プログラムの数も増加していると報告されています。衰弱性疾患と闘うために、予防接種の普及率も世界中で増加しています。
それでも、約 2,000 万人がワクチン接種を受けていない、または十分に接種を受けていないため、ワクチン接種の需要が高まっています。慢性疾患の罹患率は世界中で増加しています。そのため、ワクチン接種の必要性は高まっています。これは、予防接種プログラムとキャンペーンの拡大により、ワクチン市場が拡大すると予想されることを意味します。
インフルエンザ、感染症、ウイルス性疾患などの慢性疾患の蔓延率が高い
感染症の蔓延は世界中で増加しており、インフルエンザや細菌感染症が急増していることが分かっています。感染症の増加率により、ワクチン接種や免疫接種によって予防できる病気の予防の必要性が生じています。病気が大幅に増加しているため、集団ワクチン接種が緊急に必要とされています。集団ワクチン接種には多くの病気が必要であるため、ワクチン市場に有利な成長をもたらすことが期待されています。
感染例が増加するにつれて、人口の急増を防ぐため、集団ワクチン接種への注目も高まっています。世界中で、若いときにワクチン接種を受けていない多くの人々が予防接種を受けています。そのため、このようなニーズに応えるために、新しいワクチンの需要が高まっており、アジア太平洋地域のワクチン市場の牽引役となることが期待されています。
拘束
登録ワクチンが入手できない
ワクチンの厳格な規制承認と開発手続きに時間がかかることが、登録済みのワクチンが入手できない原因の一部となっています。規制当局は、ワクチンの評価、認可、管理、監視に困難を感じています。これらの規制により、ワクチンの世界的供給が遅れています。
したがって、さまざまな国の製造会社の規制プロセスと文書作成は、ワクチン市場の成長を抑制し、妨げる可能性があります。
機会
市場参加者による戦略的取り組み
市場プレーヤーは、ワクチン市場において、拡大、コラボレーション、買収を含むさまざまな戦略的イニシアチブを採用しています。これらのイニシアチブにより、企業の製品ポートフォリオが拡大し、市場が拡大し、顧客間の製品需要が高まり、最終的に市場プレーヤーは最大の収益を得ることができます。
As the demand for effective and novel vaccines is increasing worldwide, these strategic initiatives taken by top market players aim at enhancing business operations and earning more profitability in the market.
Various strategic initiatives adopted by the market players allowed them to expand its root in vaccines and earn more market growth. Thus, the market players operating in vaccines are adopting several strategic initiatives expected to act as an opportunity for the vaccine market growth.
CHALLENGE
SIDE EFFECTS CAUSED BY VACCINES
The vaccine is a medical product that helps in preventing different diseases. But sometimes, side effects arise due to the use of the vaccines. Some of them are mild side effects which are redness, soreness, or swelling at the injection site. But the adverse side effects of the vaccines are rare but are life-threatening.
The life-threatening side effects can give rise to fear in the population's mind. Moreover, it impacts the credibility of vaccine manufacturers, affecting product sales. This thus suggests that side effects caused by vaccines may hamper the growth of the vaccines market.
Recent Developments
- In October 2022, Indonesia Launched its First Home-Grown COVID-19 Vaccine. IndoVac vaccine has been developed jointly by Indonesia's state-owned pharmaceutical company Bio Farma and the Baylor College of Medicine, an independent health sciences center in Houston, Texas
- In November 2020, Merck Sharp & Dohme Corp., a Merck & Co., Inc. subsidiary, signed an agreement to acquire OncoImmune, a clinical-stage biopharmaceutical company. The OncoImmune Company is highly focused on the development of COVID-19 treatment options. Through this agreement, the company is expecting to develop new vaccine candidates
- In September 2020, Sanofi and GSK signed an agreement with the government of Canada to supply 72 million doses of COVID-19 vaccines. There is a high demand for COVID-19 vaccines, and the demand is increasing with the increasing pandemic. This agreement allowed the company to ensure future potential
Asia-Pacific Vaccines Market Scope
Asia-Pacific vaccines market is segmented into composition, type, kind, age of administration, diseases, route of administration, end user, and distribution channel. The growth amongst these segments will help you analyze meager growth segments in the industries and provide the users with a valuable market overview and market insights to make strategic decisions to identify core market applications.
COMPOSITION
- Combination vaccines
- Mono vaccines
On the basis of composition, the Asia-Pacific vaccines market is segmented into combination vaccines and monovaccines.
TYPE
- Subunit, recombinant, polysaccharide and conjugate vaccines
- Live-attenuated vaccines
- Inactivated vaccines
- Toxoid vaccines
On the basis of type, the Asia-Pacific vaccines market is segmented into subunit, recombinant, polysaccharide, and conjugate vaccines, live-attenuated vaccines, inactivated vaccines, and toxoid vaccines.
KIND
- Routine vaccine
- Recommended vaccine
- Required vaccine
On the basis of kind, the Asia-Pacific vaccines market is segmented into routine vaccine, recommended vaccine, and required vaccine.
AGE OF ADMINISTRATION
- Pediatric vaccine
- Adult vaccine
On the basis of age of administration, the Asia-Pacific vaccines market is segmented into pediatric vaccine and adult vaccine
DISEASES
- Pneumococcal disease
- Measles, mumps & varicella
- DPT
- Hepatitis
- Influenza
- Typhoid
- Meningococcal
- Varicella
- Rabies
- Japanese encephalitis
- Yellow fever
- Others
On the basis of diseases, the Asia-Pacific vaccines market is segmented into pneumococcal disease, measles, mumps, and varicella, DPT, hepatitis, influenza, typhoid, meningococcal, rabies, Japanese encephalitis, yellow fever, and others.
ROUTE OF ADMINISTRATION
- Injectable
- Nasal
- Oral
On the basis of route of administration, the Asia-Pacific vaccines market is segmented into injectable, oral, and nasal.
END USER
- Community hospitals
- Hospitals
- Specialty centres
- Clinics
- Others
On the basis of end user, the Asia-Pacific vaccines market is segmented into community hospitals, hospitals, specialty centres, clinics, and others.
DISTRIBUTION CHANNEL
- Hospital pharmacy
- Retail pharmacy
- Online pharmacy
On the basis of distribution channel, the Asia-Pacific vaccines market is segmented into hospital pharmacy, retail pharmacy, and online pharmacy.
Asia-Pacific Vaccines Market Regional Analysis/Insights
Asia-Pacific vaccines market is analyzed, and market size insights and trends are provided by country, composition, type, kind, age of administration, diseases, route of administration, end user, and distribution channel, as referenced above.
The countries covered in this market report are Japan, China, South Korea, India, Australia, Singapore, Thailand, Malaysia, Indonesia, Philippines, Vietnam, and the rest of Asia-Pacific.
Japan is expected to dominate the Asia-Pacific vaccine market regarding market share and revenue and will continue to flourish its dominance during the forecast period. This is due to a rising preference for preventive health check-ups.
The country section of the report also provides individual market-impacting factors and changes in regulations in the market that impact the current and future trends of the market. Data points, such as new and replacement sales, country demographics, disease epidemiology, and import-export tariffs, are some of the major pointers used to forecast the market scenario for individual countries. In addition, the presence and availability of Global brands and their challenges faced due to competition from local and domestic brands and the impact of sales channels are considered while providing forecast analysis of the country data.
Competitive Landscape and Asia-Pacific Vaccines Market Share Analysis
The vaccines market competitive landscape provides details of a competitor. Details include company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, Global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, and application dominance. The above data points are only related to the companies' focus on the vaccine market.
Some of the major players operating in the Asia-Pacific vaccines market are Bharat Biotech, Biological E Limited, Bio Farma, Serum Institute of India Pvt. Ltd., Takeda Pharmaceutical Company Limited, Merck Sharp & Dohme Corp. (a subsidiary of Merck & Co., Inc.), Abbott, AstraZeneca, Sanofi, Pfizer Inc., Janssen Global Services, LLC (a subsidiary of Johnson & Johnson Services, Inc.), F. Hoffmann-La Roche Ltd, Panacea Biotec Ltd, and BAXTER VACCINES (a subsidiary of Baxter), among others.
SKU-
世界初のマーケットインテリジェンスクラウドに関するレポートにオンラインでアクセスする
- インタラクティブなデータ分析ダッシュボード
- 成長の可能性が高い機会のための企業分析ダッシュボード
- カスタマイズとクエリのためのリサーチアナリストアクセス
- インタラクティブなダッシュボードによる競合分析
- 最新ニュース、更新情報、トレンド分析
- 包括的な競合追跡のためのベンチマーク分析のパワーを活用
目次
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF THE ASIA-PACIFIC VACCINES MARKET
1.4 LIMITATIONS
1.5 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 CURRENCY AND PRICING
2.5 DBMR TRIPOD DATA VALIDATION MODEL
2.6 MULTIVARIATE MODELLING
2.7 COMPOSITION LIFELINE CURVE
2.8 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.9 DBMR MARKET POSITION GRID
2.1 SECONDARY SOURCES
2.11 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHT
4.1 PESTEL MODEL
4.2 PORTER'S FIVE FORCES
4.3 EPIDEMIOLOGY
4.4 INDUSTRIAL INSIGHTS:
4.5 PIPELINE ANALYSIS
4.6 ASIA-PACIFIC VACCINES MARKET: SUPPLY CHAIN MANAGEMENT OF VACCINES
4.6.1 COLD CHAIN STORAGE:
4.6.2 PROCESS OF LOGISTICS
5 REGULATORY FRAMEWORK
5.1 JAPAN
5.2 CHINA
5.3 SOUTH KOREA
5.4 INDIA
5.5 AUSTRALIA
5.6 SINGAPORE
5.7 THAILAND
5.8 MALAYSIA
5.9 INDONESIA
5.1 VIETNAM
5.11 PHILIPPINES
5.12 REST OF ASIA-PACIFIC
5.12.1 TAIWAN
5.12.2 CAMBODIA
6 MARKET OVERVIEW
6.1 DRIVERS
6.1.1 GROWING IMMUNIZATION PROGRAMS AND CAMPAIGNS
6.1.2 HIGH PREVALENCE OF CHRONIC CONDITIONS SUCH AS FLU, INFECTIOUS AND VIRAL DISEASES
6.1.3 IMPROVEMENT IN TREATMENT
6.1.4 LAUNCH OF NEWER VACCINES
6.1.5 INCREASING GOVERNMENT SUPPORT
6.2 RESTRAINTS
6.2.1 UNAVAILABILITY OF REGISTERED VACCINES
6.2.2 DIFFICULTIES ASSOCIATED WITH THE TRANSPORT AND PRODUCTION OF VACCINES
6.3 OPPORTUNITIES
6.3.1 STRATEGIC INITIATIVES BY THE MARKET PLAYERS
6.3.2 PRESENCE OF PIPELINE PRODUCTS
6.3.3 RISE IN EXPENDITURE IN THE HEALTHCARE SECTOR
6.3.4 INCREASING AWARENESS FOR VACCINATION
6.4 CHALLENGES
6.4.1 SIDE EFFECTS CAUSED BY VACCINES
6.4.2 FEAR AMONG PATIENTS RELATED TO INJECTIONS AND NEEDLE STICKS
6.4.3 PRODUCT RECALL
7 ASIA-PACIFIC VACCINES MARKET, BY COMPOSITION
7.1 OVERVIEW
7.2 COMBINATION VACCINES
7.3 MONO VACCINES
8 ASIA-PACIFIC VACCINES MARKET, BY TYPE
8.1 OVERVIEW
8.2 SUBUNIT, RECOMBINANT, POLYSACCHARIDE, AND CONJUGATE VACCINES
8.2.1 PNEUMOCOCCAL DISEASE
8.2.2 HIB (HAEMOPHILUS INFLUENZA TYPE B) DISEASE
8.2.3 HPV (HUMAN PAPILLOMA VIRUS)
8.2.4 HEPATITIS B
8.2.5 MENINGOCOCCAL
8.2.6 SHINGLES
8.2.7 WHOOPING COUGH
8.2.8 OTHERS
8.3 LIVE-ATTENUTAED VACCINES
8.3.1 ROTAVIRUS
8.3.2 MEASLES
8.3.3 MUMPS
8.3.4 RUBELLA
8.3.5 SMALLPOX
8.3.6 YELLOW FEVER
8.3.7 OTHERS
8.4 INACTIVATED VACCINES
8.4.1 FLU (SHOT ONLY)
8.4.2 POLIO (SHOT ONLY)
8.4.3 HEPATITIS A
8.4.4 RABIES
8.4.5 OTHERS
8.5 TOXOID VACCINES
8.5.1 DIPHTHERIA, TETANUS & PERTUSSIS (DTP)
8.5.2 OTHERS
9 ASIA-PACIFIC VACCINES MARKET, BY KIND
9.1 OVERVIEW
9.2 ROUTINE VACCINES
9.2.1 PNEUMOCOCCAL DISEASES
9.3 DIPTHERIA, TETANUS & PERTUSIS(DPT)
9.3.1 HIB (HAEMOPHILUS INFLUENZA TYPE B) DISEASE
9.3.2 MEASLES
9.3.3 MUMPS
9.3.4 HEPATITIS B
9.3.5 RUBELLA
9.3.6 POLIO
9.3.7 OTHERS
9.4 RECOMMENDED VACCINE
9.4.1 TYPHOID FEVER VACCINE
9.5 HEPATITIS A
9.5.1 RABIES
9.5.2 JAPANESE ENCEPHALITIS
9.5.3 TICK-BORNE ENCEPHALITIS
9.5.4 CHOLERA
9.5.5 OTHERS
9.6 REQUIRED VACCINE
9.6.1 MENINGOCOCCAL
9.7 YELLOW FEVER
9.7.1 OTHERS
10 ASIA-PACIFIC VACCINES MARKET, BY AGE OF ADMINISTRATION
10.1 OVERVIEW
10.2 PEDIATRIC VACCINE
10.2.1 PNEUMOCOCCAL DISEASES
10.3 MEASLES, MUMPS & RUBELLA
10.3.1 DIPTHERIA, TETANUS & PERTUSIS (DPT)
10.3.2 ROTAVIRUS
10.3.3 MENINGOCOCCAL
10.3.4 VARICELLA
10.3.5 POLIO
10.3.6 TUBERCULOSIS
10.3.7 MALARIA
10.3.8 OTHERS
10.4 ADULT VACCINE
10.4.1 INFLUENZA
10.5 HPV (HUMAN PAPILLOMA VIRUS)
10.5.1 TYPHOID
10.5.2 HEPATITIS B
10.5.3 JAPANESE ENCEPHALITIS
10.5.4 YELLOW FEVER
10.5.5 CANCER
10.5.6 OTHERS
11 ASIA-PACIFIC VACCINES MARKET, BY DISEASES
11.1 OVERVIEW
11.2 PNEUMOCCOCAL DISEASE
11.3 MEASLES, MUMPS & RUBELLA
11.4 DPT
11.5 HEPATITIS
11.6 INFLUENZA
11.7 TYPHOID
11.8 MENINGOCOCCAL
11.9 VARICELLA
11.1 RABIES
11.11 JAPANESE ENCEPHALITIS
11.12 YELLOW FEVER
11.13 OTHERS
12 ASIA-PACIFIC VACCINES MARKET, BY ROUTE OF ADMINISTRATION
12.1 OVERVIEW
12.2 INJECTABLE
12.2.1 INTRAMUSCULAR
12.2.2 SUBCUTANEOUS
12.2.3 INTRADERMAL
12.3 ORAL
12.4 NASAL
13 ASIA-PACIFIC VACCINES MARKET, BY END USER
13.1 OVERVIEW
13.2 COMMUNITY HOSPITALS
13.3 HOSPITALS
13.4 SPECIALTY CENTERS
13.5 CLINICS
13.6 OTHERS
14 ASIA-PACIFIC VACCINES MARKET, BY DISTRIBUTION CHANNEL
14.1 OVERVIEW
14.2 HOSPITAL PHARMACY
14.3 RETAIL PHARMACY
14.4 ONLINE PHARMACY
15 ASIA-PACIFIC VACCINE MARKET
15.1 ASIA-PACIFIC
15.1.1 JAPAN
15.1.2 CHINA
15.1.3 AUSTRALIA
15.1.4 INDIA
15.1.5 SOUTH KOREA
15.1.6 SINGAPORE
15.1.7 MALAYSIA
15.1.8 THAILAND
15.1.9 INDONESIA
15.1.10 PHILIPPINES
15.1.11 VIETNAM
15.1.12 REST OF ASIA PACIFIC
16 ASIA-PACIFIC VACCINES MARKET: COMPANY LANDSCAPE
16.1 COMPANY SHARE ANALYSIS: ASIA-PACIFIC
16.2 DISEASE SHARE ANALYSIS: PFIZER, INC.
16.3 COUNTRY SHARE ANALYSIS: PFIZER, INC.
16.4 DISEASE SHARE ANALYSIS: MERCK SHARP & DOHME CORP. (A SUBSIDIARY OF MERCK & CO., INC.)
16.5 COUNTRY SHARE ANALYSIS: MERCK SHARP & DOHME CORP. (A SUBSIDIARY OF MERCK & CO., INC.)
16.6 DISEASE SHARE ANALYSIS: GLAXOSMITHKLINE PLC
16.7 COUNTRY SHARE ANALYSIS: GLAXOSMITHKLINE PLC.
16.8 DISEASE SHARE ANALYSIS: SANOFI
16.9 COUNTRY SHARE ANALYSIS: SANOFI
16.1 DISEASE SHARE ANALYSIS: SERUM INSTITUTE OF INDIA PVT. LTD.
16.11 COUNTRY SHARE ANALYSIS: SERUM INSTITUTE OF INDIA PVT. LTD.
17 SWOT ANALYSIS
18 COMPANY PROFILE
18.1 PFIZER INC.
18.1.1 COMPANY SNAPSHOT
18.1.2 REVENUE ANALYSIS
18.1.3 PRODUCT PORTFOLIO
18.1.4 RECENT DEVELOPMENTS
18.2 MERCK SHARP & DOHME CORP. (A SUBSIDIARY OF MERCK & CO., INC.)
18.2.1 COMPANY SNAPSHOT
18.2.2 REVENUE ANALYSIS
18.2.3 COMPANY WEBSITE AND PRESS RELEASES
18.2.4 PRODUCT PORTFOLIO
18.2.5 RECENT DEVELOPMENTS
18.3 GLAXOSMITHKLINE PLC.
18.3.1 COMPANY SNAPSHOT
18.3.2 REVENUE ANALYSIS
18.3.3 PRODUCT PORTFOLIO
18.3.4 RECENT DEVELOPMENTS
18.4 SANOFI
18.4.1 COMPANY SNAPSHOT
18.4.2 REVENUE ANALYSIS
18.4.3 PRODUCT PORTFOLIO
18.4.4 RECENT DEVELOPMENTS
18.5 SERUM INSTITUTE OF INDIA PVT. LTD.
18.5.1 COMPANY SNAPSHOT
18.5.2 PRODUCT PORTFOLIO
18.5.3 RECENT DEVELOPMENTS
18.6 ABBOTT
18.6.1 COMPANY SNAPSHOT
18.6.2 REVENUE ANALYSIS
18.6.3 PRODUCT PORTFOLIO
18.6.4 RECENT DEVELOPMENTS
18.7 ASTRAZENECA (2022)
18.7.1 COMPANY SNAPSHOT
18.7.2 REVENUE ANALYSIS
18.7.3 PRODUCT PORTFOLIO
18.7.4 RECENT DEVELOPMENTS
18.8 ALK
18.8.1 COMPANY SNAPSHOT
18.8.2 REVENUE ANALYSIS
18.8.3 PRODUCT PORTFOLIO
18.8.4 RECENT DEVELOPMENTS
18.9 BAXTER VACCINES (A SUBSIDIARY OF BAXTER)
18.9.1 COMPANY SNAPSHOT
18.9.2 REVENUE ANALYSIS
18.9.3 PRODUCT PORTFOLIO
18.9.4 RECENT DEVELOPMENT
18.1 BHARAT BIOTECH
18.10.1 COMPANY SNAPSHOT
18.10.2 PRODUCT PORTFOLIO
18.10.3 RECENT DEVELOPMENTS
18.11 BIO FARMA
18.11.1 COMPANY SNAPSHOT
18.11.2 PRODUCT PORTFOLIO
18.11.3 RECENT DEVELOPMENTS
18.12 BIOLOGICAL E LIMITED
18.12.1 COMPANY SNAPSHOT
18.12.2 PRODUCT PORTFOLIO
18.12.3 RECENT DEVELOPMENTS
18.13 DAIICHI SANKYO COMPANY, LIMITED
18.13.1 COMPANY SNAPSHOT
18.13.2 REVENUE ANALYSIS
18.13.3 PRODUCT PORTFOLIO
18.13.4 RECENT DEVELOPMENTS
18.14 F. HOFFMANN-LA ROCHE LTD
18.14.1 COMPANY SNAPSHOT
18.14.2 REVENUE ANALYSIS
18.14.3 PRODUCT PORTFOLIO
18.14.4 RECENT DEVELOPMENT
18.15 JANSSEN GLOBAL SERVICES, LLC (A SUBSIDIARY OF JOHNSON & JOHNSON SERVICES, INC.)
18.15.1 COMPANY SNAPSHOT
18.15.2 REVENUE ANALYSIS
18.15.3 PRODUCT PORTFOLIO
18.15.4 RECENT DEVELOPMENTS
18.16 LANZHOU BIOLOGICAL PRODUCTS RESEARCH INSTITUTE CO., LTD.,
18.16.1 COMPANY SNAPSHOT
18.16.2 PRODUCT PORTFOLIO
18.16.3 RECENT DEVELOPMENTS
18.17 PANACEA BIOTEC LTD
18.17.1 COMPANY SNAPSHOT
18.17.2 REVENUE ANALYSIS
18.17.3 PRODUCT PORTFOLIO
18.17.4 RECENT DEVELOPMENTS
18.18 SEQIRUS (A SUBSIDIARY OF CSL LIMITED)
18.18.1 COMPANY SNAPSHOT
18.18.2 REVENUE ANALYSIS
18.18.3 PRODUCT PORTFOLIO
18.18.4 RECENT DEVELOPMENTS
18.19 TAKEDA PHARMACEUTICAL COMPANY LIMITED
18.19.1 COMPANY SNAPSHOT
18.19.2 REVENUE ANALYSIS
18.19.3 COMPANY WEBSITE AND PRESS RELEASES
18.19.4 PRODUCT PORTFOLIO
18.19.5 RECENT DEVELOPMENTS
19 QUESTIONNAIRE
20 RELATED REPORTS
表のリスト
TABLE 1 ASIA-PACIFIC VACCINES MARKET, PIPELINE ANALYSIS
TABLE 2 RECOMMENDED TEMPERATURE AND STORAGE LENGTH AT VARIOUS LEVELS OF THE COLD CHAIN.
TABLE 3 LOGISTICS PROCESS ACROSS DIFFERENT REGIONS.
TABLE 4 LAWS AND REGULATIONS IN TAIWAN
TABLE 5 VACCINES UNDER CLINICAL TRIAL
TABLE 6 THE SIDE EFFECTS RELATED TO THE VACCINES
TABLE 7 ASIA-PACIFIC VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 8 ASIA-PACIFIC VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 9 ASIA-PACIFIC SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 10 ASIA-PACIFIC LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 11 ASIA-PACIFIC INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 12 ASIA-PACIFIC TOXOID VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 13 ASIA-PACIFIC VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 14 ASIA-PACIFIC ROUTINE VACCINES IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 15 ASIA-PACIFIC RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 16 ASIA-PACIFIC REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 17 ASIA-PACIFIC VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 18 ASIA-PACIFIC PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 19 ASIA-PACIFIC ADULT VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 20 ASIA-PACIFIC VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 21 ASIA-PACIFIC VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2022-2030 (USD MILLION)
TABLE 22 ASIA-PACIFIC INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2022-2030 (USD MILLION)
TABLE 23 ASIA-PACIFIC VACCINES MARKET, BY END USER, 2022-2030 (USD MILLION)
TABLE 24 ASIA-PACIFIC VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 25 ASIA-PACIFIC KNEE CARTILAGE REPAIR MARKET, BY COUNTRY, 2021-2030 (USD MILLION)
TABLE 26 JAPAN VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 27 JAPAN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 28 JAPAN SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 29 JAPAN LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 30 JAPAN INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 31 JAPAN TOXOID VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 32 JAPAN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 33 JAPAN ROUTINE VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 34 JAPAN RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 35 JAPAN REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 36 JAPAN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 37 JAPAN PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 38 JAPAN ADULT IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 39 JAPAN VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 40 JAPAN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 41 JAPAN INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 42 JAPAN VACCINES MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 43 JAPAN VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 44 CHINA VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 45 CHINA VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 46 CHINA SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 47 CHINA LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 48 CHINA INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 49 CHINA TOXOID VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 50 CHINA VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 51 CHINA ROUTINE VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 52 CHINA RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 53 CHINA REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 54 CHINA VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 55 CHINA PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 56 CHINA ADULT IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 57 CHINA VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 58 CHINA VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 59 CHINA INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 60 CHINA VACCINES MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 61 CHINA VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 62 AUSTRALIA VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 63 AUSTRALIA VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 64 AUSTRALIA SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 65 AUSTRALIA LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 66 AUSTRALIA INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 67 AUSTRALIA TOXOID VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 68 AUSTRALIA VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 69 AUSTRALIA ROUTINE VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 70 AUSTRALIA RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 71 AUSTRALIA REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 72 AUSTRALIA VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 73 AUSTRALIA PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 74 AUSTRALIA ADULT IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 75 AUSTRALIA VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 76 AUSTRALIA VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 77 AUSTRALIA INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 78 AUSTRALIA VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 79 INDIA VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 80 INDIA VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 81 INDIA SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 82 INDIA LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 83 INDIA INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 84 INDIA TOXOID VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 85 INDIA VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 86 INDIA ROUTINE VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 87 INDIA RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 88 INDIA REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 89 INDIA VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 90 INDIA PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 91 INDIA ADULT IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 92 INDIA VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 93 INDIA VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 94 INDIA INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 95 INDIA VACCINES MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 96 INDIA VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 97 SOUTH KOREA VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 98 SOUTH KOREA VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 99 SOUTH KOREA SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 100 SOUTH KOREA LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 101 SOUTH KOREA INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 102 SOUTH KOREA VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 103 SOUTH KOREA ROUTINE VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 104 SOUTH KOREA RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 105 SOUTH KOREA REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 106 SOUTH KOREA VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 107 SOUTH KOREA PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 108 SOUTH KOREA ADULT IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 109 SOUTH KOREA VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 110 SOUTH KOREA VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 111 SOUTH KOREA INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 112 SOUTH KOREA VACCINES MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 113 SOUTH KOREA VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 114 SINGAPORE VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 115 SINGAPORE SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 116 SINGAPORE LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 117 SINGAPORE INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 118 SINGAPORE TOXOID VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 119 SINGAPORE VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 120 SINGAPORE ROUTINE VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 121 SINGAPORE RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 122 SINGAPORE REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 123 SINGAPORE VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 124 SINGAPORE PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 125 SINGAPORE ADULT IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 126 SINGAPORE VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 127 SINGAPORE VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 128 SINGAPORE INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 129 SINGAPORE VACCINES MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 130 SINGAPORE VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 131 MALAYSIA VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 132 MALAYSIA VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 133 MALAYSIA SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 134 MALAYSIA LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 135 MALAYSIA INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 136 MALAYSIA TOXOID VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 137 MALAYSIA VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 138 MALAYSIA ROUTINE VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 139 MALAYSIA RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 140 MALAYSIA REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 141 MALAYSIA VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 142 MALAYSIA PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 143 MALAYSIA ADULT IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 144 MALAYSIA VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 145 MALAYSIA VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 146 MALAYSIA INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 147 MALAYSIA VACCINES MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 148 MALAYSIA VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 149 THAILAND VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 150 THAILAND VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 151 THAILAND SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 152 THAILAND LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 153 THAILAND INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 154 THAILAND TOXOID VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 155 THAILAND VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 156 THAILAND RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 157 THAILAND REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 158 THAILAND VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 159 THAILAND PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 160 THAILAND ADULT IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 161 THAILAND VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 162 THAILAND VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 163 THAILAND INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 164 THAILAND VACCINES MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 165 THAILAND VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 166 INDONESIA VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 167 INDONESIA VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 168 INDONESIA SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 169 INDONESIA LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 170 INDONESIA INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 171 INDONESIA TOXOID VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 172 INDONESIA VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 173 INDONESIA ROUTINE VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 174 INDONESIA RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 175 INDONESIA REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 176 INDONESIA VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 177 INDONESIA PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 178 INDONESIA ADULT IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 179 INDONESIA VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 180 INDONESIA VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 181 INDONESIA INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 182 INDONESIA VACCINES MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 183 INDONESIA VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 184 PHILIPPINES VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 185 PHILIPPINES VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 186 PHILIPPINES SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 187 PHILIPPINES LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 188 PHILIPPINES INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 189 PHILIPPINES TOXOID VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 190 PHILIPPINES VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 191 PHILIPPINES ROUTINE VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 192 PHILIPPINES RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 193 PHILIPPINES REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 194 PHILIPPINES VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 195 PHILIPPINES PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 196 PHILIPPINES ADULT IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 197 PHILIPPINES VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 198 PHILIPPINES VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 199 PHILIPPINES INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 200 PHILIPPINES VACCINES MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 201 PHILIPPINES VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 202 VIETNAM VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
TABLE 203 VIETNAM VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 204 VIETNAM SUBUNIT, RECOMBINANT, POLYSACCHARIDE & CONJUGATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 205 VIETNAM LIVE-ATTENUATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 206 VIETNAM INACTIVATED VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 207 VIETNAM TOXOID VACCINES IN VACCINES MARKET, BY TYPE, 2021-2030 (USD MILLION)
TABLE 208 VIETNAM VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 209 VIETNAM ROUTINE VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 210 VIETNAM RECOMMENDED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 211 VIETNAM REQUIRED VACCINE IN VACCINES MARKET, BY KIND, 2021-2030 (USD MILLION)
TABLE 212 VIETNAM VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 213 VIETNAM PEDIATRIC VACCINE IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 214 VIETNAM ADULT IN VACCINES MARKET, BY AGE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 215 VIETNAM VACCINES MARKET, BY DISEASE, 2021-2030 (USD MILLION)
TABLE 216 VIETNAM VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 217 VIETNAM INJECTABLE IN VACCINES MARKET, BY ROUTE OF ADMINISTRATION, 2021-2030 (USD MILLION)
TABLE 218 VIETNAM VACCINES MARKET, BY END USER, 2021-2030 (USD MILLION)
TABLE 219 VIETNAM VACCINES MARKET, BY DISTRIBUTION CHANNEL, 2021-2030 (USD MILLION)
TABLE 220 REST OF ASIA-PACIFIC VACCINES MARKET, BY COMPOSITION, 2021-2030 (USD MILLION)
図表一覧
FIGURE 1 ASIA-PACIFIC VACCINES MARKET: SEGMENTATION
FIGURE 2 ASIA-PACIFIC VACCINES MARKET: DATA TRIANGULATION
FIGURE 3 ASIA-PACIFIC VACCINES MARKET: DROC ANALYSIS
FIGURE 4 ASIA-PACIFIC VACCINES MARKET: ASIA-PACIFIC VS COUNTRY MARKET ANALYSIS
FIGURE 5 ASIA-PACIFIC VACCINES MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 ASIA-PACIFIC VACCINES MARKET: MULTIVARIATE MODELLING
FIGURE 7 ASIA-PACIFIC VACCINES MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 8 ASIA-PACIFIC VACCINES MARKET: DBMR MARKET POSITION GRID
FIGURE 9 ASIA-PACIFIC VACCINES MARKET: SEGMENTATION
FIGURE 10 GROWING IMMUNIZATION PROGRAMS AND CAMPAIGNS AND THE HIGH PREVALENCE OF CHRONIC CONDITIONS SUCH AS FLU AND BACTERIAL INFECTIOUS DISEASES ARE DRIVING THE ASIA-PACIFIC VACCINES MARKET IN THE FORECAST PERIOD OF 2023 TO 2030
FIGURE 11 THE COMBINATION VACCINES SEGMENT IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE ASIA-PACIFIC VACCINES MARKET IN 2023 & 2030
FIGURE 12 FDA REGULATORY REVIEW PROCESS OF VACCINES
FIGURE 13 PROCESS OF SPECIAL APPROVAL ON VACCINES DURING THE 2019 H1N1PDM PANDEMIC
FIGURE 14 REGULATION OVERVIEW FOR THERAPEUTICS IN SINGAPORE
FIGURE 15 DRIVERS, RESTRAINTS, OPPORTUNITIES AND CHALLENGES OF THE ASIA-PACIFIC VACCINES MARKET
FIGURE 16 ASIA-PACIFIC VACCINES MARKET: BY COMPOSITION, 2022
FIGURE 17 ASIA-PACIFIC VACCINES MARKET: BY COMPOSITION, 2023-2030 (USD MILLION)
FIGURE 18 ASIA-PACIFIC VACCINES MARKET: BY COMPOSITION, CAGR (2023-2030)
FIGURE 19 ASIA-PACIFIC VACCINES MARKET: BY COMPOSITION, LIFELINE CURVE
FIGURE 20 ASIA-PACIFIC VACCINES MARKET: BY TYPE, 2022
FIGURE 21 ASIA-PACIFIC VACCINES MARKET: BY TYPE, 2023-2030 (USD MILLION)
FIGURE 22 ASIA-PACIFIC VACCINES MARKET: BY TYPE, CAGR (2023-2030)
FIGURE 23 ASIA-PACIFIC VACCINES MARKET: BY TYPE, LIFELINE CURVE
FIGURE 24 ASIA-PACIFIC VACCINES MARKET: BY KIND, 2022
FIGURE 25 ASIA-PACIFIC VACCINES MARKET: BY KIND, 2023-2030 (USD MILLION)
FIGURE 26 ASIA-PACIFIC VACCINES MARKET: BY KIND, CAGR (2023-2030)
FIGURE 27 ASIA-PACIFIC VACCINES MARKET: BY KIND, LIFELINE CURVE
FIGURE 28 ASIA-PACIFIC VACCINES MARKET: BY AGE OF ADMINISTRATION, 2022
FIGURE 29 ASIA-PACIFIC VACCINES MARKET: BY AGE OF ADMINISTRATION, 2023-2030 (USD MILLION)
FIGURE 30 ASIA-PACIFIC VACCINES MARKET: BY AGE OF ADMINISTRATION, CAGR (2023-2030)
FIGURE 31 ASIA-PACIFIC VACCINES MARKET: BY AGE OF ADMINISTRATION, LIFELINE CURVE
FIGURE 32 ASIA-PACIFIC VACCINES MARKET: BY DISEASES, 2022
FIGURE 33 ASIA-PACIFIC VACCINES MARKET: BY DISEASES, 2023-2030 (USD MILLION)
FIGURE 34 ASIA-PACIFIC VACCINES MARKET: BY DISEASES, CAGR (2023-2030)
FIGURE 35 ASIA-PACIFIC VACCINES MARKET: BY DISEASES, LIFELINE CURVE
FIGURE 36 ASIA-PACIFIC VACCINES MARKET: BY ROUTE OF ADMINISTRATION, 2022
FIGURE 37 ASIA-PACIFIC VACCINES MARKET: BY ROUTE OF ADMINISTRATION, 2023-2030 (USD MILLION)
FIGURE 38 ASIA-PACIFIC VACCINES MARKET: BY ROUTE OF ADMINISTRATION, CAGR (2023-2030)
FIGURE 39 ASIA-PACIFIC VACCINES MARKET: BY ROUTE OF ADMINISTRATION, LIFELINE CURVE
FIGURE 40 ASIA-PACIFIC VACCINES MARKET: BY END USER, 2022
FIGURE 41 ASIA-PACIFIC VACCINES MARKET: BY END USER, 2023-2030 (USD MILLION)
FIGURE 42 ASIA-PACIFIC VACCINES MARKET: BY END USER, CAGR (2023-2030)
FIGURE 43 ASIA-PACIFIC VACCINES MARKET: BY END USER, LIFELINE CURVE
FIGURE 44 ASIA-PACIFIC VACCINES MARKET: BY DISTRIBUTION CHANNEL, 2022
FIGURE 45 ASIA-PACIFIC VACCINES MARKET: BY DISTRIBUTION CHANNEL, 2023-2030 (USD MILLION)
FIGURE 46 ASIA-PACIFIC VACCINES MARKET: BY DISTRIBUTION CHANNEL, CAGR (2023-2030)
FIGURE 47 ASIA-PACIFIC VACCINES MARKET: BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 48 ASIA-PACIFIC VACCINE MARKET: SNAPSHOT (2022)
FIGURE 49 ASIA-PACIFIC VACCINE MARKET: BY COUNTRY (2022)
FIGURE 50 ASIA-PACIFIC VACCINE MARKET: BY COUNTRY (2023 & 2030)
FIGURE 51 ASIA-PACIFIC VACCINE MARKET: BY COUNTRY (2022 & 2030)
FIGURE 52 ASIA-PACIFIC VACCINE MARKET: BY COMPOSITION (2023-2030)
FIGURE 53 ASIA-PACIFIC VACCINES MARKET: COMPANY SHARE 2022 (%)
FIGURE 54 PFIZER, INC., ASIA-PACIFIC VACCINES MARKET: DISEASE SHARE 2022 (%)
FIGURE 55 PFIZER, INC., ASIA-PACIFIC VACCINES MARKET: COUNTRY SHARE 2022 (%)
FIGURE 56 MERCK SHARP & DOHME CORP. (A SUBSIDIARY OF MERCK & CO., INC.) ASIA-PACIFIC VACCINES MARKET: DISEASE SHARE 2022 (%)
FIGURE 57 MERCK SHARP & DOHME CORP. (A SUBSIDIARY OF MERCK & CO., INC.) ASIA-PACIFIC VACCINES MARKET: COUNTRY SHARE 2022 (%)
FIGURE 58 GLAXOSMITHKLINE PLC, ASIA-PACIFIC VACCINES MARKET: DISEASE SHARE 2022 (%)
FIGURE 59 GLAXOSMITHKLINE PLC, ASIA-PACIFIC VACCINES MARKET: COMPANY SHARE 2022 (%)
FIGURE 60 SANOFI, ASIA-PACIFIC VACCINES MARKET: DISEASE SHARE 2022 (%)
FIGURE 61 SANOFI ASIA-PACIFIC VACCINES MARKET: COUNTRY SHARE 2022 (%)
FIGURE 62 SERUM INSTITUTE OF INDIA PVT. LTD., ASIA-PACIFIC VACCINES MARKET: DISEASE SHARE 2022 (%)
FIGURE 63 SERUM INSTITUTE OF INDIA PVT. LTD. ASIA-PACIFIC VACCINES MARKET: COUNTRY SHARE 2022 (%)

調査方法
データ収集と基準年分析は、大規模なサンプル サイズのデータ収集モジュールを使用して行われます。この段階では、さまざまなソースと戦略を通じて市場情報または関連データを取得します。過去に取得したすべてのデータを事前に調査および計画することも含まれます。また、さまざまな情報ソース間で見られる情報の不一致の調査も含まれます。市場データは、市場統計モデルと一貫性モデルを使用して分析および推定されます。また、市場シェア分析と主要トレンド分析は、市場レポートの主要な成功要因です。詳細については、アナリストへの電話をリクエストするか、お問い合わせをドロップダウンしてください。
DBMR 調査チームが使用する主要な調査方法は、データ マイニング、データ変数が市場に与える影響の分析、および一次 (業界の専門家) 検証を含むデータ三角測量です。データ モデルには、ベンダー ポジショニング グリッド、市場タイムライン分析、市場概要とガイド、企業ポジショニング グリッド、特許分析、価格分析、企業市場シェア分析、測定基準、グローバルと地域、ベンダー シェア分析が含まれます。調査方法について詳しくは、お問い合わせフォームから当社の業界専門家にご相談ください。
カスタマイズ可能
Data Bridge Market Research は、高度な形成的調査のリーダーです。当社は、既存および新規のお客様に、お客様の目標に合致し、それに適したデータと分析を提供することに誇りを持っています。レポートは、対象ブランドの価格動向分析、追加国の市場理解 (国のリストをお問い合わせください)、臨床試験結果データ、文献レビュー、リファービッシュ市場および製品ベース分析を含めるようにカスタマイズできます。対象競合他社の市場分析は、技術ベースの分析から市場ポートフォリオ戦略まで分析できます。必要な競合他社のデータを、必要な形式とデータ スタイルでいくつでも追加できます。当社のアナリスト チームは、粗い生の Excel ファイル ピボット テーブル (ファクト ブック) でデータを提供したり、レポートで利用可能なデータ セットからプレゼンテーションを作成するお手伝いをしたりすることもできます。